Abstract
Selective dopamine D3 receptor (D3R) antagonists prevent reinstatement of drug-seeking behavior and decrease the rewarding effects of contextual cues associated with drug intake preclinically, suggesting that they may reduce drug craving in humans. GSK598809 is a selective D3R antagonist recently progressed in Phase I trials. The aim of this study was to establish a model, based on the determination of the occupancy of brain across species, to predict the ability of GSK598809 to reduce nicotine-seeking behavior in humans, here assessed as cigarette craving in smokers. Using ex vivo [125I](R)-trans-7-hydroxy-2-[N-propyl-N-(3′-iodo-2′-propenyl)amino] tetralin ([125I]7OH-PIPAT) autoradiography and [11C]PHNO positron emission tomography, we demonstrated a dose-dependent occupancy of the D3Rs by GSK598809 in rat, baboon, and human brains. We also showed a direct relationship between and pharmacokinetic exposure, and potencies in line with the in vitro binding affinity. Likewise, GSK598809 dose dependently reduced the expression of nicotine-induced conditioned place preference (CPP) in rats, with an effect proportional to the exposure and at every time point, and 100% effect at values ⩾72%. In humans, a single dose of GSK598809, giving submaximal levels (72–89%) of , transiently alleviated craving in smokers after overnight abstinence. These data suggest that either higher is required for a full effect in humans or that nicotine-seeking behavior in CPP rats only partially translates into craving for cigarettes in short-term abstinent smokers. In addition, they provide the first clinical evidence of potential efficacy of a selective D3R antagonist for the treatment of substance-use disorders.
Similar content being viewed by others
INTRODUCTION
The dopamine (DA) D3 receptor (D3R) is selectively expressed in important areas of the brain implicated in addiction, including the ventral striatum (VST), midbrain, and pallidum (Bouthenet et al, 1991; Staley and Mash, 1996). These findings, together with data demonstrating plasticity changes in drug-dependent subjects, such as the increase in D3R density in cocaine addicts and metamphetamine polydrug users (Staley and Mash, 1996; Boileau et al, 2012), have generated interest in developing selective D3R antagonists as potential pharmacotherapeutics for addiction (Heidbreder and Newman, 2010). Although selective D3R antagonists fail to alter significantly drug self-administration (S/A) under low fixed-ratio (FR) schedules of reinforcement in rodents, they are very potent in decreasing drug S/A when the unit dose of the drug is decreased, when the work demand for the drug is increased (eg, from an FR1 to FR10 schedule or in progressive-ratio schedules of reinforcement), or when drug-seeking behavior is maintained by a drug-associated conditioned reinforcer (see Heidbreder and Newman, 2010). Selective D3R antagonists are also potent in preventing cue-, drug-, or stress-induced reinstatement of drug-seeking behavior, and reducing the acquisition and/or expression of drug-induced conditioned place preference (CPP; Heidbreder and Newman, 2010). Taken together, these observations suggest that D3R antagonists do not alter the primary reinforcing properties of drugs of abuse. Instead, they appear to (i) regulate motivation to self-administer drugs under schedules of reinforcement that require an increase in work demand and (ii) disrupt responsiveness to stimuli that have an important role in reinstatement of drug-seeking behavior. The latter may include re-exposure to the drug itself, exposure to environmental cues previously associated with drug-taking behavior, and stress.
In humans, drug craving (the desire or urge to take the drug in response to conscious or unconscious stimuli associated with drug taking) is an important factor of the stage of the addiction cycle in which the individual reinstates drug-seeking behavior after abstinence (the preoccupation/anticipation stage; Koob and Volkow, 2010). Assuming that craving is a fundamental drive of relapse in humans, and knowing that conditioned-drug effects have a prominent role in cue-elicited craving (O’Brien et al, 1998; Bedi et al, 2011), it is reasonable to think that selective D3R antagonists may decrease craving and, therefore, act as relapse prevention medications. The hypothesis that selective D3R antagonists may reduce drug craving in humans, however, has not been tested previously because of the lack of selective D3R antagonists with appropriate profiles for clinical investigation. Moreover, the complexity, long duration, and high cost of relapse prevention trials may delay the possibility to perform Phase II studies. Small, exploratory efficacy studies, in which dosing is limited to avoid exposing human subjects to potentially unsafe concentrations, are an alternative approach to determine if selective D3R antagonists warrant further investigation as potential medications to prevent relapse.
Previous research has shown that GSK598809 (derivative 74 in Micheli et al, 2010) is a selective and potent competitive D3R antagonist with good physicochemical, pharmacokinetic (PK), and safety properties, which dose dependently decreased the expression of nicotine-induced CPP in rats. The first objective of this work was to evaluate the relationship between the ability of GSK598809 to reduce nicotine-seeking behavior in rats and decrease cigarette craving in short-term abstinent smokers, based on the occupancy of brain as a marker of efficacy for translation across species. The secondary aim of this study was to provide information to define the doses to be tested in further, more extensive Phase II studies. The was determined by ex vivo [125I]7OH-PIPAT autoradiography in rat and by [11C]PHNO positron emission tomography (PET) in baboon (to have a preliminary evaluation of the technique) and human.
An introductory overview of this work has been presented at the American College of Neuropsychopharmacology (ACNP) 50th Annual Meeting (Merlo Pich, 2011). Some of the data reported in this work have been published previously (Micheli et al, 2010; Searle et al, 2010).
MATERIALS AND METHODS
Study 1: In Vitro Pharmacological Profile
[35S]Guanosine-5′-(γ-thio)triphosphate ([35S]GTPγS) binding was performed as described by Micheli et al (2010), with the following changes: membranes were from a Chinese Hamster Ovary (CHO) cell line expressing human D3Rs (hD3Rs) after induction with mifrepistone (hD3R-CHO/inducible) and the buffer solution contained 0.02% pluronic F-127, 30 μg/ml saponin, and 10 μM GDP. [3H]-(+)-2H-naphth[1,2-b]-1,4-oxazin-9-ol, 3,4,4a,5,6,10b-hexahydro-4-propyl-([3H]PHNO) binding was carried out on membranes from CHO cell lines stably expressing hD3Rs (hD3-CHO/stable) or the long variant of human D2Rs (hD2L-CHO/stable). To mimic the physiological conditions of PET-[11C]PHNO experiments, incubation was performed at 37 °C (for 60 min) in artificial cerebral spinal fluid: 124 mM NaCl, 1.9 mM KCl, 2 mM MgSO4, 1.3 mM KH2PO4, 11 mM glucose, 25 mM NaHCO3, 1.2 mM CaCl2 (pH 7.4). [125I]7OH-PIPAT binding was carried out as described by Micheli et al (2010), with the following changes: membranes were from a CHO cell line stably expressing rat D3Rs (rD3-CHO/stable) and incubation was performed at room temperature (RT), as in the experiments aimed at determining the by means of ex vivo autoradiography. GSK598809 pKi and pA2 values were determined using the method of Arunlakshana and Schild (1959), and Koff and Kon with that of Motulsky and Mahan (1984).
