Abstract
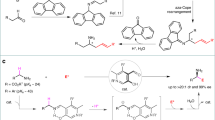
This protocol describes the catalytic asymmetric formation of all-carbon quaternary centers—a distinctive feature of many natural products and pharmaceuticals—via conjugate addition of alkylzirconium reagents to a tertiary enone. This methodology uses alkenes as starting materials and enables the incorporation of functional groups. The alkylzirconium reagent is generated in situ by mixing the alkene with the Schwartz reagent. The alkylzirconium is added to a solution containing a copper-ligand complex, and then the enone is added to the mixture. The addition of pent-4-en-1-ylbenzene to 3-methyl-2-cyclohexenone is detailed herein as a generic example. This procedure works at room temperature (∼25 °C), and it is scalable to at least 1.5 g. The setup of the reaction takes 3–5 h and the reaction goes to completion within 4–20 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Paterson, I. & Anderson, E.A. The renaissance of natural products as drug candidates. Science 310, 451–453 (2005).
Butler, M.S. Natural products to drugs: natural product-derived compounds in clinical trials. Nat. Prod. Rep. 25, 475–516 (2008).
Ishibashi, M. et al. Metachromins A and B, novel antineoplastic sesquiterpenoids from the Okinawan sponge Hippospongia cf. metachromia. J. Org. Chem. 53, 2855–2858 (1988).
Majetich, G. & Shimkus, J.M. The taiwaniaquinoids: a review. J. Nat. Prod. 73, 284–298 (2010).
Suffness, M. (Ed) Taxol: Science and Applications (CRC Press, 1995).
Greig, N.H., Pei, X.-F., Soncrant, T.T., Ingram, D.K. & Brossi, A. Phenserine and ring C hetero-analogues: drug candidates for the treatment of Alzheimer's disease. Med. Res. Rev. 15, 3–31 (1995).
Kavanagh, F., Hervey, A. & Robbins, W.J. Antibiotic substances from basidiomycetes: VIII. Pleurotus Multilus (Fr.) Sacc. and Pleurotus Passeckerianus Pilat. Proc. Natl. Acad. Sci. USA 37, 570–574 (1951).
Brady, S.F., Singh, M.P., Janso, J.E. & Clardy, J. Guanacastepene, a fungal-derived diterpene antibiotic with a new carbon skeleton. J. Am. Chem. Soc. 122, 2116–2117 (2000).
Wellington, K.D., Cambie, R.C., Rutledge, P.S. & Bergquist, P.R. Chemistry of sponges. 19. novel bioactive metabolites from Hamigera tarangaensis. J. Nat. Prod. 63, 79–85 (1999).
Corey, E.J. & Guzman-Perez, A. The catalytic enantioselective construction of molecules with quaternary carbon stereocenters. Angew. Chem. Int. Ed. 37, 388–401 (1998).
Christoffers, J. & Baro, A. Construction of quaternary stereocenters: new perspectives through enantioselective Michael reactions. Angew. Chem. Int. Ed. 42, 1688–1690 (2003).
Douglas, C.J. & Overman, L.E. Catalytic asymmetric synthesis of all-carbon quaternary stereocenters. Proc. Natl. Acad. Sci. USA 101, 5363–5367 (2004).
Trost, B.M. & Jiang, C.H. Catalytic enantioselective construction of all-carbon quaternary stereocenters. Synthesis 2006, 369–396 (2006).
Hawner, C. & Alexakis, A. Metal-catalyzed asymmetric conjugate addition reaction: formation of quaternary stereocenters. Chem. Commun. 46, 7295–7306 (2010).
Das, J.P. & Marek, I. Enantioselective synthesis of all-carbon quaternary stereogenic centers in acyclic systems. Chem. Commun. 47, 4593–4623 (2011).
Jerphagnon, T., Pizzuti, M.G., Minnaard, A.J. & Feringa, B.L. Recent advances in enantioselective copper-catalyzed 1,4-addition. Chem. Soc. Rev. 38, 1039–1075 (2009).
Alexakis, A., Backvall, J.E., Krause, N., Pamies, O. & Dieguez, M. Enantioselective copper-catalyzed conjugate addition and allylic substitution reactions. Chem. Rev. 108, 2796–2823 (2008).
