Abstract
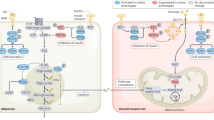
Acromegaly is a rare disease characterized by excess secretion of growth hormone (GH) and increased circulating insulin-like growth factor 1 (IGF-1) concentrations. The disease is associated with increased morbidity and premature mortality, but these effects can be reduced if GH levels are decreased to <2.5 μg/l and IGF-1 levels are normalized. Therapy for acromegaly is targeted at decreasing GH and IGF-1 levels, ameliorating patients' symptoms and decreasing any local compressive effects of the pituitary adenoma. The therapeutic options for acromegaly include surgery, radiotherapy and medical therapies, such as dopamine agonists, somatostatin receptor ligands and the GH receptor antagonist pegvisomant. Medical therapy is currently most widely used as secondary treatment for persistent or recurrent acromegaly following noncurative surgery, although it is increasingly used as primary therapy. This Review provides an overview of current and future pharmacological therapies for patients with acromegaly.
Key Points
-
Acromegaly is associated with increased morbidity and mortality
-
Targets for therapy in acromegaly include normalization of levels of growth hormone and insulin-like growth factor 1, reduction of mortality, amelioration of symptoms and reduction in tumor volume
-
Treatment options for acromegaly include surgery, radiotherapy and medical therapies such as somatostatin receptor ligands, dopamine agonists and pegvisomant
-
Medical therapy can be used as primary or secondary therapy in acromegaly
-
Treatment with somatostatin receptor ligands (SRLs) can result in biochemical control and tumor shrinkage; long-acting SRLs are more widely used due to increased compliance and patient convenience
-
Pegvisomant treatment is associated with normalization of levels of insulin-like growth factor 1 in a large proportion of patients but does not lead to tumor shrinkage
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Alexander, L., Appleton, D., Hall, R., Ross, W. M. & Wilkinson, R. Epidemiology of acromegaly in the Newcastle region. Clin. Endocrinol. (Oxf.) 12, 71–79 (1980).
Ayuk, J. et al. Growth hormone and pituitary radiotherapy, but not serum insulin-like growth factor-I concentrations, predict excess mortality in patients with acromegaly. J.Clin. Endocrinol.Metab. 89, 1613–1617 (2004).
Bates, A. S., Van't Hoff, W., Jones, J. M. & Clayton, R. N. An audit of outcome of treatment in acromegaly. Q. J. Med. 86, 293–299 (1993).
Beauregard, C., Truong, U., Hardy, J. & Serri, O. Long-term outcome and mortality after transsphenoidal adenomectomy for acromegaly. Clin. Endocrinol. (Oxf.) 58, 86–91 (2003).
Dekkers, O. M., Biermasz, N. R., Pereira, A. M., Romijn, J. A. & Vandenbroucke, J. P. Mortality in acromegaly: a metaanalysis. J. Clin. Endocrinol.Metab. 93, 61–67 (2008).
Nabarro, J. D. Acromegaly. Clin. Endocrinol. (Oxf.) 26, 481–512 (1987).
Orme, S. M., McNally, R. J., Cartwright, R. A. & Belchetz, P. E. Mortality and cancer incidence in acromegaly: a retrospective cohort study. United Kingdom Acromegaly Study Group. J. Clin. Endocrinol. Metab. 83, 2730–2734 (1998).
Rajasoorya, C., Holdaway, I. M., Wrightson, P., Scott, D. J. & Ibbertson, H. K. Determinants of clinical outcome and survival in acromegaly. Clin.Endocrinol. (Oxf.) 41, 95–102 (1994).
Swearingen, B. et al. Long-term mortality after transsphenoidal surgery and adjunctive therapy for acromegaly. J. Clin. Endocrinol. Metab. 83, 3419–3426 (1998).
Wright, A. D., Hill, D. M., Lowy, C. & Fraser, T. R. Mortality in acromegaly. Q. J. Med. 39, 1–16 (1970).
Biermasz, N. R. et al. Determinants of survival in treated acromegaly in a single center: predictive value of serial insulin-like growth factor I measurements. J. Clin. Endocrinol. Metab. 89, 2789–2796 (2004).