In Vivo Studies
All in vivo studies were pre-reviewed and approved by a local animal care committee in accordance with the guidelines of the ‘Principles of Laboratory Animal Care’ (NIH publication no. 86-23, revised 1985) and with a project license that was obtained according to the Italian law (Art. 7, Legislative Decree no. 116, 27 January 1992), which acknowledges European Directive 86/609/EEC on the care and welfare of laboratory animals.
A preliminary investigation of GSK598809 PKs following intraperitoneal administration in rat showed that the total GSK598809 concentration in blood (Ct,blood) reached the peak concentration (Ct,bloodmax) after 20 min (Tmax), and was then relatively steady for 40 min (Ct,blood at 1 h=84±5% Ct,bloodmax; mean±SD, n=3), halved at 2 h (Ct,blood at 2 h=47±16% Ct,bloodmax), and was almost undetectable after 24 h (Ct,blood at 24 h=1±0.3% Ct,bloodmax).
Study 2: D3R Occupancy in Rat
In the first experiment (dose–response), rats received an intraperitoneal administration of vehicle (saline) or GSK598809 (range 0.01–3 mg/kg) and were then terminated by decapitation after 1 h; in the second experiment (time–course), rats received vehicle or GSK598809 (1 mg/kg) and were terminated at different times between 0.75 and 24 h. Blood sampling, for GSK598809 quantification, was performed from the tail vein immediately before decapitation. After discarding the cerebellum, the brain was divided along the longitudinal fissure: one hemisphere was used for GSK598809 quantification in the brain tissue, and the other was frozen in isopentane, and subsequently cut into 14-μm-thick coronal hemisections at +2.70, +2.20, and +1.60 mm from bregma. [125I]7OH-PIPAT autoradiography was performed with a protocol derived from Burris et al (1994) and Stanwood et al (2000), with some modifications. Briefly, sections were rinsed (5 min, RT) in a solution consisting of 50 mM Tris buffer (pH=7.4), 40 mM NaCl, and 300 μM guanosine 5′-triphosphate (GTP). Incubation (15 min, RT) with 0.4 nM [125I]7OH-PIPAT was performed in a solution consisting of 50 mM Tris buffer (pH=7.0), 40 mM NaCl, 300 μM GTP, and 5 μM 1,3-di(2-tolyl)guanidine. Nonspecific binding was determined in the presence of 10 μM DA. The reaction was stopped by washing (90 min, 4 °C) the slides in 40 mM NaCl/50 mM Tris buffer (pH=7.4). After a dip in water (4 °C), sections were dried and put in contact to Fuji Imaging Plates BAS-SR2025 overnight. The plates were read at the Bio-image Analyzer BAS5000 (Fuji Photo Film, Japan). Preliminary experiments (not reported) were performed to assess the specificity of [125I]7OH-PIPAT binding to D3Rs in these conditions and reduce GSK598809 dissociation from the receptor to minimize the risk of underestimating occupancy (Li et al, 2006).
Quantitative analysis was performed by computer-assisted microdensitometry (MCID basic; Imaging Research, Canada). Photostimulated luminescence per mm2 values were converted to the corresponding radioligand concentration (fmol/mg) by referring to iodine standards (Microscales; Amersham Biosciences) on the same plate. [125I]7OH-PIPAT binding was measured in the shell of the nucleus accumbens (AcbSh), a subregion of the VST containing high levels of D3Rs. The , numerically equivalent to the occupancy of the [125I]7OH-PIPAT binding sites (OPIPAT) in these binding conditions, was calculated with the following equation:

where  is the specific binding (SB) in the vehicle-treated animals (mean value) and SBGSK598809 the SB in the GSK598809-treated animal. Occupancy data were fit by nonlinear regression analysis using the GraphPad Prism 4.0 (GraphPad Software) with the following equations:
is the specific binding (SB) in the vehicle-treated animals (mean value) and SBGSK598809 the SB in the GSK598809-treated animal. Occupancy data were fit by nonlinear regression analysis using the GraphPad Prism 4.0 (GraphPad Software) with the following equations:

where Ct,blood and Ct,brain are the total concentration in the blood and brain, respectively, OCt,blood50 and OCt,brain50 are the total concentration, giving 50% of maximal occupancy , in the blood and brain, respectively, and γ the Hill coefficient of this function.
Study 3: Nicotine CPP in Rat
Similar to the RO study, two different CPP experiments were performed (dose–response and time–course). Animal husbandry, the apparatus, and the first three phases (acclimation, handling, and conditioning) of the nicotine CPP procedure were exactly as described in Micheli et al (2010). In the last phase (testing), rats from the dose–response experiment received vehicle or GSK598809 (range 0.05–3 mg/kg, intraperitoneally) 30 min before they were placed in the apparatus. In the time–course experiment, rats received vehicle or GSK598809 (range 0.3–3.0 mg/kg, intraperitoneally) 4 or 8 h before they were placed in the apparatus. In both experiments, the animals were allowed to remain in the apparatus for 15 min, and the time spent in each chamber was automatically recorded (see Micheli et al, 2010).
The conditioning properties of nicotine were quantified as preference index (PI), defined as the time spent in the nicotine-paired chamber minus the time spent in the unpaired chamber. The ability of the antagonist (GSK598809) to reduce nicotine preference, or inhibitory effect (E), was calculated with the following equation:

in which  is the PI in the vehicle-treated animals (mean value) and PIGSK598809 the PI in the GSK598809-treated animal. Data were fit using a nonlinear regression analysis (GraphPad Prism 4.0) as follows:
is the PI in the vehicle-treated animals (mean value) and PIGSK598809 the PI in the GSK598809-treated animal. Data were fit using a nonlinear regression analysis (GraphPad Prism 4.0) as follows:

where C̄t,blood and C̄t,brain are the total concentration (in the blood or brain, respectively) of the animals receiving the same dose of GSK598809 in the satellite RO experiment (median value), and ECt,blood50 and ECt,brain50 the total concentration, giving 50% of maximal effect (Emax), in the blood or brain, respectively, and γ the Hill coefficient.
