Abstract
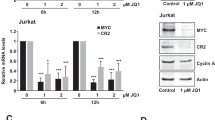
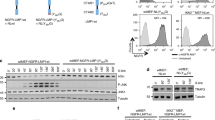
Rel/NF-κB transcription factors are critical arbiters of immune responses, cell survival, and transformation, and are frequently deregulated in cancer. The p50 NF-κB1 component of Rel/NF-κB DNA-binding dimers regulates genes involved in both cell cycle traverse and apoptosis. Nfkb1 loss accelerates B cell growth and leads to increased B cell turnover in vivo, phenotypes akin to those manifested in B cells of Eμ-Myc transgenic mice, a model of human Burkitt lymphoma. Interestingly, Eμ-Myc B cells express reduced levels of cytoplasmic and nuclear NF-κB1 and have reduced Rel/NF-κB DNA-binding activity, suggesting that Myc-mediated repression of NF-κB1 might mediate its proliferative and apoptotic effects on B cells. Furthermore, Nfkb1 expression was reduced in the majority of Eμ-Myc lymphomas and was also suppressed in human Burkitt lymphoma. Nonetheless, loss of Nfkb1 did not appreciably affect Myc's proliferative or apoptotic responses in B cells and had no effect on lymphoma development in Eμ-Myc mice. Therefore, Nfkb1 is dispensable for Myc-induced lymphomagenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Adams JM, Harris AW, Pinkert CA, Corcoran LM, Alexander WS, Cory S, Palmiter RD and Brinster RL . (1985). Nature, 318, 533–538.
Alt JR, Greiner TC, Cleveland JL and Eischen CM . (2003). EMBO J., 22, 1442–1450.
Askew DS, Ashmun RA, Simmons BC and Cleveland JL . (1991). Oncogene, 6, 1915–1922.
Baldwin AS . (2001). J. Clin. Invest., 107, 241–246.
Baudino TA, Maclean KH, Brennan J, Parganas E, Yang C, Aslanian A, Lees JA, Sherr CJ, Roussel MF and Cleveland JL . (2003). Mol. Cell, 11, 905–914.
Bello-Fernandez C, Packham G and Cleveland JL . (1993). Proc. Natl. Acad. Sci. USA, 90, 7804–7808.
Bouchard C, Staller P and Eilers M . (1998). Trends Cell Biol., 8, 202–206.
Caamano JH, Rizzo CA, Durham SK, Barton DS, Raventos-Suarez C, Snapper CM and Bravo R . (1998). J. Exp. Med., 187, 185–196.
Cavallieri F and Goldfarb M . (1987). Mol. Cell. Biol., 7, 3554–3560.
Coppola JA and Cole MD . (1986). Nature, 320, 760–763.
de Alboran IM, O'Hagan RC, Gartner F, Malynn B, Davidson L, Rickert R, Rajewsky K, DePinho RA and Alt FW . (2001). Immunity, 14, 45–55.
Egle A, Harris AW, Bouillet P and Cory S . (2004). Proc. Natl. Acad. Sci. USA, 101, 6164–6169.
Eischen CM, Weber JD, Roussel MF, Sherr CJ and Cleveland JL . (1999). Genes Dev., 13, 2658–2669.
Eischen CM, Woo D, Roussel MF and Cleveland JL . (2001). Mol. Cell. Biol., 21, 5063–5070.
Evan GI and Vousden KH . (2001). Nature, 411, 342–348.
Evan GI, Wyllie AH, Gilbert CS, Littlewood TD, Land H, Brooks M, Waters CM, Penn LZ and Hancock DC . (1992). Cell, 69, 119–128.
Gerondakis S, Grossmann M, Nakamura Y, Pohl T and Grumont R . (1999). Oncogene, 18, 6888–6895.
Ghosh S and Karin M . (2002). Cell, 109, S81–S96.
Grandori C, Cowley SM, James LP and Eisenman RN . (2000). Annu. Rev. Cell. Dev. Biol., 16, 653–699.
Grumont RJ, Rourke IJ, O'Reilly LA, Strasser A, Miyake K, Sha W and Gerondakis S . (1998). J. Exp. Med., 187, 663–674.
Grumont RJ, Strasser A and Gerondakis S . (2002). Mol. Cell, 10, 1283–1294.
Hanahan D and Weinberg RA . (2000). Cell, 100, 57–70.
Hayden MS and Ghosh S . (2004). Genes Dev., 18, 2195–2224.
Jeffers JR, Parganas E, Lee Y, Yang C, Wang J, Brennan J, MacLean KH, Han J, Chittenden T, Ihle JN, McKinnon PJ, Cleveland JL and Zambetti GP . (2003). Cancer Cell, 4, 321–328.
Kanda K, Hu H-M, Zhang L, Grandchamps J and Boxer LM . (2000). J. Biol. Chem., 275, 32338–32346.
Klefstrom J, Arighi E, Littlewood T, Jaattela M, Saksela E, Evan GI and Alitalo K . (1997). EMBO J., 16, 7382–7392.
