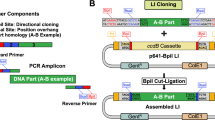
Abstract
Engineered zinc finger nucleases can stimulate gene targeting at specific genomic loci in insect, plant and human cells. Although several platforms for constructing artificial zinc finger arrays using “modular assembly” have been described, standardized reagents and protocols that permit rapid, cross-platform “mixing-and-matching” of the various zinc finger modules are not available. Here we describe a comprehensive, publicly available archive of plasmids encoding more than 140 well-characterized zinc finger modules together with complementary web-based software (termed ZiFiT) for identifying potential zinc finger target sites in a gene of interest. Our reagents have been standardized on a single platform, enabling facile mixing-and-matching of modules and transfer of assembled arrays to expression vectors without the need for specialized knowledge of zinc finger sequences or complicated oligonucleotide design. We also describe a bacterial cell-based reporter assay for rapidly screening the DNA-binding activities of assembled multi-finger arrays. This protocol can be completed in approximately 24–26 d.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Beumer, K., Bhattacharyya, G., Bibikova, M., Trautman, J.K. & Carroll, D. Efficient gene targeting in Drosophila with zinc-finger nucleases. Genetics 172, 2391–2403 (2006).
Bibikova, M., Beumer, K., Trautman, J.K. & Carroll, D. Enhancing gene targeting with designed zinc finger nucleases. Science 300, 764 (2003).
Bibikova, M., Golic, M., Golic, K.G. & Carroll, D. Targeted chromosomal cleavage and mutagenesis in Drosophila using zinc-finger nucleases. Genetics 161, 1169–1175 (2002).
Wright, D.A. et al. High-frequency homologous recombination in plants mediated by zinc-finger nucleases. Plant J. 44, 693–705 (2005).
Porteus, M.H. & Baltimore, D. Chimeric nucleases stimulate gene targeting in human cells. Science 300, 763 (2003).
Urnov, F.D. et al. Highly efficient endogenous human gene correction using designed zinc-finger nucleases. Nature 435, 646–651 (2005).
Alwin, S. et al. Custom zinc-finger nucleases for use in human cells. Mol. Ther. 12, 610–617 (2005).
Kim, Y.G., Cha, J. & Chandrasegaran, S. Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain. Proc. Natl. Acad. Sci. USA 93, 1156–1160 (1996).
Smith, J., Berg, J.M. & Chandrasegaran, S. A detailed study of the substrate specificity of a chimeric restriction enzyme. Nucleic Acids Res. 27, 674–681 (1999).
Smith, J. et al. Requirements for double-strand cleavage by chimeric restriction enzymes with zinc finger DNA-recognition domains. Nucleic Acids Res. 28, 3361–3369 (2000).
Mani, M., Smith, J., Kandavelou, K., Berg, J.M. & Chandrasegaran, S. Binding of two zinc finger nuclease monomers to two specific sites is required for effective double-strand DNA cleavage. Biochem. Biophys. Res. Commun. 334, 1191–1197 (2005).
Porteus, M.H. Mammalian gene targeting with designed zinc finger nucleases. Mol. Ther. 13, 438–446 (2006).
Dreier, B., Beerli, R.R., Segal, D.J., Flippin, J.D. & Barbas, C.F. 3rd Development of zinc finger domains for recognition of the 5′-ANN-3′ family of DNA sequences and their use in the construction of artificial transcription factors. J. Biol. Chem. 276, 29466–29478 (2001).
Dreier, B. et al. Development of zinc finger domains for recognition of the 5′-CNN-3′ family DNA sequences and their use in the construction of artificial transcription factors. J. Biol. Chem. 280, 35588–35597 (2005).
Dreier, B., Segal, D.J. & Barbas, C.F., 3rd Insights into the molecular recognition of the 5′-GNN-3′ family of DNA sequences by zinc finger domains. J. Mol. Biol. 303, 489–502 (2000).
Segal, D.J., Dreier, B., Beerli, R.R. & Barbas, C.F. 3rd Toward controlling gene expression at will: selection and design of zinc finger domains recognizing each of the 5′-GNN-3′ DNA target sequences. Proc. Natl. Acad. Sci. USA 96, 2758–2763 (1999).
Segal, D.J. The use of zinc finger peptides to study the role of specific factor binding sites in the chromatin environment. Methods 26, 76–83 (2002).
Desjarlais, J.R. & Berg, J.M. Use of a zinc-finger consensus sequence framework and specificity rules to design specific DNA binding proteins. Proc. Natl. Acad. Sci. USA 90, 2256–2260 (1993).
Mandell, J.G. & Barbas, C.F. 3rd Zinc Finger Tools: custom DNA-binding domains for transcription factors and nucleases. Nucleic Acids Res. 34, W516–523 (2006).
