Abstract
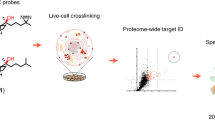
Cells produce electrophilic products with the potential to modify and affect the function of proteins. Chemoproteomic methods have provided a means to qualitatively inventory proteins targeted by endogenous electrophiles; however, ascertaining the potency and specificity of these reactions to identify the sites in the proteome that are most sensitive to electrophilic modification requires more quantitative methods. Here we describe a competitive activity–based profiling method for quantifying the reactivity of electrophilic compounds against >1,000 cysteines in parallel in the human proteome. Using this approach, we identified a select set of proteins that constitute 'hot spots' for modification by various lipid-derived electrophiles, including the oxidative stress product 4-hydroxy-2-nonenal (HNE). We show that one of these proteins, ZAK kinase, is labeled by HNE on a conserved, active site–proximal cysteine and that the resulting enzyme inhibition creates a negative feedback mechanism that can suppress the activation of JNK pathways normally induced by oxidative stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Walsh, C.T. Posttranslational Modification of Proteins: Expanding Nature's Inventory (Roberts & Company, 2005).
Jacobs, A.T. & Marnett, L.J. Systems analysis of protein modification and cellular responses induced by electrophile stress. Acc. Chem. Res. 43, 673–683 (2010).
Leonard, S.E. & Carroll, K.S. Chemical 'omics' approaches for understanding protein cysteine oxidation in biology. Curr. Opin. Chem. Biol. 15, 88–102 (2011).
Guéraud, F. et al. Chemistry and biochemistry of lipid peroxidation products. Free Radic. Res. 44, 1098–1124 (2010).
Fritz, K.S. & Petersen, D.R. An overview of the chemistry and biology of reactive aldehydes. Free Radic. Biol. Med. 59, 85–91 (2013).
Rudolph, T.K. & Freeman, B.A. Transduction of redox signaling by electrophile-protein reactions. Sci. Signal. 2, re7 (2009).
Fritz, K.S. & Petersen, D.R. Exploring the biology of lipid peroxidation-derived protein carbonylation. Chem. Res. Toxicol. 24, 1411–1419 (2011).
Leonarduzzi, G., Robbesyn, F. & Poli, G. Signaling kinases modulated by 4-hydroxynonenal. Free Radic. Biol. Med. 37, 1694–1702 (2004).
Jacobs, A.T. & Marnett, L.J. Heat shock factor 1 attenuates 4-Hydroxynonenal-mediated apoptosis: critical role for heat shock protein 70 induction and stabilization of Bcl-XL. J. Biol. Chem. 282, 33412–33420 (2007).
Surh, Y.J. et al. 15-Deoxy-Δ12,14-prostaglandin J2, an electrophilic lipid mediator of anti-inflammatory and pro-resolving signaling. Biochem. Pharmacol. 82, 1335–1351 (2011).
Chipuk, J.E. et al. Sphingolipid metabolism cooperates with BAK and BAX to promote the mitochondrial pathway of apoptosis. Cell 148, 988–1000 (2012).
Vila, A. et al. Identification of protein targets of 4-hydroxynonenal using click chemistry for ex vivo biotinylation of azido and alkynyl derivatives. Chem. Res. Toxicol. 21, 432–444 (2008).
Codreanu, S.G., Zhang, B., Sobecki, S.M., Billheimer, D.D. & Liebler, D.C. Global analysis of protein damage by the lipid electrophile 4-hydroxy-2-nonenal. Mol. Cell. Proteomics 8, 670–680 (2009).
Han, B., Hare, M., Wickramasekara, S., Fang, Y. & Maier, C.S. A comparative 'bottom up' proteomics strategy for the site-specific identification and quantification of protein modifications by electrophilic lipids. J. Proteomics 75, 5724–5733 (2012).
Roe, M.R., Xie, H., Bandhakavi, S. & Griffin, T.J. Proteomic mapping of 4-hydroxynonenal protein modification sites by solid-phase hydrazide chemistry and mass spectrometry. Anal. Chem. 79, 3747–3756 (2007).
Kim, H.Y., Tallman, K.A., Liebler, D.C. & Porter, N.A. An azido-biotin reagent for use in the isolation of protein adducts of lipid-derived electrophiles by streptavidin catch and photorelease. Mol. Cell Proteomics 8, 2080–2089 (2009).
