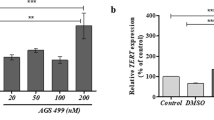
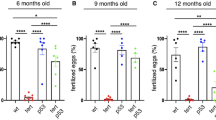
Abstract
The expression level of the telomerase catalytic subunit (telomerase reverse transcriptase, TERT) positively correlates with cell survival after exposure to several lethal stresses. However, whether the protective role of TERT is independent of telomerase activity has not yet been clearly explored. Here, we genetically evaluated the protective roles of both TERT and telomerase activity against cell death induced by staurosporine (STS) and N-methyl-D-aspartic acid (NMDA). First generation (G1) TERT-deficient mouse embryonic fibroblasts (MEFs) displayed an increased sensitivity to STS, while TERT transgenic MEFs were more resistant to STS-induced apoptosis than wild-type. Deletion of the telomerase RNA component (TERC) failed to alter the sensitivity of TERT transgenic MEFs to STS treatment. Similarly, NMDA-induced excitotoxic cell death of primary neurons was suppressed by TERT, but not by TERC both in vitro and in vivo. Specifically, NMDA accelerated death of TERT-deficient mice, while TERT transgenic mice showed enhanced survival when compared with wild-type littermates after administration of NMDA. In addition, the transgenic expression of TERT protected motor neurons from apoptosis induced by sciatic nerve axotomy. These results indicate that telomerase activity is not essential for the protective function of TERT. This telomerase activity-independent TERT function may contribute to cancer development and aging independently of telomere lengthening.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Artandi SE, Alson S, Tietze MK, Sharpless NE, Ye S, Greenberg RA et al. (2002). Constitutive telomerase expression promotes mammary carcinomas in aging mice. Proc Natl Acad Sci USA 99: 8191–8196.
Autexier C, Pruzan R, Funk WD, Greider CW . (1996). Reconstitution of human telomerase activity and identification of a minimal functional region of the human telomerase RNA. EMBO J 15: 5928–5935.
Beattie TL, Zhou W, Robinson MO, Harrington L . (1998). Reconstitution of human telomerase activity in vitro. Curr Biol 8: 177–180.
Blackburn EH . (2001). Switching and signaling at the telomere. Cell 106: 661–673.
Blasco MA, Funk W, Villeponteau B, Greider CW . (1995). Functional characterization and developmental regulation of mouse telomerase RNA. Science 269: 1267–1270.
Blasco MA, Lee HW, Hande MP, Samper E, Lansdorp PM, DePinho RA et al. (1997). Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91: 25–34.
Boklan J, Nanjangud G, MacKenzie KL, May C, Sadelain M, Moore MA . (2002). Limited proliferation and telomere dysfunction following telomerase inhibition in immortal murine fibroblasts. Cancer Res 62: 2104–2114.
Cao Y, Li H, Deb S, Liu JP . (2002). TERT regulates cell survival independent of telomerase enzymatic activity. Oncogene 21: 3130–3138.
Cerezo A, Kalthoff H, Schuermann M, Schafer B, Boukamp P . (2002). Dual regulation of telomerase activity through c-Myc-dependent inhibition and alternative splicing of hTERT. J Cell Sci 115: 1305–1312.
Chen JL, Greider CW . (2003). Template boundary definition in mammalian telomerase. Genes Dev 17: 2747–2752.
Chung HK, Cheong C, Song J, Lee HW . (2005). Extratelomeric functions of telomerase. Curr Mol Med 5: 233–241.
Collado M, Blasco MA, Serrano M . (2007). Cellular senescence in cancer and aging. Cell 130: 223–233.
Counter CM, Hahn WC, Wei W, Caddle SD, Beijersbergen RL, Lansdorp PM et al. (1998). Dissociation among in vitro telomerase activity, telomere maintenance, and cellular immortalization. Proc Natl Acad Sci USA 95: 14723–14728.
Deckwerth TL, Elliott JL, Knudson CM, Johnson Jr EM, Snider WD, Korsmeyer SJ . (1996). BAX is required for neuronal death after trophic factor deprivation and during development. Neuron 17: 401–411.
Deveraux QL, Takahashi R, Salvesen GS, Reed JC . (1997). X-linked IAP is a direct inhibitor of cell-death proteases. Nature 388: 300–304.
