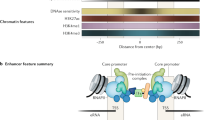
Key Points
-
This Review presents a unifying quantitative and conceptual framework for translating DNA sequences into transcriptional behaviours.
-
Each DNA-binding molecule has specific DNA sequence preferences (affinities) and, thus, every regulatory sequence defines a unique affinity landscape for each molecule.
-
At given concentrations of DNA-binding molecules, the unique affinity landscape of a regulatory sequence dictates a distinct distribution of molecule-binding configurations and, consequently, a distinct transcriptional output.
-
Accurate models of the DNA sequence preferences of nucleosomes and of many transcription factors are now available.
-
The intrinsic DNA sequence preferences of nucleosomes are major determinants of nucleosome organization in vivo, and partly account for the depletion of nucleosomes around the starts and ends of genes.
-
Nucleosome depletion around transcription factor-binding sites is partly encoded in the nucleosome affinity landscape of the genome and might assist in directing factors to their appropriate genomic sites.
-
Differences in the intrinsic nucleosome affinity landscapes in which factor-binding sites are embedded might allow the same factor to regulate its different targets with different activation dynamics.
-
Two factors that have adjacent binding sites can show indirect binding cooperativity through competition with nucleosomes, allowing some factors to function as activators on some regulatory sequences but as inhibitors on others.
-
The affinity landscape of a regulatory sequence dictates when factors must compete with nucleosomes for access to the DNA, partly explaining the differential requirement for chromatin remodellers at different loci.
-
DNA sequence changes that directly alter the nucleosome affinity landscape of a regulatory sequence might help to drive phenotypic diversity across evolution.
-
Variability in cell-to-cell expression and in DNA replication can be partly explained in terms of the affinity landscape of a DNA sequence for transcription factors and nucleosomes.
Abstract
Complex transcriptional behaviours are encoded in the DNA sequences of gene regulatory regions. Advances in our understanding of these behaviours have been recently gained through quantitative models that describe how molecules such as transcription factors and nucleosomes interact with genomic sequences. An emerging view is that every regulatory sequence is associated with a unique binding affinity landscape for each molecule and, consequently, with a unique set of molecule-binding configurations and transcriptional outputs. We present a quantitative framework based on existing methods that unifies these ideas. This framework explains many experimental observations regarding the binding patterns of factors and nucleosomes and the dynamics of transcriptional activation. It can also be used to model more complex phenomena such as transcriptional noise and the evolution of transcriptional regulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Casadaban, M. J. Transposition and fusion of the lac genes to selected promoters in Escherichia coli using bacteriophage lambda and Mu. J. Mol. Biol. 104, 541–555 (1976).
Guarente, L. & Ptashne, M. Fusion of Escherichia coli lacZ to the cytochrome c gene of Saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 78, 2199–2203 (1981).
Bellen, H. J. et al. P-element-mediated enhancer detection: a versatile method to study development in Drosophila. Genes Dev. 3, 1288–1300 (1989).
Wilson, M. D. et al. Species-specific transcription in mice carrying human chromosome 21. Science 322, 434–438 (2008).
Rajewsky, N., Vergassola, M., Gaul, U. & Siggia, E. D. Computational detection of genomic cis-regulatory modules applied to body patterning in the early Drosophila embryo. BMC Bioinformatics 3, 30 (2002).
Sinha, S., van Nimwegen, E. & Siggia, E. D. A probabilistic method to detect regulatory modules. Bioinformatics 19 (Suppl. 1), i292–i301 (2003).
Granek, J. A. & Clarke, N. D. Explicit equilibrium modeling of transcription-factor binding and gene regulation. Genome Biol. 6, R87 (2005).
Segal, E. et al. A genomic code for nucleosome positioning. Nature 442, 772–778 (2006). This paper shows that genomes encode the location of many of their nucleosomes and that this intrinsically encoded nucleosome organization might facilitate specific chromosome functions.
Ioshikhes, I. P., Albert, I., Zanton, S. J. & Pugh, B. F. Nucleosome positions predicted through comparative genomics. Nature Genet. 38, 1210–1215 (2006). This paper shows that different nucleosome architectures might be encoded at different classes of regulatory sequences.
