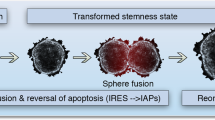
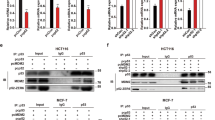
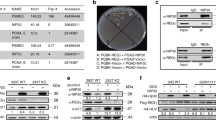
Abstract
p53 is frequently mutated by genetic alternation or suppressed by various kinds of cellular signaling pathways in human cancers. Recently, we have revealed that p53 is suppressed and eliminated from cells by direct binding with oncogenic K-Ras-induced Snail. On the basis of the fact, we generated specific inhibitors against p53-Snail binding (GN25 and GN29). These chemicals can induce p53 expression and functions in K-Ras-mutated cells. However, it does not show cytotoxic effect on normal cells or K-Ras-wild-type cells. Moreover, GN25 can selectively activate wild-type p53 in p53WT/MT cancer cells. But single allelic mt p53 containing cell line, Panc-1, does not respond to our chemical. In vivo xenograft test also supports the antitumor effect of GN25 in K-Ras-mutated cell lines. These results suggest that our compounds are strong candidate for anticancer drug against K-Ras-initiated human cancers including pancreatic and lung cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Blander G, Kipnis J, Leal JF, Yu CE, Schellenberg GD, Oren M . (1999). Physical and functional interaction between p53 and the Werner's syndrome protein. J Biol Chem 274: 29463–29469.
Brooks CL, Gu W . (2006). p53 ubiquitination: Mdm2 and beyond. Mol Cell 21: 307–315.
Butz J, Wickstrom E, Edwards J . (2003). Characterization of mutations and loss of heterozygosity of p53 and K-ras2 in pancreatic cancer cell lines by immobilized polymerase chain reaction. BMC Biotechnol 3: 11.
Cano A, Perez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG et al. (2000). The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol 2: 76–83.
Deramaudt T, Rustgi AK . (2005). Mutant KRAS in the initiation of pancreatic cancer. Biochim Biophys Acta 1756: 97–101.
Downward J . (1998). Ras signalling and apoptosis. Curr Opin Genet Dev 8: 49–54.
Ha NC, Tonozuka T, Stamos JL, Choi HJ, Weis WI . (2004). Mechanism of phosphorylation-dependent binding of APC to beta-catenin and its role in beta-catenin degradation. Mol Cell 15: 511–521.
Kajita M, McClinic KN, Wade PA . (2004). Aberrant expression of the transcription factors snail and slug alters the response to genotoxic stress. Mol Cell Biol 24: 7559–7566.
Kumaravel TS, Bristow RG . (2005). Detection of genetic instability at HER-2/neu and p53 loci in breast cancer cells using Comet-FISH. Breast Cancer Res Treat 91: 89–93.
Lee SJ, Jung YS, Lee SH, Chung HY, Park BJ . (2009a). Isolation of a chemical inhibitor against K-Ras-induced p53 suppression through natural compound screening. Int J Oncol 34: 1637–1643.
Lee SH, Lee SJ, Chung JY, Jung YS, Choi SY, Hwang SH et al. (2009b). p53, secreted by K-Ras-Snail pathway, is endocytosed by K-Ras-mutated cells; implication of target-specific drug delivery and early diagnostic marker. Oncogene 28: 2005–2014.
Lee SH, Lee SJ, Jung YS, Xu Y, Kang HS, Ha NC et al. (2009c). Blocking of p53-snail binding, promoted by oncogenic K-Ras, recovers p53 expression. Neoplasia 11: 22–31.
Leslie A, Pratt NR, Gillespie K, Sales M, Kernohan NM, Smith G et al. (2003). Mutations of APC, K-ras, and p53 are associated with specific chromosomal aberrations in colorectal adenocarcinomas. Cancer Res 63: 4656–4661.
Levine AJ . (1997). p53, the cellular gatekeeper for growth and division. Cell 88: 323–331.
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI et al. (1997). PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947.
Park BJ, Kang JW, Lee SW, Choi SJ, Shin YK, Ahn YH et al. (2005). The haploinsufficient tumor suppressor p18 upregulates p53 via interactions with ATM/ATR. Cell 120: 209–221.
Piao S, Lee SH, Kim H, Yum S, Stamos JL, Xu Y et al. (2008). Direct inhibition of GSK3beta by the phosphorylated cytoplasmic domain of LRP6 in Wnt/beta-catenin signaling. PLoS One 3: e4046.
Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW . (1997). Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88: 593–602.
Sherr CJ . (2006). Divorcing ARF and p53: an unsettled case. Nat Rev Cancer 6: 663–673.
Vassilev LT, Vu BT, Graves B, Carvajal D, Podlaski F, Filipovic Z et al. (2004). In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 303: 844–848.
Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L et al. (2007). Restoration of p53 function leads to tumour regression in vivo. Nature 445: 661–665.
Weinberg RA (ed). The Biology of Cancer, 1st edn., Ch 8, p 311. Garland Science. Whitehead Institute for Biomedical Research, MIT.
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V et al. (2007). Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445: 656–660.
Yan W, Chen X . (2009). Identification of GRO1 as a critical determinant for mutant p53 gain of function. J Biol Chem 284: 12178–12187.
Yook JI, Li XY, Ota I, Hu C, Kim NH, Cha SY et al. (2006). Wnt-Axin2-GSK3beta cascade regulates Snail1 activity in breast cancer cells. Nat Cell Biol 8: 1398–1406.
Acknowledgements
This work was supported by national cancer center of Korea (0920250; BJP), by the Bio-Scientific Research Grant funded by the Pusan National University (PNU-2008-101-20080596000; BJP) and by the Priority Research Center Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2009-0093815; GYS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Lee, SH., Shen, GN., Jung, Y. et al. Antitumor effect of novel small chemical inhibitors of Snail-p53 binding in K-Ras-mutated cancer cells. Oncogene 29, 4576–4587 (2010). https://doi.org/10.1038/onc.2010.208
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.208
Keywords
This article is cited by
-
Novel chemical inhibitor against SOD1 misfolding and aggregation protects neuron-loss and ameliorates disease symptoms in ALS mouse model
Communications Biology (2021)
-
The role of epithelial–mesenchymal transition-regulating transcription factors in anti-cancer drug resistance
Archives of Pharmacal Research (2021)
-
Pathway activity profiling of growth factor receptor network and stemness pathways differentiates metaplastic breast cancer histological subtypes
BMC Cancer (2019)
-
Hypoxic resistance of KRAS mutant tumor cells to 3-Bromopyruvate is counteracted by Prima-1 and reversed by N-acetylcysteine
BMC Cancer (2016)
-
Central role of Snail1 in the regulation of EMT and resistance in cancer: a target for therapeutic intervention
Journal of Experimental & Clinical Cancer Research (2014)