Aldini, G. et al. Identification of actin as a 15-deoxy-Δ12,14-prostaglandin J2 target in neuroblastoma cells: mass spectrometric, computational, and functional approaches to investigate the effect on cytoskeletal derangement. Biochemistry 46, 2707–2718 (2007).
Shearn, C.T., Fritz, K.S., Reigan, P. & Petersen, D.R. Modification of Akt2 by 4-hydroxynonenal inhibits insulin-dependent Akt signaling in HepG2 cells. Biochemistry 50, 3984–3996 (2011).
Bennaars-Eiden, A. et al. Covalent modification of epithelial fatty acid-binding protein by 4-hydroxynonenal in vitro and in vivo. J. Biol. Chem. 277, 50693–50702 (2002).
Higdon, A.N. et al. Methods for imaging and detecting modification of proteins by reactive lipid species. Free Radic. Biol. Med. 47, 201–212 (2009).
LoPachin, R.M., Gavin, T., Petersen, D.R. & Barber, D.S. Molecular mechanisms of 4-hydroxy-2-nonenal and acrolein toxicity: nucleophilic targets and adduct formation. Chem. Res. Toxicol. 22, 1499–1508 (2009).
Doorn, J.A. & Petersen, D.R. Covalent modification of amino acid nucleophiles by the lipid peroxidation products 4-hydroxy-2-nonenal and 4-oxo-2-nonenal. Chem. Res. Toxicol. 15, 1445–1450 (2002).
Weerapana, E. et al. Quantitative reactivity profiling predicts functional cysteines in proteomes. Nature 468, 790–795 (2010).
Rostovtsev, V.V., Green, J.G., Fokin, V.V. & Sharpless, K.B. A stepwise Huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Edn Engl. 41, 2596–2599 (2002).
Yang, J.J. et al. ZAK inhibits human lung cancer cell growth via ERK and JNK activation in an AP-1-dependent manner. Cancer Sci. 101, 1374–1381 (2010).
Bloem, L.J. et al. Tissue distribution and functional expression of a cDNA encoding a novel mixed lineage kinase. J. Mol. Cell Cardiol. 33, 1739–1750 (2001).
Wong, J. et al. Small molecule kinase inhibitors block the ZAK-dependent inflammatory effects of doxorubicin. Cancer Biol. Ther. 14, 56–63 (2013).
Wang, X. et al. Complete inhibition of anisomycin and UV radiation but not cytokine induced JNK and p38 activation by an aryl-substituted dihydropyrrolopyrazole quinoline and mixed lineage kinase 7 small interfering RNA. J. Biol. Chem. 280, 19298–19305 (2005).
Yu, X. & Bloem, L.J. Effect of C-terminal truncations on MLK7 catalytic activity and JNK activation. Biochem. Biophys. Res. Commun. 310, 452–457 (2003).
Bachovchin, D.A. et al. Academic cross-fertilization by public screening yields a remarkable class of protein phosphatase methylesterase-1 inhibitors. Proc. Natl. Acad. Sci. USA 108, 6811–6816 (2011).
Patricelli, M.P. et al. Functional interrogation of the kinome using nucleotide acyl phosphates. Biochemistry 46, 350–358 (2007).
Shen, H.M. & Liu, Z.G. JNK signaling pathway is a key modulator in cell death mediated by reactive oxygen and nitrogen species. Free Radic. Biol. Med. 40, 928–939 (2006).
Uchida, K. 4-Hydroxy-2-nonenal: a product and mediator of oxidative stress. Prog. Lipid Res. 42, 318–343 (2003).
Dubinina, E.E. & Dadali, V.A. Role of 4-hydroxy-trans-2-nonenal in cell functions. Biochemistry (Mosc.) 75, 1069–1087 (2010).
Liu, Q. et al. Developing irreversible inhibitors of the protein kinase cysteinome. Chem. Biol. 20, 146–159 (2013).
Huang, X. et al. Crystal structure of an inactive Akt2 kinase domain. Structure 11, 21–30 (2003).