Melmed, S. et al. Guidelines for acromegaly management: an update. J. Clin. Endocrinol. Metab. 94, 1509–1517 (2009).
Ben-Shlomo, A. & Melmed, S. Pituitary somatostatin receptor signaling. Trends Endocrinol. Metab. 21, 123–133 (2010).
Feelders, R. A. et al. Medical therapy of acromegaly: efficacy and safety of somatostatin analogues. Drugs 69, 2207–2226 (2009).
Taboada, G. F. et al. Quantitative analysis of somatostatin receptor subtype (SSTR1–5) gene expression levels in somatotropinomas and non-functioning pituitary adenomas. Eur. J. Endocrinol. 156, 65–74 (2007).
Hofland, L. J. & Lamberts, S. W. Somatostatin receptors in pituitary function, diagnosis and therapy. Front Horm. Res. 32, 235–252 (2004).
Jaquet, P. et al. Human somatostatin receptor subtypes in acromegaly: distinct patterns of messenger ribonucleic acid expression and hormone suppression identify different tumoral phenotypes. J. Clin. Endocrinol. Metab. 85, 781–792 (2000).
Murray, R. D. & Melmed, S. A critical analysis of clinically available somatostatin analog formulations for therapy of acromegaly. J. Clin. Endocrinol. Metab. 93, 2957–2968 (2008).
Grass, P., Marbach, P., Bruns, C. & Lancranjan, I. Sandostatin LAR (microencapsulated octreotide acetate) in acromegaly: pharmacokinetic and pharmacodynamic relationships. Metabolism 45, 27–30 (1996).
Gillis, J. C., Noble, S. & Goa, K. L. Octreotide long-acting release (LAR). A review of its pharmacological properties and therapeutic use in the management of acromegaly. Drugs 53, 681–699 (1997).
McKeage, K., Cheer, S. & Wagstaff, A. J. Octreotide long-acting release (LAR): a review of its use in the management of acromegaly. Drugs 63, 2473–2499 (2003).
Vance, M. L. & Harris, A. G. Long-term treatment of 189 acromegalic patients with the somatostatin analog octreotide. Results of the International Multicenter Acromegaly Study Group. Arch. Intern. Med. 151, 1573–1578 (1991).
Ezzat, S. et al. Octreotide treatment of acromegaly. A randomized, multicenter study. Ann. Intern. Med. 117, 711–718 (1992).
Sherlock, M. et al. Medical therapy in patients with acromegaly: predictors of response and comparison of efficacy of dopamine agonists and somatostatin analogues. J. Clin. Endocrinol. Metab. 94, 1255–1263 (2009).
Morange, I. et al. Slow release lanreotide treatment in acromegalic patients previously normalized by octreotide. J. Clin. Endocrinol. Metab. 79, 145–151 (1994).
Colao, A. et al. Long-term effects of depot long-acting somatostatin analog octreotide on hormone levels and tumor mass in acromegaly. J. Clin. Endocrinol. Metab. 86, 2779–2786 (2001).
Colao, A. et al. Effectiveness and tolerability of slow release lanreotide treatment in active acromegaly. J. Endocrinol. Invest. 22, 40–47 (1999).
Freda, P. U. et al. Long-acting somatostatin analog therapy of acromegaly: a meta-analysis. J. Clin. Endocrinol. Metab. 90, 4465–4473 (2005).
Colao, A., Auriemma, R. S., Galdiero, M., Lombardi, G. & Pivonello, R. Effects of initial therapy for five years with somatostatin analogs for acromegaly on growth hormone and insulin-like growth factor-I levels, tumor shrinkage, and cardiovascular disease: a prospective study. J. Clin. Endocrinol. Metab. 94, 3746–3756 (2009).
Cozzi, R. et al. Four-year treatment with octreotide-long-acting repeatable in 110 acromegalic patients: predictive value of short-term results? J. Clin. Endocrinol. Metab. 88, 3090–3098 (2003).
Chanson, P. et al. Comparison of octreotide acetate LAR and lanreotide SR in patients with acromegaly. Clin. Endocrinol. (Oxf.) 53, 577–586 (2000).
