Abstract
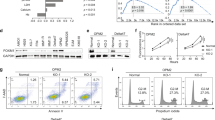
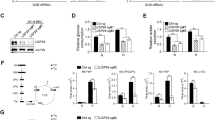
Normal cells possess adaptive mechanisms to couple energy availability with cell growth (cell size increase) and survival, and imbalances are associated with major diseases such as cancer. Inactivation of critical regulators involved in energy stress response, including adenosine monophosphate-activated protein kinase (AMPK), liver kinase B1 (LKB1), tuberous sclerosis complex 1 (TSC1) and tuberous sclerosis complex 2 (TSC2), leads to uncontrolled cell growth yet increased apoptosis under energy stress. These energy stress regulators are also important in tumor suppression and metabolism. Here, we show that forkhead box O (FoxO) transcription factor, a central regulator of tumor suppression and metabolism, plays a unique role in energy stress response. FoxOs inhibit the mammalian target of rapamycin complex 1 (mTORC1), a key regulator of cell growth, under energy stress, and inactivation of FoxOs alleviates energy stress-mediated mTORC1 repression. Surprisingly, unlike AMPK-, Lkb1- or Tsc1/2-deficient cells, FoxO-deficient cells exhibit decreased apoptosis under energy stress. FoxOs operate to inhibit mTORC1 signaling and cell survival independent of AMPK and TSC. Integrated transcriptomic and functional analyses identified BCL2/adenovirus E1B 19 kDa protein-interacting protein 3 (BNIP3)—a negative regulator of both Rheb and Bcl2 prosurvival family members—as a key downstream target of FoxOs to inhibit mTORC1 function and promote apoptosis in response to energy stress. We show that p38β, but not AMPK, is likely to function upstream of FoxO–BNIP3 to mediate energy stress response. Finally, we reveal that low expression of FoxO or BNIP3 correlates with poor clinical outcomes in renal cancer patients. Together, our study uncovers a novel signaling circuit functioning to mediate cellular energy responses to control cell growth and survival. These findings also have important implications to human cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Mihaylova MM, Shaw RJ . The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol 2011; 13: 1016–1023.
Hardie DG, Ross FA, Hawley SA . AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol 2012; 13: 251–262.
Shackelford DB, Shaw RJ . The LKB1–AMPK pathway: metabolism and growth control in tumour suppression. Nat Rev Cancer 2009; 9: 563–575.
Laplante M, Sabatini DM . mTOR signaling in growth control and disease. Cell 2012; 149: 274–293.
Wullschleger S, Loewith R, Hall MN . TOR signaling in growth and metabolism. Cell 2006; 124: 471–484.
Ma XM, Blenis J . Molecular mechanisms of mTOR-mediated translational control. Nat Rev Mol Cell Biol 2009; 10: 307–318.
Dann SG, Selvaraj A, Thomas G . mTOR complex1–S6K1 signaling: at the crossroads of obesity, diabetes and cancer. Trends Mol Med 2007; 13: 252–259.
Huang J, Manning BD . The TSC1–TSC2 complex: a molecular switchboard controlling cell growth. Biochem J 2008; 412: 179–190.
Li Y, Corradetti MN, Inoki K, Guan KL . TSC2: filling the GAP in the mTOR signaling pathway. Trends Biochem Sci 2004; 29: 32–38.
Inoki K, Zhu T, Guan KL . TSC2 mediates cellular energy response to control cell growth and survival. Cell 2003; 115: 577–590.
Gwinn DM, Shackelford DB, Egan DF, Mihaylova MM, Mery A, Vasquez DS et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell 2008; 30: 214–226.
Shaw RJ, Bardeesy N, Manning BD, Lopez L, Kosmatka M, DePinho RA et al. The LKB1 tumor suppressor negatively regulates mTOR signaling. Cancer Cell 2004; 6: 91–99.
Corradetti MN, Inoki K, Bardeesy N, DePinho RA, Guan KL . Regulation of the TSC pathway by LKB1: evidence of a molecular link between tuberous sclerosis complex and Peutz–Jeghers syndrome. Genes Dev 2004; 18: 1533–1538.
Shaw RJ, Kosmatka M, Bardeesy N, Hurley RL, Witters LA, DePinho RA et al. The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc Natl Acad Sci USA 2004; 101: 3329–3335.
