Abstract
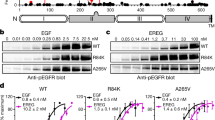
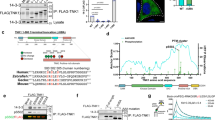
Tumor cells often subvert normal regulatory mechanisms of signal transduction. This study shows this principle by studying yet uncharacterized mutants of the epidermal growth factor receptor (EGFR) previously identified in glioblastoma multiforme, which is the most aggressive brain tumor in adults. Unlike the well-characterized EGFRvIII mutant form, which lacks a portion of the ligand-binding cleft within the extracellular domain, EGFRvIVa and EGFRvIVb lack internal segments distal to the intracellular tyrosine kinase domain. By constructing the mutants and by ectopic expression in naive cells, we show that both mutants confer an oncogenic potential in vitro, as well as tumorigenic growth in animals. The underlying mechanisms entail constitutive receptor dimerization and basal activation of the kinase domain, likely through a mechanism that relieves a restraining molecular fold, along with stabilization due to association with HSP90. Phosphoproteomic analyses delineated the signaling pathways preferentially engaged by EGFRvIVb-identified unique substrates. This information, along with remarkable sensitivities to tyrosine kinase blockers and to a chaperone inhibitor, proposes strategies for pharmacological interception in brain tumors harboring EGFRvIV mutations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
An WG, Schulte TW, Neckers LM . (2000). The heat shock protein 90 antagonist geldanamycin alters chaperone association with p210bcr-abl and v-src proteins before their degradation by the proteasome. Cell Growth Differ 11: 355–360.
Burgess AW, Cho HS, Eigenbrot C, Ferguson KM, Garrett TP, Leahy DJ et al. (2003). An open-and-shut case? Recent insights into the activation of EGF/ErbB receptors. Mol Cell 12: 541–552.
Chiara F, Bishayee S, Heldin CH, Demoulin JB . (2004). Autoinhibition of the platelet-derived growth factor beta-receptor tyrosine kinase by its C-terminal tail. J Biol Chem 279: 19732–19738.
Chiosis G, Huezo H, Rosen N, Mimnaugh E, Whitesell L, Neckers L . (2003). 17AAG: low target binding affinity and potent cell activity--finding an explanation. Mol Cancer Ther 2: 123–129.
Clarkson RW, Boland MP, Kritikou EA, Lee JM, Freeman TC, Tiffen PG et al. (2006). The genes induced by signal transducer and activators of transcription (STAT)3 and STAT5 in mammary epithelial cells define the roles of these STATs in mammary development. Mol Endocrinol 20: 675–685.
Dawson JP, Berger MB, Lin CC, Schlessinger J, Lemmon MA, Ferguson KM . (2005). Epidermal growth factor receptor dimerization and activation require ligand-induced conformational changes in the dimer interface. Mol Cell Biol 25: 7734–7742.
Ekstrand AJ, Sugawa N, James CD, Collins VP . (1992). Amplified and rearranged epidermal growth factor receptor genes in human glioblastomas reveal deletions of sequences encoding portions of the N- and/or C-terminal tails. Proc Natl Acad Sci USA 89: 4309–4313.
Frederick L, Wang XY, Eley G, James CD . (2000). Diversity and frequency of epidermal growth factor receptor mutations in human glioblastomas. Cancer Res 60: 1383–1387.
Gan HK, Kaye AH, Luwor RB . (2009). The EGFRvIII variant in glioblastoma multiforme. J Clin Neurosci 16: 748–754.
Getz G, Levine E, Domany E . (2000). Coupled two-way clustering analysis of gene microarray data. Proc Natl Acad Sci USA 97: 12079–12084.
Gotoh N, Tojo A, Hino M, Yazaki Y, Shibuya M . (1992). A highly conserved tyrosine residue at codon 845 within the kinase domain is not required for the transforming activity of human epidermal growth factor receptor. Biochem Biophys Res Commun 186: 768–774.