Lancranjan, I. & Atkinson, A. B. Results of a European multicentre study with Sandostatin LAR in acromegalic patients. Sandostatin LAR Group. Pituitary 1, 105–114 (1999).
Baldelli, R. et al. Two-year follow-up of acromegalic patients treated with slow release lanreotide (30 mg). J. Clin. Endocrinol. Metab. 85, 4099–4103 (2000).
Verhelst, J. A. et al. Slow-release lanreotide in the treatment of acromegaly: a study in 66 patients. Eur. J. Endocrinol. 143, 577–584 (2000).
Bevan, J. S. Clinical review: The antitumoral effects of somatostatin analog therapy in acromegaly. J. Clin. Endocrinol. Metab. 90, 1856–1863 (2005).
Losa, M. et al. Effects of octreotide treatment on the proliferation and apoptotic index of GH-secreting pituitary adenomas. J. Clin. Endocrinol. Metab. 86, 5194–5200 (2001).
Danila, D. C. et al. Somatostatin receptor-specific analogs: effects on cell proliferation and growth hormone secretion in human somatotroph tumors. J. Clin. Endocrinol. Metab. 86, 2976–2981 (2001).
Bevan, J. S. et al. Primary medical therapy for acromegaly: an open, prospective, multicenter study of the effects of subcutaneous and intramuscular slow-release octreotide on growth hormone, insulin-like growth factor-I, and tumor size. J. Clin. Endocrinol. Metab. 87, 4554–4563 (2002).
Plockinger, U., Reichel, M., Fett, U., Saeger, W. & Quabbe, H. J. Preoperative octreotide treatment of growth hormone-secreting and clinically nonfunctioning pituitary macroadenomas: effect on tumor volume and lack of correlation with immunohistochemistry and somatostatin receptor scintigraphy. J. Clin. Endocrinol. Metab. 79, 1416–1423 (1994).
Lundin, P., Eden Engstrom, B., Karlsson, F. A. & Burman, P. Long-term octreotide therapy in growth hormone-secreting pituitary adenomas: evaluation with serial MR. Am. J. Neuroradiol. 18, 765–772 (1997).
Abe, T. & Ludecke, D. K. Effects of preoperative octreotide treatment on different subtypes of 90 GH-secreting pituitary adenomas and outcome in one surgical centre. Eur. J. Endocrinol. 145, 137–145 (2001).
Arosio, M. et al. Effects of treatment with octreotide in acromegalic patients—a multicenter Italian study. Italian Multicenter Octreotide Study Group. Eur. J. Endocrinol. 133, 430–439 (1995).
Cozzi, R. et al. Lanreotide 60 mg, a longer-acting somatostatin analog: tumor shrinkage and hormonal normalization in acromegaly. Pituitary 3, 231–238 (2000).
Livadas, S. et al. Disappearance of a growth hormone secreting macro adenoma during long-term somatostatin analogue administration and recurrence following somatostatin withdrawal. Hormones (Athens) 5, 57–63 (2006).
Ambrosio, M. R. et al. Efficacy and safety of the new 60-mg formulation of the long-acting somatostatin analog lanreotide in the treatment of acromegaly. Metabolism 51, 387–393 (2002).
Caron, P. et al. Efficacy of the new long-acting formulation of lanreotide (lanreotide Autogel) in the management of acromegaly. J. Clin. Endocrinol. Metab. 87, 99–104 (2002).
Colao, A. et al. First-line octreotide-LAR therapy induces tumour shrinkage and controls hormone excess in patients with acromegaly: results from an open, prospective, multicentre trial. Clin. Endocrinol. (Oxf.) 64, 342–351 (2006).
Melmed, S. Medical progress: Acromegaly. N. Engl. J. Med. 355, 2558–2573 (2006).
Amato, G. et al. Long-term effects of lanreotide SR and octreotide LAR on tumour shrinkage and GH hypersecretion in patients with previously untreated acromegaly. Clin. Endocrinol. (Oxf.) 56, 65–71 (2002).