Prediction of Human Values
GSK598809 concentrations in human blood were estimated from the rat experimental data with the following equation (assuming similar brain penetration across species):

in which (XC50)human and (XC50)rat are the concentration giving 50% of or E in human and rat, respectively, (Ki)human and (Ki)rat the affinity of GSK598809 for hD3R and rD3R, respectively, and (fu)human and (fu)rat the fraction unbound (ie, 100 minus the percentage of tissue binding) in human and rat blood, respectively.
Study 4: D3R Occupancy in Baboon and Human
The PET studies in baboon (Papio anubis) and human have been described previously (Searle et al, 2010). In brief, each subject received a baseline [11C]PHNO scan, followed by 60-min intravenous infusion of 0.1–1 mg/kg GSK598809 (baboon) or oral administration of 5–175 mg GSK598809 (human). After 15 min from the end of the infusion (baboon) or 2–3 h from the oral administration (human), the subjects received the second scan. Blood samples (for determination of GSK598809 concentration in the plasma) were taken at regular intervals. Image analysis was performed as described by Rabiner et al (2009; baboon) and Searle et al (2010; human). Quantitative analysis was performed using the simplified reference tissue model with cerebellum as reference region to derive regional estimates of [11C]PHNO binding potential relative to the non-displaceable compartment (BPND). The occupancy of [11C]PHNO binding sites by GSK598809 (OPHNO) was defined with the following equation:

where BPBaselineND is the BPND at the baseline scan and BPGSK598809ND the BPND measured in the second scan (ie, in the presence of GSK598809).
As previous analysis revealed that it is reasonable to assume that GSK598809 binding to D2Rs was negligible in the current set of data (Searle et al, 2010), we adopted a simplified procedure to estimate the potency of GSK598809 in occupying D3Rs. We applied a population PK/pharmacodynamic (PD) modeling approach to model individual data using a nonlinear mixed-effect method, as implemented in NONMEM VI (Globomax, Hanover, MD, USA). Data were fit with the following equation:

in which Ct,plasma is the total GSK598809 concentration in the plasma at the beginning of the scan, OCt,plasma50 the total GV598809 concentration, in the plasma, giving 50% of maximal occupancy of PHNO binding sites (OPHNOmax), and γ the Hill coefficient. In each region, the OPHNOmax value paralleled the fraction of D3Rs in the BPBaselineND in that region (Searle et al, 2010), although subtle differences were observed (see Results). The parameter OCt,plasma50, theoretically equivalent to the total GV598809 concentration, in the plasma, giving 50% of maximal occupancy of D3Rs , was shared between the VST, the globus pallidus (GB), and the substantia nigra (SN), three regions having both good value and BPND values. The proportion of OPHNO corresponding to occupancy of was calculated, in each point, with the following equation:

Dose Selection for the Human Efficacy Study
The dose of GSK598809 to be tested in the human efficacy study (Study 5) was selected by translating the results of the GSK598809 efficacy experiment in rat to human, using as a marker of efficacy: the dose selected was designed to ensure, in the time frame during which the efficacy on craving was studied in human, a minimal occupancy level similar to that reached by the lowest dose giving maximal (100%) effect in the rat efficacy study.
The value corresponding to the lowest dose of GSK598809 giving maximal effect in the rat nicotine CPP model (Study 3) was calculated from the PK/O relationship generated in the satellite ex vivo [125I]7OH-PIPAT autoradiography experiment (Study 2). The dose for the human study was selected on the basis of the PK/O relationship generated in the human PET-[11C]PHNO/GSK598809 occupancy experiment (Study 4) and the plasma concentration time profiles of previous single-dose PK studies.
Study 5: Efficacy in Human
For the efficacy study in human, a total of 48 treatment-seeking (ie, interested in quitting smoking), healthy male and female adult current smokers were recruited from one center, the Transdisciplinary Tobacco Use Research Centre (TTURC), Philadelphia (USA). Candidates with medical or psychiatric contraindications were excluded. Written informed consent was obtained from each subject before the performance of any study-specific procedures. A total of 40 subjects completed the study (24 males and 16 females; see Table 1 for participants’ characteristics). The study was sponsored by GlaxoSmithKline (GSK study number DAN106593) and approved by the University of Pennsylvania Institutional Review Board. The protocol was posted on http://clinicaltrial.gov (identifier NCT00605241) before study initiation.
The study was a randomized, double-blind, placebo-controlled, balanced two-way crossover design. Following screening, subjects participated in two dosing sessions (with a washout period of at least 7 days), in which they received a single oral dose of 75 mg GSK598809 or placebo. Each session was conducted in the clinic in the morning. Subjects were required to abstain from smoking for 14 h before attending the clinic. In each session visit, medical and safety assessments, blood sampling, recording of adverse events, and efficacy assessments were performed. Blood samples were collected at regular intervals for the analysis of total GSK598809 concentration in the plasma. Abstinence was confirmed by measuring exhaled carbon monoxide level and nicotine, cotinine, and trans-3-hydroxycotinine plasma levels at pre-dose.
Three different behavioral tasks were carried out to assess PD response to GSK598809, at three different times post-dose. Task 1, a modified version of the classic Stroop task (Waters et al, 2003), was carried out 30 min post-dose and lasted approximately 60 min. The difference in average response times to color-name words related to cigarette smoking (smoking reaction time, RT) and neutral control words (neutral RT) was quantified and considered an index of the degree to which smoking-related content disrupts ongoing cognitive processes. Task 2, a Behavioral Economic task, adapted from Perkins et al (2002), was carried out 90 min post-dose and lasted approximately 60 min. The level of effort participants were willing to expend for cigarette puffs or money was measured (as breakpoint in a progressive-ratio schedule) and considered an index of the relative reinforcing value of smoking. Task 3, a Cigarette Choice task (Blendy et al, 2005), was carried out to measure the relative reinforcing value of nicotine between 6.5 and 8.5 h post-dose. The dependent variable was the number of times a nicotine-containing cigarette was selected.
Participants also completed the following self-report questionnaires (aimed at determining the extent of self-reported craving) at 0.5, 1.25, 2.75, 5, 8, and 10 h post-dose: the Tiffany Smoking Urges-Brief (QSU-B; Cox et al, 2001) and the Minnesota Nicotine Withdrawal Scale (MNWS; Hughes, 2007). Finally, cigarette consumption in the natural environment subsequent to the experimental session (8.5–10.5 h post-dose) was also determined.