Hafizi S, Dahlback B . (2006). Signalling and functional diversity within the Axl subfamily of receptor tyrosine kinases. Cytokine Growth Factor Rev 17: 295–304.
Huang PH, Mukasa A, Bonavia R, Flynn RA, Brewer ZE, Cavenee WK et al. (2007). Quantitative analysis of EGFRvIII cellular signaling networks reveals a combinatorial therapeutic strategy for glioblastoma. Proc Natl Acad Sci USA 104: 12867–12872.
Huang PH, Xu AM, White FM . (2009). Oncogenic EGFR signaling networks in glioma. Sci Signal 2: re6.
Hynes NE, MacDonald G . (2009). ErbB receptors and signaling pathways in cancer. Curr Opin Cell Biol 21: 177–184.
Jeuken J, Sijben A, Alenda C, Rijntjes J, Dekkers M, Boots-Sprenger S et al. (2009). Robust detection of EGFR copy number changes and EGFR variant III: technical aspects and relevance for glioma diagnostics. Brain Pathol 19: 661–671.
Jura N, Endres NF, Engel K, Deindl S, Das R, Lamers MH et al. (2009). Mechanism for activation of the EGF receptor catalytic domain by the juxtamembrane segment. Cell 137: 1293–1307.
Katz M, Amit I, Yarden Y . (2007). Regulation of MAPKs by growth factors and receptor tyrosine kinases. Biochim Biophys Acta 1773: 1161–1176.
Kuan CT, Wikstrand CJ, Bigner DD . (2001). EGF mutant receptor vIII as a molecular target in cancer therapy. Endocr Relat Cancer 8: 83–96.
Ladanyi M, Pao W . (2008). Lung adenocarcinoma: guiding EGFR-targeted therapy and beyond. Mod Pathol 21 (Suppl 2): S16–S22.
Landau M, Fleishman SJ, Ben-Tal N . (2004). A putative mechanism for downregulation of the catalytic activity of the EGF receptor via direct contact between its kinase and C-terminal domains. Structure 12: 2265–2275.
Lavictoire SJ, Parolin DA, Klimowicz AC, Kelly JF, Lorimer IA . (2003). Interaction of Hsp90 with the nascent form of the mutant epidermal growth factor receptor EGFRvIII. J Biol Chem 278: 5292–5299.
Li JJ, Liu DP, Liu GT, Xie D . (2009). EphrinA5 acts as a tumor suppressor in glioma by negative regulation of epidermal growth factor receptor. Oncogene 28: 1759–1768.
McManus MJ, Lingle WL, Salisbury JL, Maihle NJ . (1997). A transformation-associated complex involving tyrosine kinase signal adapter proteins and caldesmon links v-erbB signaling to actin stress fiber disassembly. Proc Natl Acad Sci USA 94: 11351–11356.
Nagane M, Coufal F, Lin H, Bogler O, Cavenee WK, Huang HJ . (1996). A common mutant epidermal growth factor receptor confers enhanced tumorigenicity on human glioblastoma cells by increasing proliferation and reducing apoptosis. Cancer Res 56: 5079–5086.
Pulciani S, Santos E, Lauver AV, Long LK, Barbacid M . (1982). Transforming genes in human tumors. J Cell Biochem 20: 51–61.
Ramnarain DB, Park S, Lee DY, Hatanpaa KJ, Scoggin SO, Otu H et al. (2006). Differential gene expression analysis reveals generation of an autocrine loop by a mutant epidermal growth factor receptor in glioma cells. Cancer Res 66: 867–874.
Shewchuk LM, Hassell AM, Ellis B, Holmes WD, Davis R, Horne EL et al. (2000). Structure of the Tie2 RTK domain: self-inhibition by the nucleotide binding loop, activation loop, and C-terminal tail. Structure 8: 1105–1113.
Shimamura T, Lowell AM, Engelman JA, Shapiro GI . (2005). Epidermal growth factor receptors harboring kinase domain mutations associate with the heat shock protein 90 chaperone and are destabilized following exposure to geldanamycins. Cancer Res 65: 6401–6408.
