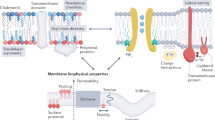
Abstract
Hydrated networks of glycans (polysaccharides)—in the form of cell walls, periplasms or gel-like matrices—are ubiquitously present adjacent to cellular plasma membranes1,2,3,4. Yet, despite their abundance, the function of glycans in the extracellular milieu is largely unknown5. Here we show that the spatial configuration of glycans controls the phase behaviour of multiphase model lipid membranes: inhomogeneous glycan networks stabilize large lipid domains at the characteristic length scale of the network, whereas homogeneous networks suppress macroscopic lipid phase separation. We also find that glycan-patterned phase separation is thermally reversible—thus indicating that the effect is thermodynamic rather than kinetic—and that phase patterning probably results from a preferential interaction of glycans with ordered lipid phases. These findings have implications for membrane-mediated transport processes6,7,8, potentially rationalize long-standing observations that differentiate the behaviour of native and model membranes9,10,11,12,13 and may indicate an intimate coupling between cellular lipidomes and glycomes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Alberts, B. et al. Molecular Biology of the Cell 4th edn (Garland Science, 2002).
Laurent, T. & Fraser, J. Hyaluronan. The FASEB J. 6, 2397–2404 (1992).
Masako, O. The ultrastructure of yeast: Cell wall structure and formation. Micron 29, 207–233 (1998).
Somerville, C. et al. Toward a systems approach to understanding plant-cell walls. Science 306, 2206–2211 (2004).
Marth, J. D. A unified vision of the building blocks of life. Nature Cell Biol. 10, 1015–1016 (2008).
Lingwood, D. & Simons, K. Lipid rafts as a membrane-organizing principle. Science 327, 46–50 (2010).
Brown, D. A. & London, E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 14, 111–136 (1998).
Engelman, D. M. Membranes are more mosaic than fluid. Nature 438, 578–580 (2005).
Munro, S. Lipid rafts: Elusive or illusive? Cell 115, 377–388 (2003).
Yethiraj, A. & Weisshaar, J. C. Why are lipid rafts not observed in vivo? Biophys. J. 93, 3113–3119 (2007).
Veatch, S. L. & Keller, S. L. Organization in lipid membranes containing cholesterol. Phys. Rev. Lett. 89, 268101 (2002).
Baumgart, T., Hess, S. T. & Webb, W. W. Imaging coexisting fluid domains in biomembrane models coupling curvature and line tension. Nature 425, 821–824 (2003).
McConnell, H. M. & Vrljic, M. Liquid–liquid immiscibility in membranes. Annu. Rev. Biophys. Biomol. Struct. 32, 469–492 (2003).
Stottrup, B. L., Veatch, S. L. & Keller, S. L. Nonequilibrium behavior in supported lipid membranes containing cholesterol. Biophys. J. 86, 2942–2950 (2004).
Parthasarathy, R., Yu, C. H. & Groves, J. T. Curvature-modulated phase separation in lipid bilayer membranes. Langmuir 22, 5095–5099 (2006).
Groves, J. T., Ulman, N. & Boxer, S. G. Micropatterning fluid lipid bilayers on solid supports. Science 275, 651–653 (1997).
Tanaka, M. & Sackmann, E. Polymer-supported membranes as models of the cell surface. Nature 437, 656–663 (2005).
Smith, H. L. et al. Model lipid membranes on a tunable polymer cushion. Phys. Rev. Lett. 102, 228102 (2009).
Wong, J. Y. et al. Polymer-cushioned bilayers. I. A structural study of various preparation methods using neutron reflectometry. Biophys. J. 77, 1445–1457 (1999).
Sackmann, E. & Tanaka, M. Supported membranes on soft polymer cushions: Fabrication, characterization and applications. Trends Biotechnol. 18, 58–64 (2000).
Watkins, E. B. et al. Structure and thermodynamics of lipid bilayers on polyethylene glycol cushions: Fact and fiction of PEG cushioned membranes. Langmuir 27, 13618–13628 (2011).
Wagner, M. L. & Tamm, L. K. Tethered polymer-supported planar lipid bilayers for reconstitution of integral membrane proteins: Silane-polyethyleneglycol-lipid as a cushion and covalent linker. Biophys. J. 79, 1400–1414 (2000).
Kraft, M. L., Weber, P. K., Longo, M. L., Hutcheon, I. D. & Boxer, S. G. Phase separation of lipid membranes analyzed with high-resolution secondary ion mass spectrometry. Science 313, 1948–1951 (2006).
Yoon, T-Y. et al. Topographic control of lipid-raft reconstitution in model membranes. Nature Mater. 5, 281–285 (2006).
Kahya, N., Scherfeld, D., Bacia, K., Poolman, B. & Schwille, P. Probing lipid mobility of raft-exhibiting model membranes by fluorescence correlation spectroscopy. J. Biol. Chem. 278, 28109–28115 (2003).
Kaizuka, Y. & Groves, J. T. Structure and dynamics of supported intermembrane junctions. Biophys. J. 86, 905–912 (2004).
Subramaniam, A. B., Lecuyer, S., Ramamurthi, K. S., Losick, R. & Stone, H. A. Particle/fluid interface replication as a means of producing topographically patterned polydimethylsiloxane surfaces for deposition of lipid bilayers. Adv. Mater. 22, 2142–2147 (2010).
Dietrich, C. et al. Lipid rafts reconstituted in model membranes. Biophys. J. 80, 1417–1428 (2001).
Pike, L. J. Rafts defined: A report on the Keystone symposium on lipid rafts and cell function. J. Lipid Res. 47, 1597–1598 (2006).
Young, M. E. et al. The Sur7p family defines novel cortical domains in Saccharomyces cerevisiae, affects sphingolipid metabolism, and is involved in sporulation. Mol. Cell. Biol. 22, 927–934 (2002).
Malinsky, J., Opekarová, M. & Tanner, W. The lateral compartmentation of the yeast plasma membrane. Yeast 27, 473–478 (2010).
Smith, A. M., Vinchurkar, M., Gronbech-Jensen, N. & Parikh, A. N. Order at the edge of the bilayer: Membrane remodeling at the edge of a planar supported bilayer is accompanied by a localized phase change. J. Am. Chem. Soc. 132, 9320–9327 (2010).
Acknowledgements
We thank the Harvard MRSEC (DMR-0820484), Harvard Center for Brain Science Imaging Facility, the Harvard Center for Nanoscale Systems, and Princeton University for partial support of this research. We also thank S. Lecuyer and M. Staykova for comments on the manuscript.
Author information
Authors and Affiliations
Contributions
A.B.S. made the initial observation of the effects of glycans on supported lipid membranes. H.A.S. and A.B.S. initiated research. A.B.S. designed and performed experiments and analysed data. A.B.S., G.G., V.N.M. and H.A.S. interpreted data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2252 kb)
Rights and permissions
About this article
Cite this article
Subramaniam, A., Guidotti, G., Manoharan, V. et al. Glycans pattern the phase behaviour of lipid membranes. Nature Mater 12, 128–133 (2013). https://doi.org/10.1038/nmat3492
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3492