RESULTS
Study 1: In Vitro Pharmacological Profile
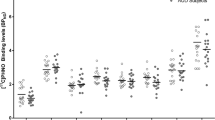
Without changing basal [35S]GTPγS levels in hD3R/inducible membranes, GSK598809 caused a parallel right-ward shift of the quinelorane-induced increase of [35S]GTPγS, with a potency value (A2) of 0.5 nM (pA2=9.3±0.1, n=3; Figure 1a). GSK598809 displaced [3H]PHNO binding to hD3R/stable and hD2LR/stable membranes, with Ki values of 6.2 nM (pKi=8.21±0.09, n=3) and 740 nM (pKi=6.13±0.02, n=3), respectively (not shown).
(a) Increase of basal [35S]guanosine-5′-(γ-thio)triphosphate ([35S]GTPγS) binding to (hD3R)/inducible cell membranes by the agonist quinelorane, alone or in the presence of increasing concentrations of GSK598809. (b) Association of [125I](R)-trans-7-hydroxy-2-[N-propyl-N-(3′-iodo-2′-propenyl)amino] tetralin ([125I]7OH-PIPAT) binding to rD3Rs/stable cell membranes, alone or in the presence of increasing concentrations of GSK598809. (c) Dissociation of [125I]7OH-PIPAT binding to rD3Rs/stable cell membranes induced by the addition of an excess of unlabeled ligand(s).
GSK598809 inhibited [125I]7OH-PIPAT binding to rD3R/stable membranes, with a Ki of 1.5 nM (pKi=8.83±0.004, n=3). Kinetic experiments showed that GSK598809 bound to rD3Rs with an association rate constant (Kon) value of 0.0508±0.0081/nM/min (n=3) and a dissociation rate constant (Koff) value of 0.2601±0.0616/min (n=3), respectively (see Figure 1b), from which it was calculated a kinetically derived dissociation constant value of 5.1 nM (pKD=8.29). Half-time (t1/2) values of few minutes for association and 3 min for dissociation were calculated from such rate constant values. GSK598809 did not change the dissociation kinetics of the radioligand, confirming the competitive interaction with the receptor (see Figure 1c).
Study 2: D3R Occupancy and PK/O Relationship in Rat
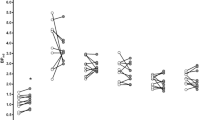
Specific [125I]7OH-PIPAT binding was higher in the brain of vehicle-treated rats than that of GSK598809-treated animals (Figure 2a). Data fitting revealed that GSK598809 occupied D3Rs (Figure 2b) with an OCt,blood50 of 15 ng/ml and an OCt,brain50 of 47 ng/g, with Hill coefficient values not significantly different from 1, and values not significantly different from 100%. Accounting for tissue binding, it was calculated that the free (unbound) concentration giving 50% of occupancy (OCU50) was 0.6 ng/ml (≈1.2 nM; pOCu,blood50=8.91) and 1.1 ng/g (≈2.3 nM; pOCu,brain50=8.64) in the blood and brain, respectively (see Table 2). The decreased with time after GSK598809 administration, in parallel to Ct,blood and Ct,brain (see Figure 2c).
(a) Ex vivo [125I](R)-trans-7-hydroxy-2-[N-propyl-N-(3′-iodo-2′-propenyl)amino] tetralin ([125I]7OH-PIPAT) binding to representative, coronal, one-hemisphere brain sections from rats treated with vehicle or GSK598809 (3 mg/kg, intraperitoneally (i.p.)). (b) Occupancy of [125I]7OH-PIPAT binding sites (=) in the rat shell of the nucleus accumbens (AcbSh) at increasing blood (●; ng/ml) and brain (○; ng/g) GSK598809 concentrations, 1 h after administration of GSK598809 (0.05–3 mg/kg intraperitoneally). (c) Time–course of the in the rat AcbSh (●) after a single administration of GSK598809 (1 mg/kg intraperitoneally); the figure also shows GSK598809 concentration in the blood (dotted line; ng/ml) and brain (dashed line; ng/g).
Study 3: Efficacy in Nicotine CPP and PK/PD Relationship in Rat
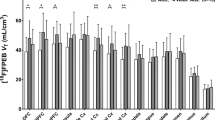
Micheli et al (2010) reported previously that GSK598809 dose dependently reduced the expression of nicotine-conditioned preference (see also Figure 3a). Data fitting vs compound exposure (as from the animals treated with the same doses in the previous PK/O experiment) indicated that GSK598809 reduced the expression of nicotine CPP with ECt,blood50 and ECt,brain50 values of 7.2 ng/ml and 21.4 ng/g, respectively, γ values of 1.7 and 2.1, respectively, and Emax values not significantly different from 100 (not shown). Considering tissue binding, it was calculated that the free concentration giving 50% of effect (ECu50) was 0.3 ng/ml (≈0.6 nM; pECu,blood50=9.23) and 0.5 ng/ml (≈1.0 nM; pECu,brain50=8.98) in the blood and brain, respectively.
Expression of nicotine-induced conditioned place preference (CPP) in rat and dose-dependent inhibition by GSK598809 at different pre-treatment times. (a) Pre-treatment time was 30 min (preference index (PI) values were calculated from data already reported in Micheli et al (2010)). (b) Pre-treatment time was 4 or 8 h, as shown in the figure. *, **, and ***P<0.01, 0.001, and 0.0001, respectively; U, unpaired rats.
The effect was lower when GSK598809 was administered 4 h before the test, and significantly different from control only at 1 and 3 mg/kg (Figure 3b). The same doses of GSK598809 had no effect when administered 8 h before the test.
Prediction of Human Values
From the rat OCt,blood50 determined in Study 2, it was possible to predict that the OCt,blood50 in human was 54 ng/ml (see Materials and Methods). By adjusting for the blood/plasma ratio in human, it was calculated that the predicted total concentration, in the plasma, to obtain 50% of occupancy in human (OCt,plasma50) was 87 ng/ml (see Table 2). In the same manner, from the rat ECt,blood50 determined in Study 3, it was possible to predict that the ECt,blood50 in human was 26 ng/ml and that the predicted total concentration, in the plasma, to obtain 50% of efficacy in human (ECt,plasma50) was 42 ng/ml.
