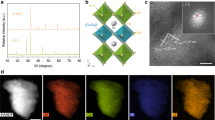
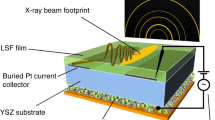
Abstract
Fast, reversible redox reactions in solids at low temperatures without thermomechanical degradation are a promising strategy for enhancing the overall performance and lifetime of many energy materials and devices. However, the robust nature of the cation’s oxidation state and the high thermodynamic barrier have hindered the realization of fast catalysis and bulk diffusion at low temperatures. Here, we report a significant lowering of the redox temperature by epitaxial stabilization of strontium cobaltites (SrCoOx) grown directly as one of two distinct crystalline phases, either the perovskite SrCoO3−δ or the brownmillerite SrCoO2.5. Importantly, these two phases can be reversibly switched at a remarkably reduced temperature (200–300 °C) in a considerably short time (< 1 min) without destroying the parent framework. The fast, low-temperature redox activity in SrCoO3−δ is attributed to a small Gibbs free-energy difference between two topotatic phases. Our findings thus provide useful information for developing highly sensitive electrochemical sensors and low-temperature cathode materials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Peña, M. A. & Fierro, J. L. G. Chemical structures and performance of perovskite oxides. Chem. Rev. 101, 1981–2017 (2001).
Maier, J. Nanoionics: Ion transport and electrochemical storage in confined systems. Nature Mater. 4, 805–815 (2005).
Nørskov, J. K., Bligaard, T., Rossmeisl, J. & Christensen, C. H. Towards the computational design of solid catalysts. Nature Chem. 1, 37–46 (2009).
Ishihara, T. Perovskite Oxide for Solid Oxide Fuel Cells (Springer, 2009).
Shao, Z. & Haile, S. M. A high-performance cathode for the next generation of solid-oxide fuel cells. Nature 431, 170–173 (2004).
Poeppelmeier, K. R., Leonowicz, M. E. & Longo, J. M. CaMnO2.5 and Ca2MnO3.5: New oxygen-defect perovskite-type oxides. J. Solid State Chem. 44, 89–98 (1982).
Hayward, M. A. et al. The hydride anion in an extended transition metal oxide array: LaSrCoO3H0.7 . Science 295, 1882–1884 (2002).
Inoue, S. et al. Anisotropic oxygen diffusion at low temperature in perovskite-structure iron oxides. Nature Chem. 2, 213–217 (2010).
Long, Y., Kaneko, Y., Ishiwata, S., Taguchi, Y. & Tokura, Y. Synthesis of cubic SrCoO3 single crystal and its anisotropic magnetic and transport properties. J. Phys. Condens. Matter 23, 245601–245606 (2011).
Takeda, T., Watanabe, H. & Yamaguchi, Y. Magnetic structure of SrCoO2.5 . J. Phys. Soc. Jpn 33, 970–972 (1972).
Bezdicka, P., Wattiaux, A., Grenier, J. C., Pouchard, M. & Hagenmuller, P. Preparation and characterization of fully stoichiometric SrCoO3 by electrochemical oxidation. Z. Anorg. Allg. Chem. 619, 7–12 (1993).
Le Toquin, R., Paulus, W., Cousson, A., Prestipino, C. & Lamberti, C. Time-resolved in situ studies of oxygen intercalation into SrCoO2.5, performed by neutron diffraction and X-ray absorption spectroscopy. J. Am. Chem. Soc. 128, 13161–13174 (2006).
Nemudry, A., Rudolf, P. & Schöllhorn, R. Topotactic electrochemical redox reactions of the defect perovskite SrCoO2.5+x . Chem. Mater. 8, 2232–2238 (1996).
Taguchi, H., Shimada, M. & Koizumi, M. The effect of oxygen vacancy on the magnetic properties in the system SrCoO3−δ (0 < δ < 0.5). J. Solid State Chem. 29, 221–225 (1979).
Pasierb, P., Komornicki, S. & Rekas, M. Comparison of the chemical diffusion of undoped and Nb-doped SrTiO3 . J. Phys. Chem. Solids 60, 1835–1844 (1999).
Goodenough, J. B. & Longo, J. M. Landolt Börnstein Vol. III/4a (Springer, 1970).
Mizusaki, J., Yamauchi, S., Fueki, K. & Ishikawa, A. Nonstoichiometry of the perovskite-type oxide La1−xSrxCrO3−δ . Solid State Ion. 12, 119–124 (1984).
Hayashi, N., Terashima, T. & Takano, M. Oxygen-holes creating different electronic phases in Fe4+-oxides: Successful growth of single crystalline films of SrFeO3 and related perovskites at low oxygen pressure. J. Mater. Chem. 11, 2235–2237 (2001).
Muñoz, A. et al. Crystallographic and magnetic structure of SrCoO2.5 brownmillerite: Neutron study coupled with band-structure calculations. Phys. Rev. B 78, 054404–054404 (2008).
Stemmer, S., Sane, A., Browning, N. D. & Mazanec, T. J. Characterization of oxygen-deficient SrCoO3−δ by electron energy-loss spectroscopy and Z-contrast imaging. Solid State Ion. 130, 71–80 (2000).