Carlsen, S. M. et al. Preoperative octreotide treatment in newly diagnosed acromegalic patients with macroadenomas increases cure short-term postoperative rates: a prospective, randomized trial. J. Clin. Endocrinol. Metab. 93, 2984–2990 (2008).
Lucas, T., Astorga, R. & Catala, M. Preoperative lanreotide treatment for GH-secreting pituitary adenomas: effect on tumour volume and predictive factors of significant tumour shrinkage. Clin. Endocrinol. (Oxf.) 58, 471–481 (2003).
Baldelli, R. et al. Glucose homeostasis in acromegaly: effects of long-acting somatostatin analogues treatment. Clin. Endocrinol. (Oxf.) 59, 492–499 (2003).
Steffin, B. et al. Effects of the long-acting somatostatin analogue Lanreotide Autogel on glucose tolerance and insulin resistance in acromegaly. Eur. J. Endocrinol. 155, 73–78 (2006).
Jaffe, C. A. & Barkan, A. L. Treatment of acromegaly with dopamine agonists. Endocrinol.Metab. Clin. North Am. 21, 713–735 (1992).
Bush, Z. M. & Vance, M. L. Management of acromegaly: Is there a role for primary medical therapy? Rev. Endocr. Metab. Disord. 9, 83–94 (2008).
Abs, R. et al. Cabergoline in the treatment of acromegaly: a study in 64 patients. J. Clin.Endocrinol. Metab. 83, 374–378 (1998).
Moyes, V., Metcalfe, K. & Drake, W. Clinical use of cabergoline as primary and adjunctive treatment for acromegaly. Eur. J. Endocrinol. 159, 541–545 (2008).
Colao, A. et al. Effect of different dopaminergic agents in the treatment of acromegaly. J. Clin.Endocrinol. Metab. 82, 518–523 (1997).
Cozzi, R. et al. Cabergoline in acromegaly: a renewed role for dopamine agonist treatment? Eur. J. Endocrinol. 139, 516–521 (1998).
Ferrari, C. et al. Long-lasting lowering of serum growth hormone and prolactin levels by single and repetitive cabergoline administration in dopamine-responsive acromegalic patients. Clin. Endocrinol. (Oxf.) 29, 467–476 (1988).
Jackson, S. N., Fowler, J. & Howlett, T. A. Cabergoline treatment of acromegaly: a preliminary dose finding study. Clin. Endocrinol. (Oxf.) 46, 745–749 (1997).
Chiodini, P. G. et al. CV 205–502 in acromegaly. Acta Endocrinol. (Copenh.) 128, 389–393 (1993).
Lombardi, G. et al. CV 205–502 treatment in therapy-resistant acromegalic patients. Eur. J. Endocrinol. 132, 559–564 (1995).
Lamberts, S. W., Klijn, J. G., van Vroonhoven, C. C., Stefanko, S. Z. & Liuzzi, A. The role of prolactin in the inhibitory action of bromocriptine on growth hormone secretion in acromegaly. Acta Endocrinol. (Copenh.) 103, 446–450 (1983).
Lamberts, S. W. et al. The value of plasma prolactin levels in the prediction of the responsiveness of growth hormone secretion to bromocriptine and TRH in acromegaly. Eur. J. Clin. Invest. 12, 151–155 (1982).
Colao, A., Merola, B., Ferone, D. & Lombardi, G. Acromegaly. J. Clin. Endocrinol. Metab. 82, 2777–2781 (1997).
Cozzi, R., Attanasio, R., Lodrini, S. & Lasio, G. Cabergoline addition to depot somatostatin analogues in resistant acromegalic patients: efficacy and lack of predictive value of prolactin status. Clin. Endocrinol. (Oxf.) 61, 209–215 (2004).
Freda, P. U. et al. Cabergoline therapy of growth hormone & growth hormone/prolactin secreting pituitary tumors. Pituitary 7, 21–30 (2004).
Selvarajah, D., Webster, J., Ross, R. & Newell-Price, J. Effectiveness of adding dopamine agonist therapy to long-acting somatostatin analogues in the management of acromegaly. Eur. J. Endocrinol. 152, 569–574 (2005).