Thaler, T. & Knochel, P. Copper-catalyzed asymmetric Michael addition of magnesium, zinc, and aluminum organometallic reagents: Efficient synthesis of chiral molecules. Angew. Chem. Int. Ed. 48, 645–648 (2009).
Lee, K.S., Brown, M.K., Hird, A.W. & Hoveyda, A.H. A practical method for enantioselective synthesis of all-carbon quaternary stereogenic centers through NHC-Cu-catalyzed conjugate additions of alkyl- and arylzinc reagents to β-substituted cyclic enones. J. Am. Chem. Soc. 128, 7182–7184 (2006).
Brown, M.K., May, T.L., Baxter, C.A. & Hoveyda, A.H. All-carbon quaternary stereogenic centers by enantioselective Cu-catalyzed conjugate additions promoted by a chiral N-heterocyclic carbene. Angew. Chem. Int. Ed. 46, 1097–1100 (2007).
Hawner, C., Li, K.Y., Cirriez, V. & Alexakis, A. Copper-catalyzed asymmetric conjugate addition of Aryl aluminum reagents to trisubstituted enones: construction of aryl-substituted quaternary centers. Angew. Chem. Int. Ed. 47, 8211–8214 (2008).
May, T.L., Brown, M.K. & Hoveyda, A.H. Enantioselective synthesis of all-carbon quaternary stereogenic centers by catalytic asymmetric conjugate additions of alkyl and aryl aluminum reagents to five-, six-, and seven-membered-ring β-substituted cyclic enones. Angew. Chem. Int. Ed. 47, 7358–7362 (2008).
Martin, D., Kehrli, S., d'Augustin, M., Clavier, H., Mauduit, M. & Alexakis, A. Copper-catalyzed asymmetric conjugate addition of Grignard reagents to trisubstituted enones. Construction of all-carbon quaternary chiral centers. J. Am. Chem. Soc. 128, 8416–8417 (2006).
Kehrli, S., Martin, D., Rix, D., Mauduit, M. & Alexakis, A. Formation of quaternary chiral centers by N-heterocyclic carbene-Cu-catalyzed asymmetric conjugate addition reactions with Grignard reagents on trisubstituted cyclic enones. Chemistry 16, 9890–9904 (2010).
Shintani, R., Duan, W.L. & Hayashi, T. Rhodium-catalyzed asymmetric construction of quaternary carbon stereocenters: Ligand-dependent regiocontrol in the 1,4-addition to substituted maleimides. J. Am. Chem. Soc. 128, 5628–5629 (2006).
Shintani, R., Tsutsumi, Y., Nagaosa, M., Nishimura, T. & Hayashi, T. Sodium tetraarylborates as effective nucleophiles in rhodium/diene-catalyzed 1,4-addition to β,β-disubstituted α,β-unsaturated ketones: catalytic asymmetric construction of quaternary carbon stereocenters. J. Am. Chem. Soc. 131, 13588–13589 (2009).
Shintani, R., Takeda, M., Nishimura, T. & Hayashi, T. Chiral tetrafluorobenzobarrelenes as effective ligands for rhodium-catalyzed asymmetric 1,4-addition of arylboroxines to β,β-disubstituted α,β-unsaturated ketones. Angew. Chem. Int. Ed. 49, 3969–3971 (2010).
Hahn, B.T., Tewes, F., Frohlich, R. & Glorius, F. Olefin-oxazolines (OlefOx): highly modular, easily tunable ligands for asymmetric catalysis. Angew. Chem. Int. Ed. 49, 1143–1146 (2010).
Kikushima, K., Holder, J.C., Gatti, M. & Stoltz, B.M. Palladium-catalyzed asymmetric conjugate addition of arylboronic acids to five-, six-, and seven-membered β-substituted cyclic enones: enantioselective construction of all-carbon quaternary stereocenters. J. Am. Chem. Soc. 133, 6902–6905 (2011).
Gottumukkala, A.L., Matcha, K., Lutz, M., de Vries, J.G. & Minnaard, A.J. Palladium-catalyzed asymmetric quaternary stereocenter formation. Chemistry 18, 6907–6914 (2012).