Segal, E., Raveh-Sadka, T., Schroeder, M., Unnerstall, U. & Gaul, U. Predicting expression patterns from regulatory sequence in Drosophila segmentation. Nature 451, 535–540 (2008). This study develops an equilibrium-based thermodynamic model that relates factor binding to gene expression in Drosophila melanogaster early embryonic patterning.
Bintu, L. et al. Transcriptional regulation by the numbers: models. Curr. Opin. Genet. Dev. 15, 116–124 (2005).
Buchler, N. E., Gerland, U. & Hwa, T. On schemes of combinatorial transcription logic. Proc. Natl Acad. Sci. USA 100, 5136–5141 (2003).
Dodd, I. B., Shearwin, K. E. & Sneppen, K. Modelling transcriptional interference and DNA looping in gene regulation. J. Mol. Biol. 369, 1200–1213 (2007).
Kuhlman, T., Zhang, Z., Saier, M. H. Jr & Hwa, T. Combinatorial transcriptional control of the lactose operon of Escherichia coli. Proc. Natl Acad. Sci. USA 104, 6043–6048 (2007).
Kaplan, N. et al. The DNA-encoded nucleosome organization of a eukaryotic genome. Nature 458, 362–366 (2009). This study directly measured the intrinsically encoded nucleosome organization of the genome and showed that it is similar to the in vivo organization. A model of nucleosome occupancy trained on in vitro reconstituted yeast nucleosomes significantly predicts nucleosome organization in Caenorhabditis elegans.
Gertz, J., Siggia, E. D. & Cohen, B. A. Analysis of combinatorial cis-regulation in synthetic and genomic promoters. Nature 457, 215–218 (2009). This paper develops and experimentally verifies an equilibrium-based thermodynamic analysis that relates transcription factor binding to gene expression, and shows that these contributions explain a significant fraction of the measured variability in gene expression.
Foat, B. C., Morozov, A. V. & Bussemaker, H. J. Statistical mechanical modeling of genome-wide transcription factor occupancy data by MatrixREDUCE. Bioinformatics 22, e141–e149 (2006).
Gupta, S. et al. Predicting human nucleosome occupancy from primary sequence. PLoS Comput. Biol. 4, e1000134 (2008).
Sinha, S., Adler, A. S., Field, Y., Chang, H. Y. & Segal, E. Systematic functional characterization of cis-regulatory motifs in human core promoters. Genome Res. 18, 477–488 (2008).
Richmond, T. J. & Davey, C. A. The structure of DNA in the nucleosome core. Nature 423, 145–150 (2003).
Gencheva, M. et al. In vitro and in vivo nucleosome positioning on the ovine β-lactoglobulin gene are related. J. Mol. Biol. 361, 216–230 (2006).
Thastrom, A., Bingham, L. M. & Widom, J. Nucleosomal locations of dominant DNA sequence motifs for histone–DNA interactions and nucleosome positioning. J. Mol. Biol. 338, 695–709 (2004).
Satchwell, S. C., Drew, H. R. & Travers, A. A. Sequence periodicities in chicken nucleosome core DNA. J. Mol. Biol. 191, 659–675 (1986).
Albert, I. et al. Translational and rotational settings of H2A.Z nucleosomes across the Saccharomyces cerevisiae genome. Nature 446, 572–576 (2007).
Johnson, S. M., Tan, F. J., McCullough, H. L., Riordan, D. P. & Fire, A. Z. Flexibility and constraint in the nucleosome core landscape of Caenorhabditis elegans chromatin. Genome Res. 16, 1505–1516 (2006).
Field, Y. et al. Distinct modes of regulation by chromatin encoded through nucleosome positioning signals. PLoS Comput. Biol. 4, e1000216 (2008). This study shows that the encoded nucleosome organization is predictive of, and might be the cause of, differences in the transcriptional noise and activation dynamics of regulatory sequences and the utilization efficiency of DNA replication origins.
Yuan, G. C. & Liu, J. S. Genomic sequence is highly predictive of local nucleosome depletion. PLoS Comput. Biol. 4, e13 (2008).