Sammells, A. F., Cook, R. L., White, J. H., Osborne, J. J. & Macduff, R. C. Rational selection of advanced solid electrolytes for intermediate temperature fuel cells. Solid State Ion. 52, 111–123 (1992).
Señarı´s-Rodrı´guez, M. A. & Goodenough, J. B. Magnetic and transport properties of the system La1−xSrxCoO3−δ (0 < x≤0.50). J. Solid State Chem. 118, 323–336 (1995).
Wu, J. & Leighton, C. Glassy ferromagnetism and magnetic phase separation in La1−xSrxCoO3 . Phys. Rev. B 67, 174408 (2003).
Suntivich, J. et al. Design principles for oxygen-reduction activity on perovskite oxide catalysts for fuel cells and metal-air batteries. Nature Chem. 3, 546–550 (2011).
Imada, M., Fujimori, A. & Tokura, Y. Metal–insulator transitions. Rev. Mod. Phys. 70, 1039–1263 (1998).
Moodenbaugh, A. R. et al. Hole-state density of La1−xSrxCoO3−δ (0≤x≤0.5) across the insulator/metal phase boundary. Phys. Rev. B 61, 5666–5671 (2000).
Karvonen, L. et al. O-K and Co-L XANES study on oxygen intercalation in perovskite SrCoO3−δ . Chem. Mater. 22, 70–76 (2010).
Xie, C. K. et al. Magnetic phase separation in SrCoOx (2.5≤x≤3). Appl. Phys. Lett. 99, 052503 (2011).
Balamurugan, S. et al. Specific-heat evidence of strong electron correlations and thermoelectric properties of the ferromagnetic perovskite SrCoO3−δ . Phys. Rev. B 74, 172406 (2006).
Ichikawa, N. et al. Reduction and oxidation of SrCoO2.5 thin films at low temperatures. Dalton Trans. 41, 10507–10510 (2012).
Lee, J. H. & Rabe, K. M. Coupled magnetic-ferroelectric metal-insulator transition in epitaxially strained SrCoO3 from first principles. Phys. Rev. Lett. 107, 067601–067601 (2011).
Potze, R. H., Sawatzky, G. A. & Abbate, M. Possibility for an intermediate spin ground state in the charge transfer material SrCoO3 . Phys. Rev. B 51, 11501–11506 (1995).
Maignan, A., Pelloquin, D., Martin, C., Hervieu, M. & Raveau, B. A new form of oxygen deficient 1201-cobaltite (Tl0.4Sr0.5Co0.1) Sr2CoO5−δ: Structure, transport and magnetic properties. J. Mater. Chem. 12, 1009–1016 (2002).
Zeng, P. Y. et al. Efficient stabilization of cubic perovskite SrCoO3−δ by B-site low concentration scandium doping combined with sol-gel synthesis. J. Alloys Compd 455, 465–470 (2008).
Tsujimoto, Y. et al. Infinite-layer iron oxide with a square-planar coordination. Nature 450, 1062–1065 (2007).
Saal, J. E. Thermodynamic Modeling of Phase Transformations: Cobalt Oxides PhD thesis, The Pennsylvania State Univ. (2010).
Acknowledgements
The work was supported by the US Department of Energy, Basic Energy Sciences, Materials Sciences and Engineering Division. The in situ XRD measurement was conducted at the Center for Nanophase Materials Sciences, which is sponsored at Oak Ridge National Laboratory by the Scientific User Facilities Division, Office of Basic Energy Sciences, US Department of Energy. Use of the Advanced Photon Source was supported by the US Department of Energy, Office of Science, under Contract No. DE-AC02-06CH11357. H.O. was supported by MEXT (25246023).
Author information
Authors and Affiliations
Contributions
H.J. conducted sample synthesis, XRD, d.c. transport and SQUID measurements with help from W.S.C., and H.J. and M.D.B. performed the high-temperature environmental XRD, under the direction of H.N.L. M.F.C. performed STEM measurements. I-C.T. and J.W.F. measured XAS and XMCD, and H.O. worked on thermopower measurements. D.S. performed the thermodynamic modelling. C.M.F. and D.D.F. worked on the catalysis measurement. H.N.L. initiated the research and supervised the work. All authors participated in writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 753 kb)
Rights and permissions
About this article
Cite this article
Jeen, H., Choi, W., Biegalski, M. et al. Reversible redox reactions in an epitaxially stabilized SrCoOx oxygen sponge. Nature Mater 12, 1057–1063 (2013). https://doi.org/10.1038/nmat3736
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3736
This article is cited by
-
Wide-range continuous tuning of the thermal conductivity of La0.5Sr0.5CoO3-δ films via room-temperature ion-gel gating
Nature Communications (2023)
-
Defect graph neural networks for materials discovery in high-temperature clean-energy applications
Nature Computational Science (2023)
-
Robotic pendant drop: containerless liquid for μs-resolved, AI-executable XPCS
Light: Science & Applications (2023)
-
Topotactically transformable antiphase boundaries with enhanced ionic conductivity
Nature Communications (2023)
-
Artificially controlled nanoscale chemical reduction in VO2 through electron beam illumination
Nature Communications (2023)