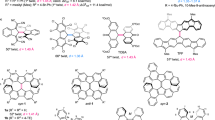
Abstract
IT has recently been shown1 by a consideration of the molecular resonance properties associated with various structural models that the observed2 centro-symmetrical nature of the hexadiene-Å"strol molecule (ppâ²-dihyaroxy-3 : 4-diphenyl-2 : 4-hexadiene) [I] requires the hexadiene chain to exist in the planar trans-trans-configuration shown below. The two benzene rings are probably rotated by some 50° out of the hexadiene plane in order to reduce the steric interference of the ortho hydrogen atoms. Similar arguments lead to a non-coplanar structure of stilbÅ"strol (ppâ²-dihydroxy-αβ-trans-diethylstilbene [II], in which the conformation of the aliphatic hexene chain must be expected to approximate to a zig-zag shape rather than to the S-form [III], the latter offering much greater steric opposition to the resonance-induced tendency of the stilbene skeleton to attain full coplanarity.
Similar content being viewed by others
Article PDF
References
Jeffrey, Koch, and Nyburg, J. Chem. Soc. (in the press).
Carlisle and Crowfoot, J. Chem. Soc., 6 (1941).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KOCH, H. Configuration of Synthetic Å‘strogens. Nature 161, 309 (1948). https://doi.org/10.1038/161309a0
Issue Date:
DOI: https://doi.org/10.1038/161309a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.