Abstract
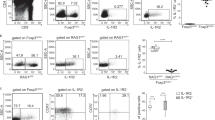
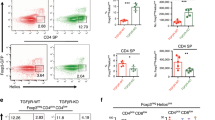
Interleukin 17 (IL-17)-producing CD4+ helper T cells (TH-17 cells) share a developmental relationship with Foxp3+ regulatory T cells (Treg cells). Here we show that a TH-17 population differentiates in the thymus in a manner influenced by recognition of self antigen and by the cytokines IL-6 and transforming growth factor-β (TGF-β). Like previously described TH-17 cells, the TH-17 cells that developed in the thymus expressed the transcription factor RORγt and the IL-23 receptor. These cells also expressed α4β1 integrins and the chemokine receptor CCR6 and were recruited to the lung, gut and liver. In the liver, these cells secreted IL-22 in response to self antigen and mediated host protection during inflammation. Thus, TH-17 cells, like Treg cells, can be selected by self antigens in the thymus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
05 October 2009
NOTE: In the version of this article initially published, two relevant papers are not cited. The following brief description of the findings of these papers has been added to the end of the fourth full paragraph on page 1131, and the citations below are now included at the end of the reference list: “Our observations follow the identification in human thymi and umbilical cord blood of CD3+CD4+CD161+ cells that express RORγt, IL-23R and CCR6 and produce IL-17 after activation and stimulation with IL-1 and IL-23 (ref. 54). This work suggested that thymus-derived cells could be precursors of peripheral TH-17 cells in humans, an idea supported by the finding that CD161+IL-17+ cells can be isolated from the intestinal tissue of patients with Crohn's disease55.” 54. Cosmi, L. et al. Human interleukin 17-producing cells originate from a CD161+CD4+ T cell precursor. J. Exp. Med. 205, 1903–1916 (2008). 55. Kleinschek. M.A. et al. Circulating and gut-resident human Th17 cells express CD161 and promote intestinal inflammation. J. Exp. Med. 206, 525–534 (2009). The error has been corrected in the HTML and PDF versions of the article.
References
Murphy, K.M. & Reiner, S.L. The lineage decisions of helper T cells. Nat. Rev. Immunol. 2, 933–944 (2002).
Abbas, A.K., Murphy, K.M. & Sher, A. Functional diversity of helper T lymphocytes. Nature 383, 787–793 (1996).
Harrington, L.E., Mangan, P.R. & Weaver, C.T. Expanding the effector CD4 T-cell repertoire: the Th17 lineage. Curr. Opin. Immunol. 18, 349–356 (2006).
Aggarwal, S., Ghilardi, N., Xie, M.H., de Sauvage, F.J. & Gurney, A.L. Interleukin-23 promotes a distinct CD4 T cell activation state characterized by the production of interleukin-17. J. Biol. Chem. 278, 1910–1914 (2003).
Ivanov, I.I. et al. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126, 1121–1133 (2006).
Yang, X.O. et al. T helper 17 lineage differentiation is programmed by orphan nuclear receptors RORα and RORγ. Immunity 28, 29–39 (2008).
Zenewicz, L.A. et al. Interleukin-22 but not interleukin-17 provides protection to hepatocytes during acute liver inflammation. Immunity 27, 647–659 (2007).
Langrish, C.L. et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005).
Bowman, E.P., Chackerian, A.A. & Cua, D.J. Rationale and safety of anti-interleukin-23 and anti-interleukin-17A therapy. Curr. Opin. Infect. Dis. 19, 245–252 (2006).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Mangan, P.R. et al. Transforming growth factor-beta induces development of the TH17 lineage. Nature 441, 231–234 (2006).
Veldhoen, M., Hocking, R.J., Atkins, C.J., Locksley, R.M. & Stockinger, B. TGFβ in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 24, 179–189 (2006).
Setoguchi, R., Hori, S., Takahashi, T. & Sakaguchi, S. Homeostatic maintenance of natural Foxp3+CD25+CD4+ regulatory T cells by interleukin (IL)-2 and induction of autoimmune disease by IL-2 neutralization. J. Exp. Med. 201, 723–735 (2005).
Laurence, A. et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity 26, 371–381 (2007).
Kryczek, I. et al. Cutting edge: opposite effects of IL-1 and IL-2 on the regulation of IL-17+ T cell pool IL-1 subverts IL-2-mediated suppression. J. Immunol. 179, 1423–1426 (2007).