Lee H, Arsura M, Wu M, Duyao M, Buckler AJ and Sonenshein GE . (1995). J. Exp. Med., 181, 1169–1177.
Lewis BC, Shim H, Li Q, Wu CS, Lee LA, Maity A and Dang CV . (1997). Mol. Cell. Biol., 17, 4967–4978.
Liou HC, Sha WC, Scott ML and Baltimore D . (1994). Mol. Cell. Biol., 14, 5349–5359.
Maclean KH, Keller UB, Rodriguez-Galindo C, Nilsson JA and Cleveland JL . (2003). Mol. Cell. Biol., 23, 7256–7270.
Martins CP and Berns A . (2002). EMBO J., 21, 3739–3748.
Nilsson JA and Cleveland JL . (2003). Oncogene, 22, 9007–9021.
Orlowski RZ and Baldwin AS . (2002). Trends Mol Med., 8, 385–389.
Owyang AM, Tumang JR, Schram BR, Hsia CY, Behrens TW, Rothstein TL and Liou HC . (2001). J. Immunol., 167, 4948–4956.
Perkins ND . (2004). Trends Cell. Biol., 14, 64–69.
Rocha S, Campbell KJ and Perkins ND . (2003). Mol. Cell, 12, 15–25.
Schmitt CA, McCurrach ME, de Stanchina E, Wallace-Brodeur RR and Lowe SW . (1999). Genes Dev., 13, 2670–2677.
Sha WC, Liou HC, Tuomanen EI and Baltimore D . (1995). Cell, 80, 321–330.
Strasser A, Harris AW, Bath ML and Cory S . (1990). Nature, 348, 331–333.
Sun Z, Arendt CW, Ellmeier W, Schaeffer EM, Sunshine MJ, Gandhi L, Annes J, Petrzilka D, Kupfer A, Schwartzberg PL and Littman DR . (2000). Nature, 404, 402–407.
Tanaka H, Matsumura I, Ezoe S, Satoh Y, Sakamaki T, Albanese C, Machii T, Pestell RG and Kanakura Y . (2002). Mol. Cell, 9, 1017–1029.
Trumpp A, Refaeli Y, Oskarsson T, Gasser S, Murphy M, Martin GR and Bishop JM . (2001). Nature, 414, 768–773.
Wu M, Lee H, Bellas RE, Schauer SL, Arsura M, Katz D, FitzGerald MJ, Rothstein TL, Sherr DH and Sonenshein GE . (1996). EMBO J., 15, 4682–4690.
You Z, Madrid LV, Saims D, Sedivy J and Wang CY . (2002). J. Biol. Chem., 277, 36671–36677.
Zelazowski P, Carrasco D, Rosas FR, Moorman MA, Bravo R and Snapper CM . (1997). J. Immunol., 159, 3133–3139.
Zindy F, Eischen CM, Randle DH, Kamijo T, Cleveland JL, Sherr CJ and Roussel MF . (1998). Genes Dev., 12, 2424–2433.
Zindy F, Williams RT, Baudino TA, Rehg JE, Skapek SX, Cleveland JL, Roussel MF and Sherr CJ . (2003). Proc. Natl. Acad. Sci. USA, 100, 15930–15935.
Acknowledgements
We thank Sara Norton, Chunying Yang and Elsie White for expert technical assistance. We also thank Mihaela Onciu and John Sandlund for providing samples from Burkitt lymphoma patients, the Animal Resource Center, the Hartwell Center and the FACS Core Facility of St Jude Children's Research Hospital. This work was supported by NIH Grant CA76379 (JLC), the Cancer Center (CORE) support Grant CA21765 and by the American Lebanese Syrian Associated Charities (ALSAC) of St Jude Children's Research Hospital. UK was supported by the Deutsche Forschungsgemeinschaft (grant KE222/5-1). JBO was supported by NRSA Grant F32 CA099478, and JAN is the recipient of the George J Mitchell endowed fellowship from ALSAC.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Keller, U., Nilsson, J., Maclean, K. et al. Nfkb1 is dispensable for Myc-induced lymphomagenesis. Oncogene 24, 6231–6240 (2005). https://doi.org/10.1038/sj.onc.1208779
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208779
Keywords
This article is cited by
-
Bptf determines oncogenic addiction in aggressive B-cell lymphomas
Oncogene (2020)
-
The NF-κB subunit c-Rel regulates Bach2 tumour suppressor expression in B-cell lymphoma
Oncogene (2016)
-
BET bromodomain inhibitors synergize with ATR inhibitors to induce DNA damage, apoptosis, senescence-associated secretory pathway and ER stress in Myc-induced lymphoma cells
Oncogene (2016)
-
The druggability of intracellular nucleotide-degrading enzymes
Cancer Chemotherapy and Pharmacology (2016)
-
Nfkb1 is a haploinsufficient DNA damage-specific tumor suppressor
Oncogene (2015)