Liu, P.Q. et al. Regulation of an endogenous locus using a panel of designed zinc finger proteins targeted to accessible chromatin regions. Activation of vascular endothelial growth factor A. J. Biol. Chem. 276, 11323–11334 (2001).
Zhang, L. et al. Synthetic zinc finger transcription factor action at an endogenous chromosomal site. Activation of the human erythropoietin gene. J. Biol. Chem. 275, 33850–33860 (2000).
Liu, Q., Xia, Z., Zhong, X. & Case, C.C. Validated zinc finger protein designs for all 16 GNN DNA triplet targets. J. Biol. Chem. 277, 3850–3856 (2002).
Bae, K.H. et al. Human zinc fingers as building blocks in the construction of artificial transcription factors. Nat. Biotechnol. 21, 275–280 (2003).
Park, K.S. et al. Phenotypic alteration of eukaryotic cells using randomized libraries of artificial transcription factors. Nat. Biotechnol. 21, 1208–1214 (2003).
Kwon, H.S., Shin, H.C. & Kim, J.S. Suppression of vascular endothelial growth factor expression at the transcriptional and post-transcriptional levels. Nucleic. Acids Res. 33, e74 (2005).
Kwon, R.J. et al. Artificial transcription factors increase production of recombinant antibodies in Chinese hamster ovary cells. Biotechnol. Lett. 28, 9–15 (2006).
Lee, D.K. et al. Toward a functional annotation of the human genome using artificial transcription factors. Genome Res. 13, 2708–2716 (2003).
Park, K.S., Jang, Y.S., Lee, H. & Kim, J.S. Phenotypic alteration and target gene identification using combinatorial libraries of zinc finger proteins in prokaryotic cells. J. Bacteriol. 187, 5496–5499 (2005).
Park, K.S. et al. Identification and use of zinc finger transcription factors that increase production of recombinant proteins in yeast and mammalian cells. Biotechnol. Prog. 21, 664–670 (2005).
Segal, D.J. et al. Evaluation of a modular strategy for the construction of novel polydactyl zinc finger DNA-binding proteins. Biochemistry 42, 2137–2148 (2003).
Hurt, J.A., Thibodeau, S.A., Hirsh, A.S., Pabo, C.O. & Joung, J.K. Highly specific zinc finger proteins obtained by directed domain shuffling and cell-based selection. Proc. Natl. Acad. Sci. USA 100, 12271–12276 (2003).
Pabo, C.O., Peisach, E. & Grant, R.A. Design and selection of novel Cys2His2 zinc finger proteins. Annu. Rev. Biochem. 70, 313–340 (2001).
Wolfe, S.A., Nekludova, L. & Pabo, C.O. DNA recognition by Cys2His2 zinc finger proteins. Annu. Rev. Biophys. Biomol. Struct. 29, 183–212 (2000).
Isalan, M., Choo, Y. & Klug, A. Synergy between adjacent zinc fingers in sequence-specific DNA recognition. Proc. Natl. Acad. Sci. USA 94, 5617–5621 (1997).
Isalan, M., Klug, A. & Choo, Y. Comprehensive DNA recognition through concerted interactions from adjacent zinc fingers. Biochemistry 37, 12026–12033 (1998).
Mani, M., Kandavelou, K., Dy, F.J., Durai, S. & Chandrasegaran, S. Design, engineering, and characterization of zinc finger nucleases. Biochem. Biophys. Res. Commun. 335, 447–457 (2005).
Dove, S.L., Joung, J.K. & Hochschild, A. Activation of prokaryotic transcription through arbitrary protein-protein contacts. Nature 386, 627–630 (1997).
Joung, J.K. Identifying and modifying protein-DNA and protein-protein interactions using a bacterial two-hybrid selection system. J. Cell. Biochem. Suppl 37: 53–57 (2001).
Joung, J.K., Ramm, E.I. & Pabo, C.O. A bacterial two-hybrid selection system for studying protein-DNA and protein-protein interactions. Proc. Natl. Acad. Sci. USA 97, 7382–7387 (2000).
Beerli, R.R., Segal, D.J., Dreier, B. & Barbas, C.F. 3rd Toward controlling gene expression at will: specific regulation of the erbB-2/HER-2 promoter by using polydactyl zinc finger proteins constructed from modular building blocks. Proc. Natl. Acad. Sci. USA 95, 14628–14633 (1998).
Beerli, R.R. & Barbas, C.F., 3rd Engineering polydactyl zinc-finger transcription factors. Nat. Biotechnol. 20, 135–141 (2002).
Blancafort, P. et al. Genetic reprogramming of tumor cells by zinc finger transcription factors. Proc. Natl. Acad. Sci. USA 102, 11716–11721 (2005).
Jamieson, A.C., Miller, J.C. & Pabo, C.O. Drug discovery with engineered zinc-finger proteins. Nat. Rev. Drug Discov. 2, 361–368 (2003).