Xu, L., Kitani, A., Fuss, I. & Strober, W. Cutting edge: regulatory T cells induce CD4+CD25−Foxp3− T cells or are self-induced to become Th17 cells in the absence of exogenous TGF-β. J. Immunol. 178, 6725–6729 (2007).
Mucida, D. et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science 317, 256–260 (2007).
Zhou, L. et al. TGF-β-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORγt function. Nature 453, 236–240 (2008).
Zhang, F., Meng, G. & Strober, W. Interactions among the transcription factors Runx1, RORγt and Foxp3 regulate the differentiation of interleukin 17–producing T cells. Nat. Immunol. 9, 1297–1306 (2008).
Gavin, M.A. et al. Foxp3-dependent programme of regulatory T-cell differentiation. Nature 445, 771–775 (2007).
Jordan, M.S. et al. Thymic selection of CD4+CD25+ regulatory T cells induced by an agonist self-peptide. Nat. Immunol. 2, 301–306 (2001).
Hsieh, C.S., Zheng, Y., Liang, Y., Fontenot, J.D. & Rudensky, A.Y. An intersection between the self-reactive regulatory and nonregulatory T cell receptor repertoires. Nat. Immunol. 7, 401–410 (2006).
Kronenberg, M. & Gapin, L. The unconventional lifestyle of NKT cells. Nat. Rev. Immunol. 2, 557–568 (2002).
Cheroutre, H. Starting at the beginning: new perspectives on the biology of mucosal T cells. Annu. Rev. Immunol. 22, 217–246 (2004).
Xiong, N. & Raulet, D.H. Development and selection of γδ T cells. Immunol. Rev. 215, 15–31 (2007).
Apostolou, I., Sarukhan, A., Klein, L. & von Boehmer, H. Origin of regulatory T cells with known specificity for antigen. Nat. Immunol. 3, 756–763 (2002).
Oehen, S., Feng, L., Xia, Y., Surh, C.D. & Hedrick, S.M. Antigen compartmentation and T helper cell tolerance induction. J. Exp. Med. 183, 2617–2626 (1996).
Grossman, Z. & Paul, W.E. Autoreactivity, dynamic tuning and selectivity. Curr. Opin. Immunol. 13, 687–698 (2001).
Monroe, R.J. et al. RAG2:GFP knockin mice reveal novel aspects of RAG2 expression in primary and peripheral lymphoid tissues. Immunity 11, 201–212 (1999).
van Santen, H.M., Benoist, C. & Mathis, D. Number of T reg cells that differentiate does not increase upon encounter of agonist ligand on thymic epithelial cells. J. Exp. Med. 200, 1221–1230 (2004).
Li, M.O., Wan, Y.Y. & Flavell, R.A. T cell-produced transforming growth factor-beta1 controls T cell tolerance and regulates Th1- and Th17-cell differentiation. Immunity 26, 579–591 (2007).
Veldhoen, M., Hocking, R.J., Flavell, R.A. & Stockinger, B. Signals mediated by transforming growth factor-β initiate autoimmune encephalomyelitis, but chronic inflammation is needed to sustain disease. Nat. Immunol. 7, 1151–1156 (2006).
Gorelik, L. & Flavell, R.A. Abrogation of TGFβ signaling in T cells leads to spontaneous T cell differentiation and autoimmune disease. Immunity 12, 171–181 (2000).
Fahlen, L. et al. T cells that cannot respond to TGF-β escape control by CD4+CD25+ regulatory T cells. J. Exp. Med. 201, 737–746 (2005).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Yoshinaga, S.K. et al. T-cell co-stimulation through B7RP-1 and ICOS. Nature 402, 827–832 (1999).
Nishimura, H., Nose, M., Hiai, H., Minato, N. & Honjo, T. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity 2, 141–151 (1999).
Ley, K., Laudanna, C., Cybulsky, M.I. & Nourshargh, S. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat. Rev. Immunol. 7, 678–689 (2007).
Reboldi, A. et al. C–C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat. Immunol. 10, 514–523 (2009).
Schutyser, E., Struyf, S. & Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev. 14, 409–426 (2003).
Liang, S.C. et al. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J. Exp. Med. 203, 2271–2279 (2006).
McGeachy, M.J. et al. TGF-β and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain TH-17 cell-mediated pathology. Nat. Immunol. 8, 1390–1397 (2007).