Wang, T., Kartika, R. & Spiegel, D.A. Exploring post-translational arginine modification using chemically synthesized methylglyoxal hydroimidazolones. J. Am. Chem. Soc. 134, 8958–8967 (2012).
Weerapana, E., Simon, G.M. & Cravatt, B.F. Disparate proteome reactivity profiles of carbon electrophiles. Nat. Chem. Biol. 4, 405–407 (2008).
Ban, H., Gavrilyuk, J. & Barbas, C.F. III. Tyrosine bioconjugation through aqueous ene-type reactions: a click-like reaction for tyrosine. J. Am. Chem. Soc. 132, 1523–1525 (2010).
Eng, J.K., McCormack, A.L. & Yates, J.R. III. An approach to correlate tandem mass spectral data of peptides with amino acid sequences in a protein database. J. Am. Soc. Mass Spectrom. 5, 976–989 (1994).
Cociorva, D., Tabb, D.L. & Yates, J.R. Validation of tandem mass spectrometry database search results using DTASelect. Curr. Protoc. Bioinformatics 16, 13.4 (2007).
Patricelli, M.P. et al. In situ kinase profiling reveals functionally relevant properties of native kinases. Chem. Biol. 18, 699–710 (2011).
Adibekian, A. et al. Click-generated triazole ureas as ultrapotent in vivo-active serine hydrolase inhibitors. Nat. Chem. Biol. 7, 469–478 (2011).
Schneider, C.A., Rasband, W.S. & Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Acknowledgements
We thank K. Backus, D. Bachovchin, B. Lanning, K. Tsuboi and A. Adibekian from the Cravatt Lab for providing reagents, and the Marletta lab at The Scripps Research Institute for sharing instrumentation for data collection. This work was supported by the US National Institutes of Health (NIH) (CA087660), an NIH/NIEHS K99/R00 Pathways to Independence Postdoctoral Award (K99ES020851, C.W.), a Pfizer Postdoctoral Fellowship (E.W.), a US National Science Foundation predoctoral fellowship (M.B.) and the Skaggs Institute for Chemical Biology.
Author information
Authors and Affiliations
Contributions
B.F.C., C.W. and E.W. conceived of the project. C.W., E.W. and M.M.B. performed experiments. B.F.C. and C.W. analyzed data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 771 kb)
Supplementary Table 1
In vitro profiling with each of the three LDEs at 100 μM in MDA-MB-231 and Ramos proteomes (XLS 6769 kb)
Supplementary Table 2
List of cysteines competed by one or more LDE with competition ratios > 5 (XLSX 51 kb)
Supplementary Table 3
In vitro profiling with HNE at 5 different concentrations in MDA-MB-231 proteomes (XLS 711 kb)
Supplementary Table 4
In situ profiling with HNE at 50 and 100 μM in MDA-MB-231 cells (XLS 422 kb)
Supplementary Table 5
List of kinases with reactive cysteines quantified by in vitro profiling with 100 μM HNE in MDA-MB-231 proteomes (XLSX 53 kb)
Supplementary Table 6
SILAC-ABPP ratios for acylphosphate-ATP probe-labeled proteins in proteomes from HEK-293T cells transfected with ZAK and treated with DMSO or 100 μM HNE in vitro (XLSX 1817 kb)
Rights and permissions
About this article
Cite this article
Wang, C., Weerapana, E., Blewett, M. et al. A chemoproteomic platform to quantitatively map targets of lipid-derived electrophiles. Nat Methods 11, 79–85 (2014). https://doi.org/10.1038/nmeth.2759
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.2759
This article is cited by
-
Z-REX: shepherding reactive electrophiles to specific proteins expressed tissue specifically or ubiquitously, and recording the resultant functional electrophile-induced redox responses in larval fish
Nature Protocols (2023)
-
N-Acryloylindole-alkyne (NAIA) enables imaging and profiling new ligandable cysteines and oxidized thiols by chemoproteomics
Nature Communications (2023)
-
A proteome-wide atlas of lysine-reactive chemistry
Nature Chemistry (2021)
-
A quantitative thiol reactivity profiling platform to analyze redox and electrophile reactive cysteine proteomes
Nature Protocols (2020)
-
Harnessing the anti-cancer natural product nimbolide for targeted protein degradation
Nature Chemical Biology (2019)