Lee, W. et al. A high-resolution atlas of nucleosome occupancy in yeast. Nature Genet. 39, 1235–1244 (2007).
Peckham, H. E. et al. Nucleosome positioning signals in genomic DNA. Genome Res. 17, 1170–1177 (2007).
Korber, P., Luckenbach, T., Blaschke, D. & Horz, W. Evidence for histone eviction in trans upon induction of the yeast PHO5 promoter. Mol. Cell. Biol. 24, 10965–10974 (2004).
Vignali, M., Hassan, A. H., Neely, K. E. & Workman, J. L. ATP-dependent chromatin-remodeling complexes. Mol. Cell. Biol. 20, 1899–1910 (2000).
Pavlovic, J., Banz, E. & Parish, R. W. The effects of transcription on the nucleosome structure of four Dictyostelium genes. Nucleic Acids Res. 17, 2315–2332 (1989).
Henikoff, S. Nucleosome destabilization in the epigenetic regulation of gene expression. Nature Rev. Genet. 9, 15–26 (2008).
Gasser, R., Koller, T. & Sogo, J. M. The stability of nucleosomes at the replication fork. J. Mol. Biol. 258, 224–239 (1996).
Johnson, D. S., Mortazavi, A., Myers, R. M. & Wold, B. Genome-wide mapping of in vivo protein–DNA interactions. Science 316, 1497–1502 (2007).
Harbison, C. T. et al. Transcriptional regulatory code of a eukaryotic genome. Nature 431, 99–104 (2004).
Bulyk, M. L., Gentalen, E., Lockhart, D. J. & Church, G. M. Quantifying DNA–protein interactions by double-stranded DNA arrays. Nature Biotechnol. 17, 573–577 (1999).
Mukherjee, S. et al. Rapid analysis of the DNA-binding specificities of transcription factors with DNA microarrays. Nature Genet. 36, 1331–1339 (2004).
Maerkl, S. J. & Quake, S. R. A systems approach to measuring the binding energy landscapes of transcription factors. Science 315, 233–237 (2007).
Berger, M. F. et al. Variation in homeodomain DNA binding revealed by high-resolution analysis of sequence preferences. Cell 133, 1266–1276 (2008).
Zhu, C. et al. High-resolution DNA binding specificity analysis of yeast transcription factors. Genome Res. 19, 556–566 (2009).
Badis, G. et al. A library of yeast transcription factor motifs reveals a widespread function for Rsc3 in targeting nucleosome exclusion at promoters. Mol. Cell 32, 878–887 (2008). This study provides direct quantitative measurements of the sequence preferences of over 100 transcription factors from yeast.
Schroeder, M. D. et al. Transcriptional control in the segmentation gene network of Drosophila. PLoS Biol. 2, e271 (2004).
Tanay, A. Extensive low-affinity transcriptional interactions in the yeast genome. Genome Res. 16, 962–972 (2006).
Miller, J. A. & Widom, J. Collaborative competition mechanism for gene activation in vivo. Mol. Cell. Biol. 23, 1623–1632 (2003).
Li, X. Y. et al. Transcription factors bind thousands of active and inactive regions in the Drosophila blastoderm. PLoS Biol. 6, e27 (2008).
Shea, M. A. & Ackers, G. K. The OR control system of bacteriophage lambda. A physical–chemical model for gene regulation. J. Mol. Biol. 181, 211–230 (1985).
Hayes, J. J. & Wolffe, A. P. The interaction of transcription factors with nucleosomal DNA. Bioessays 14, 597–603 (1992).
Thomas, G. H. & Elgin, S. C. Protein/DNA architecture of the DNase I hypersensitive region of the Drosophila hsp26 promoter. EMBO J. 7, 2191–2201 (1988).
Zeitlinger, J. et al. RNA polymerase stalling at developmental control genes in the Drosophila melanogaster embryo. Nature Genet. 39, 1512–1516 (2007).
Hendrix, D. A., Hong, J. W., Zeitlinger, J., Rokhsar, D. S. & Levine, M. S. Promoter elements associated with RNA pol II stalling in the Drosophila embryo. Proc. Natl Acad. Sci. USA 105, 7762–7767 (2008).