Study 4: D3R Occupancy and PK/O Relationship in Baboon and Human
Data fitting revealed that GSK598809 occupied baboon brain D3Rs (see Figure 4) with an ECt,plasma50 of 36 ng/ml (see Table 2). These results showed that the potency of GSK598809 in occupying brain D3Rs in baboon was not far from that predicted for the human (ECt,plasma50=87 ng/ml) from the studies in rat, encouraging further studies in human.
Occupancy of in vivo [11C]-(+)-2H-naphth[1,2-b]-1,4-oxazin-9-ol, 3,4,4a,5,6,10b-hexahydro-4-propyl-([11C]PHNO) binding sites (OPHNO) in the baboon and human ventral striatum (VST) (a), globus pallidus (GP) (b), and substantia nigra (SN) (c) at increasing plasma GSK598809 concentrations (at the beginning of the scan), as revealed by positron emission tomography (PET) methodology (original values were partially already reported in Searle et al (2010)).
GSK598809 occupied human brain D3Rs (see Figure 4) with an OCt,plasma50 of 66 ng/ml. Using blood/plasma ratio and blood tissue binding in human (see Table 2), it was estimated that the blood free concentration giving 50% of occupancy in human (OCu,blood50) was 1.9 ng/ml (≈3.9 nM; pOCu,blood50=8.41).
In each region, the OPHNOmax value was in line with the PHNO value estimated by Searle et al (2010). In the SN, OPHNOmax was 77% (95% confidence interval (CI): 70–84%), slightly lower than the 100% (95% CI: 79–100%) value reported by Searle et al (2010).
GSK598809 Dose Selection for the Human Efficacy Study
The lowest dose of GSK598809 giving maximal effect in the rodent nicotine CPP model (Study 3) was 0.3 mg/kg (see Figures 3a). Rats from the same group (0.3 mg/kg) of the satellite occupancy experiment (Study 2) had a median total GSK598809 blood concentration of 37.7 ng/ml, which corresponded to an value of 72% in the rat PK/O curve. From the PK/O relationship generated by the human PET-[11C]PHNO/GSK598809 occupancy experiment (in which an OCt,plasma50 of 66 ng/ml was determined), it was calculated that a similar occupancy value (=72%) was reached at a plasma concentration of 175 ng/ml. Previous single-dose PK studies in healthy volunteers and smokers had shown that comparable (or higher) plasma concentrations were obtained with a 75 mg oral dose in the time window between 0.5 and 10 h after administration. Therefore, the human single oral dose of 75 mg GSK598809 was chosen to yield, at the time of behavioral testing, a slightly higher to the lowest dose of GSK598809 giving maximal effect in the rat nicotine CPP experiment.
Study 5: Efficacy in Human
Following administration of an oral dose of 75 mg, GSK598809 was absorbed with a median Tmaxof 1.5 h (range from 0.5 to 8.2 h) and a mean t1/2 value of 17 h. The maximum concentration in the plasma (Ct,plasmamax) was 520 ng/ml. From these values of actual concentration in the plasma and the PK/O relationship established with the PET studies, it was estimated that the passed from zero at pre-dose to 89% at the Tmax (1.5 h) and then decreased slowly to 80% at 17 h.
In the smoking Stroop test (carried out between 0.5 and 1.5 h post-dose), the difference between the smoking RT and the neutral RT was 47.52 ms in the GSK598809 treatment condition, lower (−20 ms; 95% CI:−65, 25) than the placebo treatment condition (67.52 ms). One subject (Subject 29, treated with GSK598809) who had clear difficulties in performing the task, as demonstrated by the very low percentage (<20%) of correct scores in the incongruent word group and the longest reaction time across all the other word groups (congruent, neutral, smoking) compared with other subjects, was considered an outlier and excluded from the analysis.
There were no significant treatment-related effects of GSK598809 on performance of the Behavioral Economic task (total number of presses made for puffs of a cigarette, or a trend across trials for switching behavior) and the Cigarette Choice paradigm (number of puffs from nicotinized cigarette).
There was a significant effect of GSK598809 on responses to MNWS-R item 4 (‘desire or craving to smoke’) at 30 min post-dose: mean (SD)=2.30 (1.07) for placebo, 1.82 (1.28) for drug, t (39 d.f.)=2.21, P=0.033 (paired t-test). The comparison of responses to MNWS-R item 4 at 30 min post-dose was repeated using a nonparametric test (Wilcoxon’s matched-pairs signed-rank test) and the effect remained significant (Z=−2.25, P=0.024). There was no significant GSK598809 effect on summary scores for the MNWS-R. A significant effect of GSK598809 on response to the QSU-brief, factor 1 (‘desire and intention to smoke, with smoking anticipated as pleasurable’) scores was found at 30 min post-dose: mean (SD)=27.22 (7.09) for placebo, 25.35 (7.92) for GSK598809; t (39 d.f.)=2.06, P=0.046 (paired t-test). There was no effect of GSK598809 on factor 2 (‘anticipation of relief from negative affect and nicotine withdrawal, with an urgent desire to smoke’) of the QSU-brief, or on any craving measures at the later assessment points.
Finally, 8.5–10.5 h after smoking, subjects took, on average, 4.05 total puffs more during ad lib smoking when on GSK598809 compared with placebo (95% CI: 1.67–6.43). Similarly, subjects smoked 0.35 cigarettes more during ad lib smoking when on GSK598809 compared with placebo (95% CI: 0.12–0.59).
DISCUSSION
In this work we show, for the first time, that a selective D3R antagonist modifies cigarette craving in short-term abstinent smokers, partially validating the D3R as a target for the treatment of substance-use disorders. In addition, we establish a relationship between the ability of GSK598809 to reduce nicotine-seeking behavior preclinically and decrease craving in humans, based on the occupancy of brain D3Rs () and provide a model to early predict the right doses of novel D3R antagonists for the treatment of addiction in larger Phase II clinical trials.
Historically, technical issues related to the low density of D3Rs, binding occlusion by endogenous DA, and low specific activity of radioligands, have hampered the determination of after systemic administration of dopaminergic ligands (Schotte et al, 1996; Langlois et al, 2005). In this work, we have first confirmed that GSK598809 is a high-affinity, competitive antagonist at D3Rs, with more than 100-fold selectivity over D2LRs and fast association and dissociation kinetics. Then, we have developed an ex vivo [125I]7OH-PIPAT autoradiography protocol for the measurement of and showed that GSK598809 occupies brain D3Rs with a direct (time-independent) relationship between exposure and occupancy, and with a potency, expressed as a function of the bio-phase concentration (OCu,brain50=2.3 nM), in agreement with the binding affinity (Ki=1.5 nM for rD3R). Likewise, we showed that GSK598809 dose dependently reduces the expression of nicotine-induced CPP, with an effect directly related to the exposure and with a similar potency (ECu,brain50=0.6 nM).