Muller, D. & Alexakis, A. Rhodium and copper-catalyzed asymmetric conjugate addition of alkenyl nucleophiles. Chem. Commun. 48, 12037–12049 (2012).
Wu, J., Mampreian, D.M. & Hoveyda, A.H. Enantioselective synthesis of nitroalkanes bearing all-carbon quaternary stereogenic centers through Cu-catalyzed asymmetric conjugate additions. J. Am. Chem. Soc. 127, 4584–4585 (2005).
Hird, A.W. & Hoveyda, A.H. Catalytic enantioselective alkylations of tetrasubstituted olefins. Synthesis of all-carbon quaternary stereogenic centers through Cu-catalyzed asymmetric conjugate additions of alkylzinc reagents to enones. J. Am. Chem. Soc. 127, 14988–14989 (2005).
Fillion, E. & Wilsily, A. Asymmetric synthesis of all-carbon benzylic quaternary stereocenters via Cu-catalyzed conjugate addition of dialkylzinc reagents to 5-(1-arylalkylidene) Meldrum's acids. J. Am. Chem. Soc. 128, 2774–2775 (2006).
Dumas, A.M. & Fillion, E. Meldrum's acids and 5-alkylidene Meldrum's acids in catalytic carbon-carbon bond-forming processes. Acc. Chem. Res. 43, 440–454 (2010).
d'Augustin, M., Palais, L. & Alexakis, A. Enantioselective copper-catalyzed conjugate addition to trisubstituted cyclohexenones: construction of stereogenic quaternary centers. Angew. Chem. Int. Ed. 44, 1376–1378 (2005).
Palais, L. et al. SimplePhos monodentate ligands: synthesis and application in copper-catalyzed reactions. Angew. Chem. Int. Ed. 46, 7462–7465 (2007).
Vuagnoux-d'Augustin, M. & Alexakis, A. Copper-catalyzed asymmetric conjugate addition of trialkylaluminium reagents to trisubstituted enones: construction of chiral quaternary centers. Chemistry 13, 9647–9662 (2007).
Palais, L. & Alexakis, A. Copper-catalyzed asymmetric conjugate addition with chiral SimplePhos ligands. Chemistry 15, 10473–10485 (2009).
Mendoza, A., Ishihara, Y. & Baran, P.S. Scalable enantioselective total synthesis of taxanes. Nat. Chem. 4, 21–25 (2012).
Henon, H., Mauduit, M. & Alexakis, A. Regiodivergent 1,4 versus 1,6 asymmetric copper-catalyzed conjugate addition. Angew. Chem. Int. Ed. 47, 9122–9124 (2008).
Matsumoto, Y., Yamada, K.I. & Tomioka, K. C(2) symmetric chiral NHC ligand for asymmetric quaternary carbon constructing copper-catalyzed conjugate addition of Grignard reagents to 3-substituted cyclohexenones. J. Org. Chem. 73, 4578–4581 (2008).
Germain, N., Magrez, M., Kehrli, S., Mauduit, M. & Alexakis, A. Chiral NHC ligands for the copper-catalyzed asymmetric conjugate addition of Grignard reagents. Eur. J. Org. Chem. 5301–5306 (2012).
Tissot, M. et al. Formation of quaternary stereogenic centers by NHC-Cu-catalyzed asymmetric conjugate addition reactions with Grignard reagents on polyconjugated cyclic enones. Chemistry 18, 8731–8747 (2012).
Mak, J.Y.W. & Williams, C.M. Enantioselective total synthesis of (−)-neovibsanin G and (−)-14-epi-neovibsanin G. Chem. Commun. 48, 287–289 (2012).
Brown, M.K. & Hoveyda, A.H. Enantioselective total synthesis of clavirolide C. applications of Cu-catalyzed asymmetric conjugate additions and Ru-catalyzed ring-closing metathesis. J. Am. Chem. Soc. 130, 12904–12906 (2008).
Vuagnoux-d'Augustin, M., Kehrli, S. & Alexakis, A. Enantioselective copper-catalyzed conjugate addition to 2- or 3-substituted cyclopent-2-en-1-ones: construction of stereogenic quaternary carbon centers. Synlett 2057–2060 (2007).