Mavrich, T. N. et al. Nucleosome organization in the Drosophila genome. Nature 453, 358–362 (2008).
Mavrich, T. N. et al. A barrier nucleosome model for statistical positioning of nucleosomes throughout the yeast genome. Genome Res. 18, 1073–1083 (2008).
Shivaswamy, S. et al. Dynamic remodeling of individual nucleosomes across a eukaryotic genome in response to transcriptional perturbation. PLoS Biol. 6, e65 (2008).
Yuan, G. C. et al. Genome-scale identification of nucleosome positions in S. cerevisiae. Science 309, 626–630 (2005). The first high-resolution analysis of nucleosome positions in vivo at a genomic scale. The results show that there are stereotyped nucleosome organizations at promoters and add to the evidence that poly(dA:dT) tracts are unfavourable to nucleosome occupancy genome-wide.
Liu, X., Lee, C. K., Granek, J. A., Clarke, N. D. & Lieb, J. D. Whole-genome comparison of Leu3 binding in vitro and in vivo reveals the importance of nucleosome occupancy in target site selection. Genome Res. 16, 1517–1528 (2006).
Sekinger, E. A., Moqtaderi, Z. & Struhl, K. Intrinsic histone–DNA interactions and low nucleosome density are important for preferential accessibility of promoter regions in yeast. Mol. Cell 18, 735–748 (2005). An early demonstration that particular regulatory sequences encode low nucleosome occupancy.
Valouev, A. et al. A high-resolution, nucleosome position map of C. elegans reveals a lack of universal sequence-dictated positioning. Genome Res. 18, 1051–1063 (2008).
Kornberg, R. D. & Stryer, L. Statistical distributions of nucleosomes: nonrandom locations by a stochastic mechanism. Nucleic Acids Res. 16, 6677–6690 (1988). This paper develops the idea of statistical nucleosome positioning. Simple steric exclusion together with the high density of nucleosomes along the genome leads to a periodic ordering of nucleosomes away from boundary constraints.
Schones, D. E. et al. Dynamic regulation of nucleosome positioning in the human genome. Cell 132, 887–898 (2008).
Raveh-Sadka, T., Levo, M. & Segal, E. Incorporating nucleosomes into thermodynamic models of transcription regulation. Genome Res. 19 May 2009 (doi:10.1101/gr.088260.108).
Khorasanizadeh, S. The nucleosome: from genomic organization to genomic regulation. Cell 116, 259–272 (2004).
Narlikar, G. J., Fan, H. Y. & Kingston, R. E. Cooperation between complexes that regulate chromatin structure and transcription. Cell 108, 475–487 (2002).
Kornberg, R. D. & Lorch, Y. Twenty-five years of the nucleosome, fundamental particle of the eukaryote chromosome. Cell 98, 285–294 (1999).
Lam, F. H., Steger, D. J. & O'Shea, E. K. Chromatin decouples promoter threshold from dynamic range. Nature 453, 246–250 (2008). This paper shows experimentally that the relationship between nucleosome organization and the factor-binding affinity landscape determines the dynamics of activation of Pho genes in yeast.
Kim, H. D. & O'Shea, E. K. A quantitative model of transcription factor-activated gene expression. Nature Struct. Mol. Biol. 15, 1192–1198 (2008).
Polach, K. J. & Widom, J. A model for the cooperative binding of eukaryotic regulatory proteins to nucleosomal target sites. J. Mol. Biol. 258, 800–812 (1996).
Giniger, E. & Ptashne, M. Cooperative DNA binding of the yeast transcriptional activator GAL4. Proc. Natl Acad. Sci. USA 85, 382–386 (1988).
Tanaka, M. Modulation of promoter occupancy by cooperative DNA binding and activation-domain function is a major determinant of transcriptional regulation by activators in vivo. Proc. Natl Acad. Sci. USA 93, 4311–4315 (1996).
Ma, X., Yuan, D., Diepold, K., Scarborough, T. & Ma, J. The Drosophila morphogenetic protein Bicoid binds DNA cooperatively. Development 122, 1195–1206 (1996).