Various studies have shown that selective D3R antagonists reduce the expression of heroin-, cocaine-, metamphetamine-, and nicotine-induced CPP and prevent reinstatement to alcohol-, cocaine-, metamphetamine-, and nicotine-seeking behavior (Heidbreder and Newman, 2010), but correlation with the has never been reported. Here we show a strong relationship between the exposure of GSK598809, the in an important area of the addiction circuit (the AcbSh, a main neuroanatomical target of addictive drugs) and the efficacy in an animal model of drug-seeking behavior (the nicotine CPP).
By fitting GSK598809 plasma concentration and the D3Rs component of [11C]PHNO occupancy from our previous PET study (Searle et al, 2010), it was possible to show that GSK598809 occupied human brain D3Rs with an OCt,plasma50 of 66 ng/ml, in line with the value predicted from the rat RO studies (OCt,plasma50=87 ng/ml). In addition, we found an OCu,blood50 value of 3.9 nM, in line with GSK598809 affinity for hD3Rs (Ki=6.2 nM), suggesting that the free drug hypothesis (for a freely diffusible molecule that passively permeates through the blood–brain barrier and is not excluded or uptaken by carrier-mediated systems) is valid for GSK598809 and that the Cu,blood can be reasonably considered as a surrogate for the bio-phase concentration (ie, the Cu,brain; Read and Braggio, 2010). Overall, these results showed that the PK/O relationship was consistent between the rat, baboon, and human species, and that was a biomarker with good translational value.
As a limitation of our approach, however, prediction of GSK598809 potency in human was based on Ki values determined with different radioligands (namely [125I]7OH-PIPAT for rD3R and [3H]PHNO for hD3R) and different binding conditions. Even if these radioligands present similar properties (in that they are both high-affinity, dual D3R/D2R agonists), more accurate predictions might be obtained by measuring the affinity of GSK598809 for the rD3R and hD3R with exactly the same binding assay. In addition, our method of estimating the potency in occupying D3Rs in human, based on the simultaneous, one-site fitting of the data from the regions containing consistent amount of D3Rs, produced slightly different results from those of Searle et al (2010), who performed a simultaneous, two-site fitting of all data (including the data from the regions containing a substantial amount of D2Rs, or very low amounts of [11C]PHNO binding). Interestingly, we found that the maximal occupancy of [11C]PHNO sites by GSK598809 in the SN was slightly lower than the 100% value reported previously (Searle et al, 2010), suggesting the presence of D2Rs also in this region, as reported in human post-mortem studies (Murray et al, 1994; Gurevich and Joyce, 1999).
In the light of the results obtained in the occupancy studies, we selected the lowest GSK598809 dose that would reach values giving maximal effects in the rat nicotine CPP model (ie, ⩾72%), and tested its effect in human, to make a parallel between this preclinical paradigm of nicotine-seeking behavior and commonly used human behavioral investigations to measure craving and reward.
In the smoking Stroop test, abstinent smokers took significantly longer to color-name words related to cigarette smoking than to color-name neutral control words, confirming previous findings that abstinence decreases the ability to ignore the meaning of smoking-related information (Gross et al, 1993). This attentional bias is considered a mark of craving during abstinence. It is worth noting that the difference between the smoking and neutral RT was lower (although not significantly) in the group treated with a single oral dose of 75 mg GSK598809, suggesting that GSK598809 partially reversed the attentional bias of abstinent smokers. In addition, GSK598809 significantly decreased subjective craving (MNWS-R item 4 and QSU-brief factor 1) shortly after administration, but such effects were not maintained at later time points. Overall, these findings were in agreement with the studies reporting an association of the smoking Stroop effects and self-reported craving (Mogg and Bradley, 2002; Mogg et al, 2003; Zack et al, 2001), and suggest that GSK598809 was effective in decreasing craving. Yet, the same findings partially disagree with the effects observed in the rat CPP model, in which the level of reasonably paralleled the degree of efficacy in blocking the rewarding properties of nicotine-associated cues at any time point. In the first place, this difference may suggest that possibly higher levels (ie, higher than the 72–89% range estimated in this study), maintained over time, are needed to obtain a robust effect on craving in shortly abstinent smokers. Alternatively, it is worth to consider that nicotine CPP in rat is only a clearcut measure of a process (incentive motivation) in which place-related cues (conditioned stimuli) previously associated to nicotine intake (unconditioned stimuli) allow the animal to seek out and anticipate the reward related to nicotine intake in that specific environment. In smokers, many other factors may affect the intensity of craving, from the variety of cues previously associated with their individual smoking habit, to the length of their smoking history and/or severity of nicotine dependence, which might explain the lower efficacy of GSK598809 in blocking craving in human. Finally, it should be noted that, in contrast to the expression of CPP model, where rats were abstinent from 24 h and in a completely nicotine-free condition, the smokers in this study were abstinent only overnight (14 h) and self-administered nicotine during the Cigarette Choice paradigm, which they knew they would be doing later in the morning. Craving in the subjects of this study, therefore, might be different from the craving of smokers who have been abstinent for a longer time and/or are completely free of nicotine.
The mechanisms by which selective D3R antagonists partially reverse the attentional bias of abstinent smokers in the Stroop test are unknown. Recent preclinical studies suggest that blockade of D3Rs facilitates cholinergic transmission in the frontal cortex and have pro-cognitive effects (Lacroix et al, 2003; Millan et al, 2007; Glickstein et al, 2005; Laszy et al, 2005; Loiseau and Millan, 2009), which may explain the improved performance of GSK598809-treated patients in the Stroop test. Interestingly, not only acetylcholine but also DA was reported to increase in the frontal cortex following D3R antagonist administration (Lacroix et al, 2003). Considering that lower DA levels are observed after a prolonged period of drug dependence (Volkow et al, 2009), it can be hypothesized that D3Rs antagonists may restore DA levels to normal or, interfering with the cortical glutamatergic signaling to the Acb and ventral tegmental area (Surmeier et al, 2007; Kalivas et al, 2009), decrease the salience of smoke-associated cues in the Stroop test.
No differences were detected between a single oral dose of 75 mg GSK598809 and placebo when measuring the relative reinforcing value of smoking and nicotine with the Behavioral Economic task and Cigarette Choice paradigm. Nevertheless, GSK598809-treated subjects, as compared with placebo, slightly increased cigarette consumption and puffs/cigarette when subjects were allowed to smoke freely in the natural environment subsequent to the experimental session, suggesting less satisfaction from the same number of puffs and compensatory increases.