Djebaili M, Rondouin G, Baille V, Bockaert J . (2000). p53 and Bax implication in NMDA induced-apoptosis in mouse hippocampus. Neuroreport 11: 2973–2976.
Dubois-Dauphin M, Frankowski H, Tsujimoto Y, Huarte J, Martinou JC . (1994). Neonatal motoneurons overexpressing the bcl-2 protooncogene in transgenic mice are protected from axotomy-induced cell death. Proc Natl Acad Sci USA 91: 3309–3313.
Erdmann N, Liu Y, Harrington L . (2004). Distinct dosage requirements for the maintenance of long and short telomeres in mTert heterozygous mice. Proc Natl Acad Sci USA 101: 6080–6085.
Finkel T, Serrano M, Blasco MA . (2007). The common biology of cancer and ageing. Nature 448: 767–774.
Folini M, Brambilla C, Villa R, Gandellini P, Vignati S, Paduano F et al. (2005). Antisense oligonucleotide-mediated inhibition of hTERT, but not hTERC, induces rapid cell growth decline and apoptosis in the absence of telomere shortening in human prostate cancer cells. Eur J Cancer 41: 624–634.
Greenberg RA, Allsopp RC, Chin L, Morin GB, DePinho RA . (1998). Expression of mouse telomerase reverse transcriptase during development, differentiation and proliferation. Oncogene 16: 1723–1730.
Hahn WC, Stewart SA, Brooks MW, York SG, Eaton E, Kurachi A et al. (1999). Inhibition of telomerase limits the growth of human cancer cells. Nat Med 5: 1164–1170.
Kang HJ, Choi YS, Hong SB, Kim KW, Woo RS, Won SJ et al. (2004). Ectopic expression of the catalytic subunit of telomerase protects against brain injury resulting from ischemia and NMDA-induced neurotoxicity. J Neurosci 24: 1280–1287.
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL et al. (1994). Specific association of human telomerase activity with immortal cells and cancer. Science 266: 2011–2015.
Lakhani SA, Masud A, Kuida K, Porter Jr GA, Booth CJ, Mehal WZ et al. (2006). Caspases 3 and 7: key mediators of mitochondrial events of apoptosis. Science 311: 847–851.
Lee HW, Blasco MA, Gottlieb GJ, Horner II JW, Greider CW, DePinho RA . (1998). Essential role of mouse telomerase in highly proliferative organs. Nature 392: 569–574.
Li K, Li Y, Shelton JM, Richardson JA, Spencer E, Chen ZJ et al. (2000). Cytochrome c deficiency causes embryonic lethality and attenuates stress-induced apoptosis. Cell 101: 389–399.
Massard C, Zermati Y, Pauleau AL, Larochette N, Métivier D, Sabatier L et al. (2006). hTERT: a novel endogenous inhibitor of the mitochondrial cell death pathway. Oncogene 25: 4505–4514.
McEachern MJ, Krauskopf A, Blackburn EH . (2000). Telomeres and their control. Annu Rev Genet 34: 331–358.
Middleman EJ, Choi J, Venteicher AS, Cheung P, Artandi SE . (2006). Regulation of cellular immortalization and steady-state levels of the telomerase reverse transcriptase through its carboxy-terminal domain. Mol Cell Biol 26: 2146–2159.
Morales CP, Holt SE, Ouellette M, Kaur KJ, Yan Y, Wilson KS et al. (1999). Absence of cancer-associated changes in human fibroblasts immortalized with telomerase. Nat Genet 21: 115–118.
Murphy AN, Bredesen DE, Cortopassi G, Wang E, Fiskum G . (1996). Bcl-2 potentiates the maximal calcium uptake capacity of neural cell mitochondria. Proc Natl Acad Sci USA 93: 9893–9898.
Nakayama J, Tahara H, Tahara E, Saito M, Ito K, Nakamura H et al. (1998). Telomerase activation by hTRT in human normal fibroblasts and hepatocellular carcinomas. Nat Genet 18: 65–68.
Perez-Navarro E, Gavalda N, Gratacos E, Alberch J . (2005). Brain-derived neurotrophic factor prevents changes in Bcl-2 family members and caspase-3 activation induced by excitotoxicity in the striatum. J Neurochem 92: 678–691.
Rahman R, Latonen L, Wiman KG . (2005). hTERT antagonizes p53-induced apoptosis independently of telomerase activity. Oncogene 24: 1320–1327.