Shtiegman K, Kochupurakkal BS, Zwang Y, Pines G, Starr A, Vexler A et al. (2007). Defective ubiquitinylation of EGFR mutants of lung cancer confers prolonged signaling. Oncogene 26: 6968–6978.
Shu HK, Pelley RJ, Kung HJ . (1991). Dissecting the activating mutations in v-erbB of avian erythroblastosis virus strain R. J Virol 65: 6173–6180.
Stamos J, Sliwkowski MX, Eigenbrot C . (2002). Structure of the epidermal growth factor receptor kinase domain alone and in complex with a 4-anilinoquinazoline inhibitor. J Biol Chem 277: 46265–46272.
Tzahar E, Waterman H, Chen X, Levkowitz G, Karunagaran D, Lavi S et al. (1996). A hierarchical network of interreceptor interactions determines signal transduction by Neu differentiation factor/neuregulin and epidermal growth factor. Mol Cell Biol 16: 5276–5287.
Wong AJ, Bigner SH, Bigner DD, Kinzler KW, Hamilton SR, Vogelstein B . (1987). Increased expression of the epidermal growth factor receptor gene in malignant gliomas is invariably associated with gene amplification. Proc Natl Acad Sci USA 84: 6899–6903.
Wood ER, Truesdale AT, McDonald OB, Yuan D, Hassell A, Dickerson SH et al. (2004). A unique structure for epidermal growth factor receptor bound to GW572016 (Lapatinib): relationships among protein conformation, inhibitor off-rate, and receptor activity in tumor cells. Cancer Res 64: 6652–6659.
Wykosky J, Gibo DM, Stanton C, Debinski W . (2005). EphA2 as a novel molecular marker and target in glioblastoma multiforme. Mol Cancer Res 3: 541–551.
Zenz R, Wagner EF . (2006). Jun signalling in the epidermis: from developmental defects to psoriasis and skin tumors. Int J Biochem Cell Biol 38: 1043–1049.
Zhang X, Gureasko J, Shen K, Cole PA, Kuriyan J . (2006). An allosteric mechanism for activation of the kinase domain of epidermal growth factor receptor. Cell 125: 1137–1149.
Zhang Y, Wolf-Yadlin A, Ross PL, Pappin DJ, Rush J, Lauffenburger DA et al. (2005). Time-resolved mass spectrometry of tyrosine phosphorylation sites in the epidermal growth factor receptor signaling network reveals dynamic modules. Mol Cell Proteomics 4: 1240–1250.
Acknowledgements
YY is the incumbent of the Harold and Zelda Goldenberg Professorial Chair. Our work is supported by research grants from the Goldhirsh Foundation, the US National Cancer Institute (NCI; CA072981 to YY, CA118705, CA141556 and U54-CA112967 to FMW), the Israel Science Foundation and Dr Miriam and Sheldon G Adelson Medical Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Pines, G., Huang, P., Zwang, Y. et al. EGFRvIV: a previously uncharacterized oncogenic mutant reveals a kinase autoinhibitory mechanism. Oncogene 29, 5850–5860 (2010). https://doi.org/10.1038/onc.2010.313
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.313
Keywords
This article is cited by
-
Targeting EGFR exon 20 insertion mutations in non-small cell lung cancer
Signal Transduction and Targeted Therapy (2019)
-
The architecture of EGFR’s basal complexes reveals autoinhibition mechanisms in dimers and oligomers
Nature Communications (2018)
-
The EGFR-HER2 module: a stem cell approach to understanding a prime target and driver of solid tumors
Oncogene (2016)
-
Asymmetric kinase dimer formation is crucial for the activation of oncogenic EGFRvIII but not for ERBB3 phosphorylation
Cell Communication and Signaling (2013)
-
A novel epidermal growth factor receptor variant lacking multiple domains directly activates transcription and is overexpressed in tumors
Oncogene (2012)