Choo AY, Kim SG, Vander Heiden MG, Mahoney SJ, Vu H, Yoon SO et al. Glucose addiction of TSC null cells is caused by failed mTORC1-dependent balancing of metabolic demand with supply. Mol Cell 2010; 38: 487–499.
Zheng M, Wang YH, Wu XN, Wu SQ, Lu BJ, Dong MQ et al. Inactivation of Rheb by PRAK-mediated phosphorylation is essential for energy-depletion-induced suppression of mTORC1. Nat Cell Biol 2011; 13: 263–272.
Kim SG, Hoffman GR, Poulogiannis G, Buel GR, Jang YJ, Lee KW et al. Metabolic stress controls mTORC1 lysosomal localization and dimerization by regulating the TTT–RUVBL1/2 complex. Mol Cell 2013; 49: 172–185.
Efeyan A, Zoncu R, Chang S, Gumper I, Snitkin H, Wolfson RL et al. Regulation of mTORC1 by the Rag GTPases is necessary for neonatal autophagy and survival. Nature 2013; 493: 679–683.
Accili D, Arden KC . FoxOs at the crossroads of cellular metabolism, differentiation, and transformation. Cell 2004; 117: 421–426.
van der Horst A, Burgering BM . Stressing the role of FoxO proteins in lifespan and disease. Nat Rev Mol Cell Biol 2007; 8: 440–450.
Calnan DR, Brunet A . The FoxO code. Oncogene 2008; 27: 2276–2288.
Shaw RJ, Cantley LC . Ras, PI(3)K and mTOR signalling controls tumour cell growth. Nature 2006; 441: 424–430.
Kwiatkowski DJ, Zhang H, Bandura JL, Heiberger KM, Glogauer M, el-Hashemite N et al. A mouse model of TSC1 reveals sex-dependent lethality from liver hemangiomas, and up-regulation of p70S6 kinase activity in Tsc1 null cells. Hum Mol Genet 2002; 11: 525–534.
Kobayashi T, Minowa O, Sugitani Y, Takai S, Mitani H, Kobayashi E et al. A germ-line Tsc1 mutation causes tumor development and embryonic lethality that are similar, but not identical to, those caused by Tsc2 mutation in mice. Proc Natl Acad Sci USA 2001; 98: 8762–8767.
Gan B, Lim C, Chu G, Hua S, Ding Z, Collins M et al. FoxOs enforce a progression checkpoint to constrain mTORC1-activated renal tumorigenesis. Cancer Cell 2010; 18: 472–484.
Paik JH, Kollipara R, Chu G, Ji H, Xiao Y, Ding Z et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell 2007; 128: 309–323.
Bardeesy N, Sinha M, Hezel AF, Signoretti S, Hathaway NA, Sharpless NE et al. Loss of the Lkb1 tumour suppressor provokes intestinal polyposis but resistance to transformation. Nature 2002; 419: 162–167.
Onda H, Lueck A, Marks PW, Warren HB, Kwiatkowski DJ . Tsc2(+/−) mice develop tumors in multiple sites that express gelsolin and are influenced by genetic background. J Clin Invest 1999; 104: 687–695.
Hahn-Windgassen A, Nogueira V, Chen CC, Skeen JE, Sonenberg N, Hay N . Akt activates the mammalian target of rapamycin by regulating cellular ATP level and AMPK activity. J Biol Chem 2005; 280: 32081–32089.
Zhang J, Ney PA . Role of BNIP3 and NIX in cell death, autophagy, and mitophagy. Cell Death Differ 2009; 16: 939–946.
Chinnadurai G, Vijayalingam S, Gibson SB . BNIP3 subfamily BH3-only proteins: mitochondrial stress sensors in normal and pathological functions. Oncogene 2008; 27 (Suppl 1): S114–S127.
Burton TR, Gibson SB . The role of Bcl-2 family member BNIP3 in cell death and disease: NIPping at the heels of cell death. Cell Death Differ 2009; 16: 515–523.
Li Y, Wang Y, Kim E, Beemiller P, Wang CY, Swanson J et al. Bnip3 mediates the hypoxia-induced inhibition on mammalian target of rapamycin by interacting with Rheb. J Biol Chem 2007; 282: 35803–35813.
Mammucari C, Milan G, Romanello V, Masiero E, Rudolf R, Del Piccolo P et al. FoxO3 controls autophagy in skeletal muscle in vivo. Cell Metab 2007; 6: 458–471.