Howell, G.P. Asymmetric and diastereoselective conjugate addition reactions: C-C bond formation at large scale. Org. Proc. Res. Develop. 16, 1258–1272 (2012).
Young, I.S. & Baran, P.S. Protecting-group-free synthesis as an opportunity for invention. Nat. Chem. 1, 193–205 (2009).
Maksymowicz, R.M., Roth, P.M.C. & Fletcher, S.P. Catalytic asymmetric carbon-carbon bond formation using alkenes as alkylmetal equivalents. Nat. Chem. 4, 649–654 (2012).
Maksymowicz, R.M., Roth, P.M.C., Thompson, A.L. & Fletcher, S.P. Hydrometallation-asymmetric conjugate addition: application to complex molecule synthesis. Chem. Commun. 49, 4211–4213 (2013).
Sidera, M., Roth, P.M.C., Maksymowicz, R.M. & Fletcher, S.P. Formation of quaternary centers by copper-catalyzed asymmetric conjugate addition of alkylzirconium reagents. Angew. Chem. Int. Ed. 52, 7995–7999 (2013).
Maksymowicz, R.M., Sidera, M., Roth, P.M.C. & Fletcher, S.P. A convenient catalytic asymmetric conjugate addition reaction to enones using alkylzirconium reagents. Synthesis 45, 2662–2668 (2013).
Sebesta, R. Asymmetric copper-catalyzed 1,4-additions and allylic substitutions with nucleophiles formed by the hydrometalation of alkenes. ChemCatChem 5, 1069–1071 (2013).
Schwartz, J. & Labinger, J.A. Hydrozirconation: new transition-metal reagent for organic-synthesis. Angew. Chem. Int. Ed. 15, 333–340 (1976).
Buchwald, S.L., LaMaire, S.J. & Nielsen, R.B. Schwartz reagent. Org. Synth. 71, 77–82 (1993).
Yoshida, M., Ohmiya, H. & Sawamura, M. Enantioselective conjugate addition of alkylboranes catalyzed by a copper-N-heterocyclic carbene complex. J. Am. Chem. Soc. 134, 11896–11899 (2012).
Teichert, J.F. & Feringa, B.L. Phosphoramidites: privileged ligands in asymmetric catalysis. Angew. Chem. Int. Ed. 49, 2486–2528 (2010).
Vij, A., Zheng, Y.Y., Kirchmeier, R.I. & Shreeve, J.M. Electrophilic addition and substitution reactions of bis(trifluoromethyl)sulfonyl)amide and its N-chloro derivative. Inorg. Chem. 33, 3281–3288 (1994).
Wipf, P. & Smitrovich, J.H. Transmetalation reactions of alkylzirconocenes—copper-catalyzed conjugate addition to enones. J. Org. Chem. 56, 6494–6496 (1991).
Acknowledgements
We thank the UK Engineering and Physical Sciences Research Council for generous support of this research in the form of a Career Acceleration Fellowship to S.P.F. (EP/H003711/1).
Author information
Authors and Affiliations
Contributions
P.M.C.R., M.S. and R.M.M. conducted the experiments. All authors designed the experiments and analyzed the data. S.P.F. guided the research. P.M.C.R., M.S. and S.P.F. wrote the manuscript. All authors contributed to discussions.
Corresponding author
Ethics declarations
Competing interests
The authors are all named as inventors on a UK patent application filed by Isis Innovation, which is the technology transfer arm of the University of Oxford.
Supplementary information
Supplementary Methods
Synthesis of diphenyl isopropyl binol phosphoramidite ligand A (PDF 106 kb)
Rights and permissions
About this article
Cite this article
Roth, P., Sidera, M., Maksymowicz, R. et al. Copper-catalyzed asymmetric conjugate addition of alkylzirconium reagents to cyclic enones to form quaternary centers. Nat Protoc 9, 104–111 (2014). https://doi.org/10.1038/nprot.2013.169
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.169
This article is cited by
-
Desymmetrization of meso-bisphosphates using copper catalysis and alkylzirconocene nucleophiles
Nature Communications (2019)
-
Computational ligand design in enantio- and diastereoselective ynamide [5+2] cycloisomerization
Nature Communications (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.