Klug, A. Towards therapeutic applications of engineered zinc finger proteins. FEBS Lett 579, 892–894 (2005).
Lee, D.K., Seol, W. & Kim, J.S. Custom DNA-binding proteins and artificial transcription factors. Curr. Topics Med. Chem. 3, 645–657 (2003).
Falke, D. & Juliano, R.L. Selective gene regulation with designed transcription factors: implications for therapy. Curr. Opin. Mol. Ther. 5, 161–166 (2003).
Beerli, R.R., Dreier, B. & Barbas, C.F., 3rd Positive and negative regulation of endogenous genes by designed transcription factors. Proc. Natl. Acad. Sci. USA 97, 1495–1500 (2000).
Ren, D., Collingwood, T.N., Rebar, E.J., Wolffe, A.P. & Camp, H.S. PPARgamma knockdown by engineered transcription factors: exogenous PPARgamma2 but not PPARgamma1 reactivates adipogenesis. Genes Dev. 16, 27–32 (2002).
Liang, Y. et al. Activation of vascular endothelial growth factor A transcription in tumorigenic glioblastoma cell lines by an enhancer with cell type-specific DNase I accessibility. J. Biol. Chem. 277, 20087–20094 (2002).
Falke, D., Fisher, M., Ye, D. & Juliano, R.L. Design of artificial transcription factors to selectively regulate the pro-apoptotic bax gene. Nucleic Acids Res. 31, e10 (2003).
Blancafort, P., Magnenat, L. & Barbas, C.F. Scanning the human genome with combinatorial transcription factor libraries. Nat. Biotechnol. 21, 269–274 (2003).
Blancafort, P., Segal, D.J. & Barbas, C.F. 3rd Designing transcription factor architectures for drug discovery. Mol. Pharmacol. 66, 1361–1371 (2004).
Bibikova, M. et al. Stimulation of homologous recombination through targeted cleavage by chimeric nucleases. Mol. Cell Biol. 21, 289–297 (2001).
Porteus, M.H. & Carroll, D. Gene targeting using zinc finger nucleases. Nat. Biotechnol. 23, 967–973 (2005).
Aslanidis, C. & de Jong, P.J. Ligation-independent cloning of PCR products (LIC-PCR). Nucleic Acids Res 18, 6069–6074 (1990).
Thibodeau, S.A., Fang, R. & Joung, J.K. High-throughput beta-galactosidase assay for bacterial cell-based reporter systems. Biotechniques 36, 410–415 (2004).
Miller, J.H. A Short Course in Bacterial Genetics. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1992).
Rebar, E.J. & Pabo, C.O. Zinc finger phage: affinity selection of fingers with new DNA-binding specificities. Science 263, 671–673 (1994).
Greisman, H.A. & Pabo, C.O. A general strategy for selecting high-affinity zinc finger proteins for diverse DNA target sites. Science 275, 657–661 (1997).
Isalan, M., Klug, A. & Choo, Y. A rapid, generally applicable method to engineer zinc fingers illustrated by targeting the HIV–1 promoter. Nat. Biotechnol. 19, 656–660 (2001).
Acknowledgements
We respectfully acknowledge the previously published work of Carlos Barbas' group, Sangamo Biosciences and ToolGen, Inc., which described and characterized the various zinc finger modules we used as the basis for our composite archive. We thank J.-S. Kim for providing the full amino acid sequences of the ToolGen human zinc finger modules. We thank members of our groups, especially P. Zaback and J. Townsend for helpful suggestions. J.D.S. is supported by USDA MGET 2001-52100-11506. A.S.H. was supported by NIH T32 CA09216. M.H.P is supported by the NIH (R01 HL0792595 and R21 CA120681). D.F.V. is supported by NSF grant DBI 0501678. J.K.J. is supported by the NIH (R01 GM069906 and R01 GM072621) and the MGH Department of Pathology. J.K.J. dedicates this protocol to the memory of Robert L. Burghoff, a patient teacher and friend who always knew how to make molecular biology experiments work. Note added in proof: Caroll, Segal and colleagues have recently described PCR-boned methods for assembling zinc finger modules into arrays and methods for purifying ZFNs and testing their activities in vitro (Caroll, D., Morton, J.J., Beumer, K.J. & Segal, D.J. Design, construction and in vitro testing of zinc finger nucleones. Nat. Protocols 3, 1329–1341).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Wright, D., Thibodeau-Beganny, S., Sander, J. et al. Standardized reagents and protocols for engineering zinc finger nucleases by modular assembly. Nat Protoc 1, 1637–1652 (2006). https://doi.org/10.1038/nprot.2006.259
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.259
This article is cited by
-
An intracellular membrane protein GEP1 regulates xanthurenic acid induced gametogenesis of malaria parasites
Nature Communications (2020)
-
Systemic loss of Sarm1 protects Schwann cells from chemotoxicity by delaying axon degeneration
Communications Biology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.