Greer EL, Oskoui PR, Banko MR, Maniar JM, Gygi MP, Gygi SP et al. The energy sensor AMP-activated protein kinase directly regulates the mammalian FOXO3 transcription factor. J Biol Chem 2007; 282: 30107–30119.
Ho KK, McGuire VA, Koo CY, Muir KW, de Olano N, Maifoshie E et al. Phosphorylation of FOXO3a on Ser-7 by p38 promotes its nuclear localization in response to doxorubicin. J Biol Chem 2012; 287: 1545–1555.
Linehan WM, Zbar B . Focus on kidney cancer. Cancer Cell 2004; 6: 223–228.
Rini BI, Campbell SC, Escudier B . Renal cell carcinoma. Lancet 2009; 373: 1119–1132.
Davila D, Connolly NMC, Bonner H, Weisova P, Dussmann H, Concannon CG et al. Two-step activation of FOXO3 by AMPK generates a coherent feed-forward loop determining excitotoxic cell fate. Cell Death Differ 2012; 19: 1677–1688.
Jones RG, Thompson CB . Tumor suppressors and cell metabolism: a recipe for cancer growth. Genes Dev 2009; 23: 537–548.
Khatri S, Yepiskoposyan H, Gallo CA, Tandon P, Plas DR . FOXO3a regulates glycolysis via transcriptional control of tumor suppressor TSC1. J Biol Chem 2010; 285: 15960–15965.
Chen CC, Jeon SM, Bhaskar PT, Nogueira V, Sundararajan D, Tonic I et al. FoxOs inhibit mTORC1 and activate Akt by inducing the expression of Sestrin3 and Rictor. Dev Cell 2010; 18: 592–604.
Gan B, Sahin E, Jiang S, Sanchez-Aguilera A, Scott KL, Chin L et al. mTORC1-dependent and -independent regulation of stem cell renewal, differentiation, and mobilization. Proc Natl Acad Sci USA 2008; 105: 19384–19389.
Gan B, Hu J, Jiang S, Liu Y, Sahin E, Zhuang L et al. Lkb1 regulates quiescence and metabolic homeostasis of haematopoietic stem cells. Nature 2010; 468: 701–704.
Laderoute KR, Amin K, Calaoagan JM, Knapp M, Le T, Orduna J et al. 5′-AMP-activated protein kinase (AMPK) is induced by low-oxygen and glucose deprivation conditions found in solid-tumor microenvironments. Mol Cell Biol 2006; 26: 5336–5347.
Gan B, Melkoumian ZK, Wu X, Guan KL, Guan JL . Identification of FIP200 interaction with the TSC1–TSC2 complex and its role in regulation of cell size control. J Cell Biol 2005; 170: 379–389.
Acknowledgements
We thank Drs Junjie Chen and Hui-Kuan Lin for critical reading of the paper. We also thank Dr Benoit Viollet for providing AMPK WT and KO MEFs and Dr Hsiao-Sheng Liu for providing BNIP3 expression plasmid. BG would also like to thank Ron DePinho for his encouragement and suggestions on this study. This study has been supported by grants from MD Anderson Cancer Center, US Department of Defense (TS093049 and PC100356), LAM Foundation (LAM092P01-12), Concern Foundation and Sidney Kimmel Foundation (to BG) and the TCGA grant U24CA143883 from NCI/NIH (to JY). BG is a Kimmel Scholar and a member of the MD Anderson Cancer Center (CA016672).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Lin, A., Yao, J., Zhuang, L. et al. The FoxO–BNIP3 axis exerts a unique regulation of mTORC1 and cell survival under energy stress. Oncogene 33, 3183–3194 (2014). https://doi.org/10.1038/onc.2013.273
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.273
Keywords
This article is cited by
-
Genome-wide expression analysis reveals different heat shock responses in indigenous (Bos indicus) and crossbred (Bos indicus X Bos taurus) cattle
Genes and Environment (2023)
-
SLC7A11 expression level dictates differential responses to oxidative stress in cancer cells
Nature Communications (2023)
-
Actin cytoskeleton vulnerability to disulfide stress mediates disulfidptosis
Nature Cell Biology (2023)
-
A targetable CoQ-FSP1 axis drives ferroptosis- and radiation-resistance in KEAP1 inactive lung cancers
Nature Communications (2022)
-
Whole-exome sequencing in eccrine porocarcinoma indicates promising therapeutic strategies
Cancer Gene Therapy (2022)