Arvelo, M.B. et al. A20 protects mice from D-galactosamine/lipopolysaccharide acute toxic lethal hepatitis. Hepatology 35, 535–543 (2002).
Michel, M.L. et al. Critical role of ROR-γt in a new thymic pathway leading to IL-17-producing invariant NKT cell differentiation. Proc. Natl. Acad. Sci. USA 105, 19845–19850 (2008).
Mattner, J. et al. Exogenous and endogenous glycolipid antigens activate NKT cells during microbial infections. Nature 434, 525–529 (2005).
Hayday, A. & Tigelaar, R. Immunoregulation in the tissues by γδ T cells. Nat. Rev. Immunol. 3, 233–242 (2003).
Lahn, M. et al. Negative regulation of airway responsiveness that is dependent on γδ T cells and independent of αβ T cells. Nat. Med. 5, 1150–1156 (1999).
Jahng, A.W. et al. Activation of natural killer T cells potentiates or prevents experimental autoimmune encephalomyelitis. J. Exp. Med. 194, 1789–1799 (2001).
Roark, C.L., Simonian, P.L., Fontenot, A.P., Born, W.K. & O'Brien, R.L. γδ T cells: an important source of IL-17. Curr. Opin. Immunol. 20, 353–357 (2008).
Chien, Y.H. & Konigshofer, Y. Antigen recognition by γδ T cells. Immunol. Rev. 215, 46–58 (2007).
Yang, X.O. et al. Regulation of inflammatory responses by IL-17F. J. Exp. Med. 205, 1063–1075 (2008).
Aggarwal, S., Xie, M.H., Maruoka, M., Foster, J. & Gurney, A.L. Acinar cells of the pancreas are a target of interleukin-22. J. Interferon Cytokine Res. 21, 1047–1053 (2001).
Ito, T. et al. Two functional subsets of FOXP3+ regulatory T cells in human thymus and periphery. Immunity 28, 870–880 (2008).
Cosmi, L. et al. Human interleukin 17-producing cells originate from a CD161+CD4+ T cell precursor. J. Exp. Med. 205, 1903–1916 (2008).
Kleinschek, M.A. et al. Circulating and gut-resident human Th17 cells express CD161 and promote intestinal inflammation. J. Exp. Med. 206, 525–534 (2009).
Acknowledgements
We thank S. Hedrick (University of California, San Diego) and S. Oehan (University Hospital, Zurich) for AND- and PCC-transgenic mice; F. Alt (The Children's Hospital and Harvard Medical School) and D. Schatz (Yale University School of Medicine) for RAG2:GFP mice; H. van Santen, D. Mathis and C. Benoist (Joslin Diabetes Center and Harvard Medical School) for TA.TIM mice; L. Bockenstedt (Yale University School of Medicine) for CD1d-deficient mice; E. Marks and other members of the Craft laboratory for discussions; L. Zenewicz and E. Espluges for advice; and R. Medzhitov and S. Kaech for critical reading of the manuscript. Supported by the US National Institutes of Heath (AR40072, AR44076, AI56219 and AR053495; and 5T32GM07205 to B.R.M.), the Arthritis Foundation, Rheuminations and the Connecticut chapter of the Lupus Foundation.
Author information
Authors and Affiliations
Contributions
B.R.M. and J.C. designed experiments; B.R.M., H.N.N., J.Y. C., A.C.P. and J.M.O. did the experiments; B.R.M., H.N.N., J.Y.C., R.A.F. and J.C. analyzed the data; and B.R.M. and J.C. wrote the paper.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 and Supplementary Tables 1–2 (PDF 1112 kb)
Rights and permissions
About this article
Cite this article
Marks, B., Nowyhed, H., Choi, JY. et al. Thymic self-reactivity selects natural interleukin 17–producing T cells that can regulate peripheral inflammation. Nat Immunol 10, 1125–1132 (2009). https://doi.org/10.1038/ni.1783
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1783
This article is cited by
-
IL-17 and IL-17-producing cells in protection versus pathology
Nature Reviews Immunology (2023)
-
The star target in SLE: IL-17
Inflammation Research (2023)
-
Mucocutaneous IL-17 immunity in mice and humans: host defense vs. excessive inflammation
Mucosal Immunology (2018)
-
The complex alteration in the network of IL-17-type cytokines in patients with hereditary angioedema
Clinical and Experimental Medicine (2018)
-
Apoptosis in response to microbial infection induces autoreactive TH17 cells
Nature Immunology (2016)