Overall, these results suggest that when a single dose of GSK598809 is administered to smokers, it does not affect the rewarding properties of nicotine per se (in line with preclinical findings showing that selective D3R antagonists do not interfere with the primary reinforcing properties of drugs of abuse), while reducing self-reported craving and slightly increasing smoking, probably as a compensatory mechanism. Further experiments, possibly in different types of smokers (interested and not interested in quitting smoking) are needed to understand if D3R antagonists may decrease the rewarding properties of nicotine in specific circumstances in humans.
In conclusion, we found that GSK598809 PK exposure is predictive of brain at any time point, in both rat and human species. In rats repeatedly exposed to nicotine and abstinent for 24 h, the level of produced by a single dose of GSK598809 reasonably paralleled the degree of efficacy in blocking nicotine-seeking behavior driven by associated cues. In humans, a single oral dose of 75 mg GSK598809 estimated to reach submaximal levels of only partially alleviated craving in smokers abstinent for 14 h. These data suggested that higher and persistent by a selective DA D3R antagonist or additional effects on other biological mechanisms are needed to more efficiently block craving for cigarettes in smokers under transient (hours) abstinence in humans. Studies with long-term abstinent smokers motivated to quit, with repeated dose treatment designed to achieve higher and more sustained levels of , and incorporating tests that better reflect the cue associative nature of the preclinical CPP test, are probably needed to further investigate the role of GSK598809 or other DA D3R antagonists in human craving.
References
Arunlakshana O, Schild HO (1959). Some quantitative uses of drug antagonists. Br J Pharmacol Chemother 14: 48–58.
Bedi G, Preston KL, Epstein DH, Heishman SJ, Marrone GF, Shaham Y et al (2011). Incubation of cue-induced cigarette craving during abstinence in human smokers. Biol Psychiatry 69: 708–711.
Blendy JA, Strasser A, Walters CL, Perkins KA, Patterson F, Berkowitz R et al (2005). Reduced nicotine reward in obesity: cross-comparison in human and mouse. Psychopharmacology (Berl) 180: 306–315.
Boileau I, Payer D, Houle S, Behzadi A, Rusjan PM, Tong Y et al (2012). Higher binding of the dopamine D3 receptor-preferring ligand [11C]-(+)-propyl-hexahydro-naphtho-oxazin in methamphetamine polydrug users: a positron emission tomography study. J Neurosci 32: 1353–1359.
Bouthenet ML, Souil E, Martres MP, Sokoloff P, Giros B, Schwartz JC (1991). Localization of dopamine D3 receptor mRNA in the rat brain using in situ hybridization histochemistry: comparison with dopamine D2 receptor mRNA. Brain Res 564: 203–219.
Burris KD, Filtz TM, Chumpradit S, Kung MP, Foulon C, Hensler JG et al (1994). Characterization of [125I](R)-trans-7-hydroxy-2-[N-propyl-N-(3′-iodo-2′-propenyl)amino] tetralin binding to dopamine D3 receptors in rat olfactory tubercle. J Pharmacol Exp Ther 268: 935–942.
Cox LS, Tiffany ST, Christen AG (2001). Evaluation of the brief questionnaire of smoking urges (QSU-brief) in laboratory and clinical settings. Nicotine Tob Res 3: 7–16.
Glickstein SB, Desteno DA, Hof PR, Schmauss C (2005). Mice lacking dopamine D2 and D3 receptors exhibit differential activation of prefrontal cortical neurons during tasks requiring attention. Cereb Cortex 15: 1016–1024.
Gross TM, Jarvik ME, Rosenblatt MR (1993). Nicotine abstinence produces content-specific Stroop interference. Psychopharmacology (Berl) 110: 333–336.
Gurevich EV, Joyce JN (1999). Distribution of dopamine D3 receptor expressing neurons in the human forebrain: comparison with d2 receptor expressing neurons. Neuropsychopharmacology 20: 60–80.
Heidbreder CA, Newman AH (2010). Current perspectives on selective dopamine D(3) receptor antagonists as pharmacotherapeutics for addictions and related disorders. Ann N Y Acad Sci 1187: 4–34.
Hughes JR (2007). Measurement of the effects of abstinence from tobacco: A qualitative review. Psychol Addict Behav 21: 127–137.
Kalivas PW, Lalumiere RT, Knackstedt L, Shen H (2009). Glutamate transmission in addiction. Neuropharmacology 56 (Suppl 1): 169–173.
Koob GF, Volkow ND (2010). Neurocircuitry of addiction. Neuropsychopharmacology 35: 217–238.
Lacroix LP, Hows ME, Shah AJ, Hagan JJ, Heidbreder CA (2003). Selective antagonism at dopamine D3 receptors enhances monoaminergic and cholinergic neurotransmission in the rat anterior cingulate cortex. Neuropsychopharmacology 28: 839–849.
Langlois X, te Riele P, Ashton D (2005). In vivo receptor occupancy profile of aripiprazole in rat brain. Soc Neurosci Abstracts 444: 3.
Laszy J, Laszlovszky I, Gyertyán I (2005). Dopamine D3 receptor antagonists improve the learning performance in memory-impaired rats. Psychopharmacology (Berl) 179: 567–575.
Li J, Fish RL, Cook SM, Tattersall FD, Atack JR (2006). Comparison of in vivo and ex vivo [3H]flumazenil binding assays to determine occupancy at the benzodiazepine binding site of rat brain GABAA receptors. Neuropharmacology 51: 168–172.
Loiseau F, Millan MJ (2009). Blockade of dopamine D(3) receptors in frontal cortex, but not in sub-cortical structures, enhances social recognition in rats: similar actions of D(1) receptor agonists, but not of D(2) antagonists. Eur Neuropsychopharmacol 19: 23–33.
Merlo Pich E (2011). Translational approach to dopamine D3 receptor: from mechanism of action to clinical studies. Neuropsychopharmacology 36: S13.
Micheli F, Arista L, Bonanomi G, Blaney FE, Braggio S, Capelli AM et al (2010). 1,2,4-Triazolyl azabicyclo[3.1.0]hexanes: a new series of potent and selective dopamine D(3) receptor antagonists. J Med Chem 53: 374–391.