Sarin KY, Cheung P, Gilison D, Lee E, Tennen RI, Wang E et al. (2005). Conditional telomerase induction causes proliferation of hair follicle stem cells. Nature 436: 1048–1052.
Seleznev K, Zhao C, Zhang XH, Song K, Ma ZA . (2006). Calcium-independent phospholipase A2 localizes in and protects mitochondria during apoptotic induction by staurosporine. J Biol Chem 281: 22275–22288.
Shay JW, Zou Y, Hiyama E, Wright WE . (2001). Telomerase and cancer. Hum Mol Genet 10: 677–685.
Siegl-Cachedenier I, Flores I, Klatt P, Blasco MA . (2007). Telomerase reverses epidermal hair follicle stem cell defects and loss of long-term survival associated with critically short telomeres. J Cell Biol 179: 277–290.
Sofia RD, Gordon R, Gels M, Diamantis W . (1994). Comparative effects of felbamate and other compounds on N-methyl-D-aspartic acid-induced convulsions and lethality in mice. Pharmacol Res 29: 139–144.
Sun W, Gould TW, Vinsant S, Prevette D, Oppenheim RW . (2003). Neuromuscular development after the prevention of naturally occurring neuronal death by Bax deletion. J Neurosci 23: 7298–7310.
Sung YH, Choi YS, Cheong C, Lee HW . (2005). The pleiotropy of telomerase against cell death. Mol Cells 19: 303–309.
Wallace DC . (2005). A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: a dawn for evolutionary medicine. Annu Rev Genet 39: 359–407.
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ et al. (2001). Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292: 727–730.
Wong KK, Chang S, Weiler SR, Ganesan S, Chaudhuri J, Zhu C et al. (2000). Telomere dysfunction impairs DNA repair and enhances sensitivity to ionizing radiation. Nat Genet 26: 85–88.
Wu YL, Dudognon C, Nguyen E, Hillion J, Pendino F, Tarkanyi I et al. (2006). Immunodetection of human telomerase reverse-transcriptase (hTERT) re-appraised: nucleolin and telomerase cross paths. J Cell Sci 119: 2797–2806.
Yi X, White DM, Aisner DL, Baur JA, Wright WE, Shay JW . (2000). An alternate splicing variant of the human telomerase catalytic subunit inhibits telomerase activity. Neoplasia 2: 433–440.
Yuan X, Ishibashi S, Hatakeyama S, Saito M, Nakayama J, Nikaido R et al. (1999). Presence of telomeric G-strand tails in the telomerase catalytic subunit TERT knockout mice. Genes Cells 4: 563–572.
Zhang P, Chan SL, Fu W, Mendoza M, Mattson MP . (2003). TERT suppresses apoptotis at a premitochondrial step by a mechanism requiring reverse transcriptase activity and 14-3-3 protein-binding ability. FASEB J 17: 767–769.
Acknowledgements
This work was supported by 21C Frontier Projects (Functional Human Genome Project, M106KB010014-07K0201-01410 and Brain Research Center, M103KV010025-07K2201-02510) from the MOST, SRC grant (MTRC, R11-2000-080-11004-0) from KOSEF, MOEHRD and the US National Institute for Aging (R01 AG23145, WCH). YHS is supported by BK21 of Yonsei University from MOEHRD.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Lee, J., Sung, Y., Cheong, C. et al. TERT promotes cellular and organismal survival independently of telomerase activity. Oncogene 27, 3754–3760 (2008). https://doi.org/10.1038/sj.onc.1211037
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1211037
Keywords
This article is cited by
-
Knock-down of LRP/LR influences signalling pathways in late-stage colorectal carcinoma cells
BMC Cancer (2021)
-
Telomerase reverse transcriptase preserves neuron survival and cognition in Alzheimer’s disease models
Nature Aging (2021)
-
Telomerase activation in the treatment of aging or degenerative diseases: a systematic review
Molecular and Cellular Biochemistry (2021)
-
Aqueous extract of Phragmites communis rhizomes attenuates phototoxicity in skin cells
Molecular & Cellular Toxicology (2021)
-
Human telomerase reverse transcriptase positively regulates mitophagy by inhibiting the processing and cytoplasmic release of mitochondrial PINK1
Cell Death & Disease (2020)