Gatta, B., Hau, D. H., Catargi, B., Roger, P. & Tabarin, A. Re-evaluation of the efficacy of the association of cabergoline to somatostatin analogues in acromegalic patients. Clin. Endocrinol. (Oxf.) 63, 477–478 (2005).
Webster, J. et al. A comparison of cabergoline and bromocriptine in the treatment of hyperprolactinemic amenorrhea. Cabergoline Comparative Study Group. N. Engl. J. Med. 331, 904–909 (1994).
Biller, B. M. et al. Treatment of prolactin-secreting macroadenomas with the once-weekly dopamine agonist cabergoline. J. Clin. Endocrinol. Metab. 81, 2338–2343 (1996).
Van Camp, G. et al. Treatment of Parkinson's disease with pergolide and relation to restrictive valvular heart disease. Lancet 363, 1179–1183 (2004).
Zanettini, R. et al. Valvular heart disease and the use of dopamine agonists for Parkinson's disease. N. Engl. J. Med. 356, 39–46 (2007).
Schade, R., Andersohn, F., Suissa, S., Haverkamp, W. & Garbe, E. Dopamine agonists and the risk of cardiac-valve regurgitation. N. Engl. J. Med. 356, 29–38 (2007).
Bogazzi, F. et al. Treatment with low doses of cabergoline is not associated with increased prevalence of cardiac valve regurgitation in patients with hyperprolactinaemia. Int. J. Clin. Pract. 62, 1864–1869 (2008).
Colao, A. et al. Increased prevalence of tricuspid regurgitation in patients with prolactinomas chronically treated with cabergoline. J. Clin. Endocrinol. Metab. 93, 3777–3784 (2008).
Kars, M., Pereira, A., Bax, J. & Romijn, J. Cabergoline and cardiac valve disease in prolactinoma patients: additional studies during long-term treatment are required. Eur. J. Endocrinol. 159, 363–367 (2008).
Lancellotti, P. et al. Cabergoline and the risk of valvular lesions in endocrine disease. Eur. J. Endocrinol. 159, 1–5 (2008).
Vallette, S. et al. Long-term cabergoline therapy is not associated with valvular heart disease in patients with prolactinomas. Pituitary 12, 153–157 (2008).
Wakil, A., Rigby, A., Clark, A. & Atkin, S. Low dose cabergoline for hyperprolactinaemia is not associated with clinically significant valvular heart disease. Eur. J. Endocrinol. 159, R11–R14 (2008).
Colao, A., Ferone, D., Marzullo, P. & Lombardi, G. Systemic complications of acromegaly: epidemiology, pathogenesis, and management. Endocr. Rev. 25, 102–152 (2004).
Higham, C. E. & Trainer, P. J. Growth hormone excess and the development of growth hormone receptor antagonists. Exp. Physiol. 93, 1157–1169 (2008).
Pokrajac, A. et al. Variation in GH and IGF-I assays limits the applicability of international consensus criteria to local practice. Clin. Endocrinol. (Oxf.) 67, 65–70 (2007).
Trainer, P. J. et al. Treatment of acromegaly with the growth hormone-receptor antagonist pegvisomant. N. Engl. J. Med. 342, 1171–1177 (2000).
van der Lely, A. J. et al. Long-term treatment of acromegaly with pegvisomant, a growth hormone receptor antagonist. Lancet 358, 1754–1759 (2001).
Mukherjee, A., Monson, J. P., Jonsson, P. J., Trainer, P. J. & Shalet, S. M. Seeking the optimal target range for insulin-like growth factor I during the treatment of adult growth hormone disorders. J. Clin. Endocrinol. Metab. 88, 5865–5870 (2003).
Herman-Bonert, V. S., Zib, K., Scarlett, J. A. & Melmed, S. Growth hormone receptor antagonist therapy in acromegalic patients resistant to somatostatin analogs. J. Clin. Endocrinol. Metab. 85, 2958–2961 (2000).
Colao, A. et al. Efficacy of 12-month treatment with the GH receptor antagonist pegvisomant in patients with acromegaly resistant to long-term, high-dose somatostatin analog treatment: effect on IGF-I levels, tumor mass, hypertension and glucose tolerance. Eur. J. Endocrinol. 154, 467–477 (2006).