Millan MJ, Di Cara B, Dekeyne A, Panayi F, De Groote L, Sicard D et al (2007). Selective blockade of dopamine D(3) versus D(2) receptors enhances frontocortical cholinergic transmission and social memory in rats: a parallel neurochemical and behavioural analysis. J Neurochem 100: 1047–1061.
Mogg K, Bradley BP (2002). Selective processing of smoking-related cues in smokers: manipulation of deprivation level and comparison of three measures of processing bias. J Psychopharmacol 16: 385–392.
Mogg K, Bradley BP, Field M, De Houwer J (2003). Eye movements to smoking-related pictures in smokers: relationship between attentional biases and implicit and explicit measures of stimulus valence. Addiction 98: 825–836.
Motulsky HJ, Mahan LC (1984). The kinetics of competitive radioligand binding predicted by the law of mass action. Mol Pharmacol 25: 1–9.
Murray AM, Ryoo HL, Gurevich E, Joyce JN (1994). Localization of dopamine D3 receptors to mesolimbic and D2 receptors to mesostriatal regions of human forebrain. Proc Natl Acad Sci USA 91: 11721–11275.
O’Brien CP, Childress AR, Ehrman R, Robbins SJ (1998). Conditioning factors in drug abuse: can they explain compulsion? J Psychopharmacol 12: 15–22.
Perkins KA, Broge M, Gerlach D, Sanders M, Grobe JE, Cherry C et al (2002). Acute nicotine reinforcement, but not chronic tolerance, predicts withdrawal and relapse after quitting smoking. Health Psychol 21: 332–339.
Rabiner EA, Slifstein M, Nobrega J, Plisson C, Huiban M, Raymond R et al (2009). In vivo quantification of regional dopamine-D3 receptor binding potential of (+)-PHNO: Studies in non-human primates and transgenic mice. Synapse 63: 782–793.
Read KD, Braggio S (2010). Assessing brain free fraction in early drug discovery. Expert Opin Drug Metab Toxicol 6: 337–344.
Schotte A, Janssen PF, Gommeren W, Luyten WH, Van GP, Lesage AS et al (1996). Risperidone compared with new and reference antipsychotic drugs: in vitro and in vivo receptor binding. Psychopharmacology (Berl) 124: 57–73.
Searle G, Beaver JD, Comley RA, Bani M, Tziortzi A, Slifstein M et al (2010). Imaging dopamine D(3) receptors in the human brain with positron emission tomography, [(11)C]PHNO, and a selective D(3) receptor antagonist. Biol Psychiatry 68: 392–399.
Staley JK, Mash DC (1996). Adaptive increase in D3 dopamine receptors in the brain reward circuits of human cocaine fatalities. J Neurosci 16: 6100–6106.
Stanwood GD, Artymyshyn RP, Kung MP, Kung HF, Lucki I, McGonigle P (2000). Quantitative autoradiographic mapping of rat brain dopamine D3 binding with [(125)I]7-OH-PIPAT: evidence for the presence of D3 receptors on dopaminergic and nondopaminergic cell bodies and terminals. J Pharmacol Exp Ther 295: 1223–1231.
Surmeier DJ, Ding J, Day. M, Wang Z, Shen W (2007). D1 and D2 dopamine-receptor modulation of striatal glutamatergic signaling in striatal medium spiny neurons. Trends Neurosci 30: 228–235.
Volkow ND, Fowler JS, Wang GJ, Baler R, Telang F (2009). Imaging dopamine’s role in drug abuse and addiction. Neuropharmacology 56 (Suppl 1): 3–8.
Waters AJ, Shiffman S, Bradley BP, Mogg K (2003). Attentional shifts to smoking cues in smokers. Addiction 98: 1409–1417.
Zack M, Belsito L, Scher R, Eissenberg T, Corrigall WA (2001). Effects of abstinence and smoking on information processing in adolescent smokers. Psychopharmacology (Berl) 153: 249–257.
Acknowledgements
We thank Professor Caryn Lerman, Dr Christopher Jepson, and Dr Andrew Strasser from the Transdisciplinary Tobacco Use Research Centre (TTURC; now Center for Interdisciplinary Research on Nicotine Addiction, CIRNA), University of Pennsylvania (Philadelphia, PA, USA) for the execution of the efficacy study in human, Dr Charles R Ashby Jr, Department of Pharmaceutical Sciences, Saint John’s University (Jamaica, NY, USA) for performing the CPP experiments in rat, and Prof. Anissa Abi-Dargham and Dr Mark Slifstein, Columbia University PET Centre, for the expert execution of the baboon occupancy studies. We also thank Dr Simone Braggio, Drug Metabolism and Pharmacokinetics (DMPK) Department, GSK Medicines Research Centre (now Aptuit Srl, Verona, Italy) for interpretation of pharmacokinetic data and Antonio Felici, Project Management, GSK Medicines Research Centre (now Aptuit Srl, Verona, Italy) for his constant commitment to the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
This study was sponsored by GlaxoSmithKline. All authors were full-time employees of GlaxoSmithKline at the time the study was conducted. EMP declares that during the past 3 years he was full-time employee of GlaxoSmithKline in the period 2010–2011 and since 2012 he has been a full-time employee of F Hoffmann-La Roche, Basel. JB has been full-time employee of Maccine Pte Ltd (Singapore) in the period November 2010–March 2012 and is full-time employee of Abbott Laboratories (Greater Chicago Area, USA) since April 2012. The other authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Mugnaini, M., Iavarone, L., Cavallini, P. et al. Occupancy of Brain Dopamine D3 Receptors and Drug Craving: A Translational Approach. Neuropsychopharmacol 38, 302–312 (2013). https://doi.org/10.1038/npp.2012.171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2012.171
Keywords
This article is cited by
-
Exploring regulation and function of dopamine D3 receptors in alcohol use disorder. A PET [11C]-(+)-PHNO study
Neuropsychopharmacology (2021)
-
Exploring the role of the Ser9Gly (rs6280) Dopamine D3 receptor polymorphism in nicotine reinforcement and cue-elicited craving
Scientific Reports (2020)
-
Occupancy of dopamine D2 and D3 receptors by a novel D3 partial agonist BP1.4979: a [11C]-(+)-PHNO PET study in humans
Neuropsychopharmacology (2019)
-
Validation of gallbladder absorbed radiation dose reduction simulation: human dosimetry of [18F]fluortriopride
EJNMMI Physics (2018)
-
Acute D3 Antagonist GSK598809 Selectively Enhances Neural Response During Monetary Reward Anticipation in Drug and Alcohol Dependence
Neuropsychopharmacology (2017)