Rubin-Bejerano, I., Mandel, S., Robzyk, K. & Kassir, Y. Induction of meiosis in Saccharomyces cerevisiae depends on conversion of the transcriptional represssor Ume6 to a positive regulator by its regulated association with the transcriptional activator Ime1. Mol. Cell. Biol. 16, 2518–2526 (1996).
Ma, J. Crossing the line between activation and repression. Trends Genet. 21, 54–59 (2005).
Felsenfeld, G. & Groudine, M. Controlling the double helix. Nature 421, 448–453 (2003).
Robert, F. et al. Global position and recruitment of HATs and HDACs in the yeast genome. Mol. Cell 16, 199–209 (2004).
Dion, M. F. et al. Dynamics of replication-independent histone turnover in budding yeast. Science 315, 1405–1408 (2007).
Newman, J. R. et al. Single-cell proteomic analysis of S. cerevisiae reveals the architecture of biological noise. Nature 441, 840–846 (2006).
Zhang, Z., Gu, J. & Gu, X. How much expression divergence after yeast gene duplication could be explained by regulatory motif evolution? Trends Genet. 20, 403–407 (2004).
Tirosh, I., Weinberger, A., Bezalel, D., Kaganovich, M. & Barkai, N. On the relation between promoter divergence and gene expression evolution. Mol. Syst. Biol. 4, 159 (2008).
Field, Y. et al. Gene expression divergence in yeast is coupled to evolution of DNA-encoded nucleosome organization. Nature Genet. 41, 438–445 (2009). This paper shows that DNA sequence changes that directly alter the intrinsically encoded nucleosome organization of the genome are associated with, and might be important drivers of, evolutionary divergence in gene expression patterns.
Bar-Even, A. et al. Noise in protein expression scales with natural protein abundance. Nature Genet. 38, 636–643 (2006).
Raser, J. M. & O'Shea, E. K. Control of stochasticity in eukaryotic gene expression. Science 304, 1811–1814 (2004).
Landry, C. R., Lemos, B., Rifkin, S. A., Dickinson, W. J. & Hartl, D. L. Genetic properties influencing the evolvability of gene expression. Science 317, 118–121 (2007).
Tirosh, I., Weinberger, A., Carmi, M. & Barkai, N. A genetic signature of interspecies variations in gene expression. Nature Genet. 38, 830–834 (2006).
Blake, W. J., M, K. A., Cantor, C. R. & Collins, J. J. Noise in eukaryotic gene expression. Nature 422, 633–637 (2003).
Heichinger, C., Penkett, C. J., Bahler, J. & Nurse, P. Genome-wide characterization of fission yeast DNA replication origins. EMBO J. 25, 5171–5179 (2006).
Kim, S. M., Zhang, D. Y. & Huberman, J. A. Multiple redundant sequence elements within the fission yeast ura4 replication origin enhancer. BMC Mol. Biol. 2, 1 (2001).
Rippe, K., von Hippel, P. H. & Langowski, J. Action at a distance: DNA-looping and initiation of transcription. Trends Biochem. Sci. 20, 500–506 (1995).
Fyodorov, D. V. & Kadonaga, J. T. Dynamics of ATP-dependent chromatin assembly by ACF. Nature 418, 897–900 (2002).
Yang, J. G., Madrid, T. S., Sevastopoulos, E. & Narlikar, G. J. The chromatin-remodeling enzyme ACF is an ATP-dependent DNA length sensor that regulates nucleosome spacing. Nature Struct. Mol. Biol. 13, 1078–1083 (2006).
Whitehouse, I., Rando, O. J., Delrow, J. & Tsukiyama, T. Chromatin remodelling at promoters suppresses antisense transcription. Nature 450, 1031–1035 (2007).
Rippe, K. et al. DNA sequence- and conformation-directed positioning of nucleosomes by chromatin-remodeling complexes. Proc. Natl Acad. Sci. USA 104, 15635–15640 (2007).
von Hippel, P. H. & Delagoutte, E. A general model for nucleic acid helicases and their “coupling” within macromolecular machines. Cell 104, 177–190 (2001).
Dechering, K. J., Cuelenaere, K., Konings, R. N. & Leunissen, J. A. Distinct frequency-distributions of homopolymeric DNA tracts in different genomes. Nucleic Acids Res. 26, 4056–4062 (1998).