Trainer, P. J. ACROSTUDY: the first 5 years. Eur. J. Endocrinol. 161 (Suppl. 1), S19–S24 (2009).
Buhk, J. H. et al. Tumor volume of growth hormone-secreting pituitary adenomas during treatment with pegvisomant: a prospective multicenter study. J. Clin. Endocrinol. Metab. 95, 552–558 (2010).
Jimenez, C. et al. Follow-up of pituitary tumor volume in patients with acromegaly treated with pegvisomant in clinical trials. Eur. J. Endocrinol. 159, 517–523 (2008).
Feenstra, J. et al. Combined therapy with somatostatin analogues and weekly pegvisomant in active acromegaly. Lancet 365, 1644–1646 (2005).
Neggers, S. J. et al. Long-term efficacy and safety of combined treatment of somatostatin analogs and pegvisomant in acromegaly. J. Clin. Endocrinol. Metab. 92, 4598–4601 (2007).
Jehle, S., Reyes, C. M., Sundeen, R. E. & Freda, P. U. Alternate-day administration of pegvisomant maintains normal serum insulin-like growth factor-I levels in patients with acromegaly. J. Clin. Endocrinol. Metab. 90, 1588–1593 (2005).
Higham, C. E., Thomas, J. D., Bidlingmaier, M., Drake, W. M. & Trainer, P. J. Successful use of weekly pegvisomant administration in patients with acromegaly. Eur. J. Endocrinol. 161, 21–25 (2009).
Feenstra, J., van Aken, M. O., de Herder, W. W., Feelders, R. A. & van der Lely, A. J. Drug-induced hepatitis in an acromegalic patient during combined treatment with pegvisomant and octreotide long-acting repeatable attributed to the use of pegvisomant. Eur. J. Endocrinol. 154, 805–806 (2006).
Biering, H. et al. Elevated transaminases during medical treatment of acromegaly: a review of the German pegvisomant surveillance experience and a report of a patient with histologically proven chronic mild active hepatitis. Eur. J. Endocrinol. 154, 213–220 (2006).
van der Hoek, J. et al. A single-dose comparison of the acute effects between the new somatostatin analog SOM230 and octreotide in acromegalic patients. J. Clin. Endocrinol. Metab. 89, 638–645 (2004).
Petersenn, S. et al. Pasireotide (SOM230) demonstrates efficacy and safety in patients with acromegaly: a randomized, multicenter, phase II trial. J. Clin. Endocrinol. Metab. 95, 2781–2789 (2010).
Jaquet, P. et al. BIM-23A760, a chimeric molecule directed towards somatostatin and dopamine receptors, vs universal somatostatin receptors ligands in GH-secreting pituitary adenomas partial responders to octreotide. J. Endocrinol. Invest. 28 (Suppl.), 21–27 (2005).
Jaquet, P. et al. Efficacy of chimeric molecules directed towards multiple somatostatin and dopamine receptors on inhibition of GH and prolactin secretion from GH-secreting pituitary adenomas classified as partially responsive to somatostatin analog therapy. Eur. J. Endocrinol 153, 135–141 (2005).
Author information
Authors and Affiliations
Contributions
M. Sherlock, C. Woods and M. C. Sheppard wrote the article, researched data for the article, provided a substantial contribution to discussion of the content and reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sherlock, M., Woods, C. & Sheppard, M. Medical therapy in acromegaly. Nat Rev Endocrinol 7, 291–300 (2011). https://doi.org/10.1038/nrendo.2011.42
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.42
This article is cited by
-
Effectiveness of lanreotide autogel 120 mg at extended dosing intervals for acromegaly
Endocrine (2020)
-
Targeting growth hormone function: strategies and therapeutic applications
Signal Transduction and Targeted Therapy (2019)
-
Cabergoline in acromegaly
Pituitary (2017)
-
New therapeutic agents for acromegaly
Nature Reviews Endocrinology (2016)
-
Molecular Characterization of Growth Hormone-producing Tumors in the GC Rat Model of Acromegaly
Scientific Reports (2015)