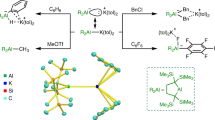
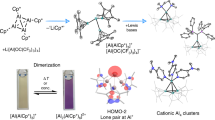
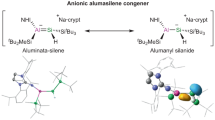
Abstract
THE existence of the dimer aluminium trimethyl has aroused considerable interest. Electron diffraction determinations1,2 are in satisfactory agreement with an ethane structure (I). However, this structure is for many reasons most unlikely: (1) it should differ from the bridge structures of the related aluminium halides3 Al2X6, aluminium dimethyl-halides2 Al2Me4X2 as well as of the boron and probably aluminium hydrides4; (2) it cannot be accounted for by any existing valence theory; no forces are available for joining the two AlMe3 molecules; (3) it requires a shorter interatomic distance (about 2·20 A.) between the equally charged Al atoms than that of a covalent Al–Al linkage (>2·48 A.); (4) the instability (non-existence) of a dimer boron trimethyl cannot be explained.
Similar content being viewed by others
Article PDF
References
Davidson, Hugill, Skinner and Sutton, Trans. Far. Soc., 36, 1212 (1940).
Broackway and Davidson, J. Amer. Chem. Soc., 63, 3287 (1941).
Palmer and Elliot, J. Amer. Chem. Soc., 60, 1852 (1938).
Longuet-Higgins and Bell, J. Chem. Soc., 250 (1943).
Kohlrausch and Wagner, Z. phys. Chem., B, 52, 153 (1942) (from Brit. Chem. Abstr., A I, 50 (1943)).
Burawoy, Nature, in the press.
Burawoy, Gibson and others, J. Chem. Soc., 860 (1934); 217, 219, 1024 (1935); 1690 (1937): cf. also Phillips and Powell, Proc. Roy. Soc., A, 173. 147 (1939).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BURAWOY, A. Structure of Aluminium Trimethyl. Nature 155, 269 (1945). https://doi.org/10.1038/155269a0
Issue Date:
DOI: https://doi.org/10.1038/155269a0
This article is cited by
-
Aluminium Trimethyl
Nature (1945)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.