Acknowledgements
We thank the members of our laboratories for discussions and critical comments on the manuscript. E.S. acknowledges research support from the National Institutes of Health and the European Research Council and J.W. acknowledges research support from the National Institutes of Health. E.S. is the incumbent of the Soretta and Henry Shapiro career development chair.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Saccharomyces Genome Database
FURTHER INFORMATION
Glossary
- Nucleosome
-
The basic unit of chromatin, which contains 147 bp of DNA wrapped around a histone protein octamer.
- Binding configuration
-
A particular arrangement of molecules along a DNA sequence, which includes specification of the precise position and orientation (or DNA strand) at which each molecule is bound.
- Transcriptional noise
-
The variability in the transcription rate (or in steady state mRNA levels) of genes across different cells from an isogenic cell population grown in the same conditions.
- Chromatin remodeller
-
A protein or protein complex that has the capacity to alter the structure of chromatin. Some remodellers require ATP hydrolysis for their activity.
- Footprinting
-
A method for detecting protein–DNA interactions by using an enzyme to cut DNA, followed by analysis of the resulting cleavage pattern. The method is based on the fact that a protein bound to DNA protects that DNA from enzymatic cleavage.
- Gel-shift analysis
-
A technique that uses native gel electrophoresis to determine whether, and how tightly, a protein of interest can bind a given DNA sequence.
- Southwestern blotting
-
A method that involves identifying DNA-binding proteins after SDS–PAGE and transfer to a membrane using their ability to bind to specific oligonucleotide probes.
- SELEX
-
(Systematic evolution of ligands by exponential enrichment). A combinatorial technique for producing DNAs that bind specifically and with high affinity to a DNA-binding protein of interest.
- ChIP–chip
-
A technique (also known as ChIP-on-chip) that combines chromatin immunoprecipitation (ChIP) with microarray technology (chip). It is a high-throughput method for identifying, on a genome-wide scale, DNA regions that are bound in vivo by a target protein of interest.
- ChIP–seq
-
A similar technique to ChIP–chip, but the resulting interactions are read out by high-throughput parallel sequencing and not by microarrays as in ChIP–chip.
- Protein-binding microarray
-
A method that allows the high-throughput characterization of the in vitro DNA-binding site sequence specificities of transcription factors. In this approach, a DNA-binding protein of interest is expressed, purified and then bound directly to a dsDNA microarray that contains a large number of different potential DNA-binding sites.
- Microfluidic platform
-
A high-throughput platform for measuring protein–DNA affinities on the basis of mechanically induced trapping of molecular interactions.
- DNA looping
-
A conformation of a dsDNA sequence in which two regions of the DNA that are separated along the DNA in one dimension are brought close together in three-dimensional space.
- Legal configuration
-
An arrangement of molecules along a DNA sequence in which there is no steric overlap between any two molecules on the DNA.
- TATA sequence
-
A DNA sequence with a core sequence of 5′–TATA–3′ found in the promoter region of many genes. It is typically bound by a corresponding TATA-binding protein during the process of recruiting RNA polymerase to a promoter.
Rights and permissions
About this article
Cite this article
Segal, E., Widom, J. From DNA sequence to transcriptional behaviour: a quantitative approach. Nat Rev Genet 10, 443–456 (2009). https://doi.org/10.1038/nrg2591
Issue Date:
DOI: https://doi.org/10.1038/nrg2591
This article is cited by
-
Generating specificity in genome regulation through transcription factor sensitivity to chromatin
Nature Reviews Genetics (2022)
-
Differential transcriptional regulation of the NANOG gene in chicken primordial germ cells and embryonic stem cells
Journal of Animal Science and Biotechnology (2021)
-
Deciphering eukaryotic gene-regulatory logic with 100 million random promoters
Nature Biotechnology (2020)
-
Exercise adaptations: molecular mechanisms and potential targets for therapeutic benefit
Nature Reviews Endocrinology (2020)
-
Transcriptional regulatory proteins in central carbon metabolism of Pichia pastoris and Saccharomyces cerevisiae
Applied Microbiology and Biotechnology (2020)