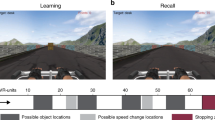
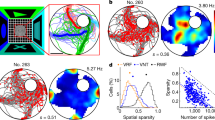
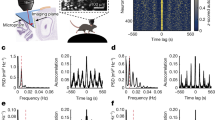
Abstract
Theories on the functions of the hippocampal system are based largely on two fundamental discoveries: the amnestic consequences of removing the hippocampus and associated structures in the famous patient H.M. and the observation that spiking activity of hippocampal neurons is associated with the spatial position of the rat. In the footsteps of these discoveries, many attempts were made to reconcile these seemingly disparate functions. Here we propose that mechanisms of memory and planning have evolved from mechanisms of navigation in the physical world and hypothesize that the neuronal algorithms underlying navigation in real and mental space are fundamentally the same. We review experimental data in support of this hypothesis and discuss how specific firing patterns and oscillatory dynamics in the entorhinal cortex and hippocampus can support both navigation and memory.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
O'Keefe, J. & Nadel, L. The Hippocampus as a Cognitive Map (Oxford Univ. Press, New York, 1978).
McNaughton, B.L. et al. Deciphering the hippocampal polyglot: the hippocampus as a path integration system. J. Exp. Biol. 199, 173–185 (1996).
O'Keefe, J. & Burgess, N. Geometric determinants of the place fields of hippocampal neurons. Nature 381, 425–428 (1996).
Thompson, E. & Varela, F.J. Radical embodiment: neural dynamics and consciousness. Trends Cogn. Sci. 5, 418–425 (2001).
McNaughton, B.L., Battaglia, F.P., Jensen, O., Moser, E.I. & Moser, M.B. Path integration and the neural basis of the 'cognitive map'. Nat. Rev. Neurosci. 7, 663–678 (2006).
Knierim, J.J., Kudrimoti, H.S. & McNaughton, B.L. Interactions between idiothetic cues and external landmarks in the control of place cells and head direction cells. J. Neurophysiol. 80, 425–446 (1998).
Derdikman, D. et al. Fragmentation of grid cell maps in a multicompartment environment. Nat. Neurosci. 12, 1325–1332 (2009).
Gothard, K.M., Skaggs, W.E., Moore, K.M. & McNaughton, B.L. Binding of hippocampal CA1 neural activity to multiple reference frames in a landmark-based navigation task. J. Neurosci. 16, 823–835 (1996).
Scoville, W.B. & Milner, B. Loss of recent memory after bilateral hippocampal lesions. J. Neurol. Neurosurg. Psychiatry 20, 11–21 (1957).
Squire, L.R. Memory and the hippocampus: a synthesis from findings with rats, monkeys, and humans. Psychol. Rev. 99, 195–231 (1992).
Suddendorf, T. & Corballis, M.C. The evolution of foresight: what is mental time travel, and is it unique to humans? Behav. Brain Sci. 30, 299–313; discussion 313–251 (2007).
Tulving, E., Donaldson, W., Bower, G.H. & United States Office of Naval Research. Organization of Memory (Academic, New York, 1972).
Buckner, R.L. The role of the hippocampus in prediction and imagination. Annu. Rev. Psychol. 61, 27–48, C21–C28 (2010).
Tulving, E. Chronesthesia: conscious awareness of subjective time. in Principles of Frontal Lobe Function (eds. Stuss, D.T. & Knight, R.C.) 311–325 (Oxford Univ. Press, New York, 2002).
Buzsáki, G. Theta rhythm of navigation: link between path integration and landmark navigation, episodic and semantic memory. Hippocampus 15, 827–840 (2005).
Eichenbaum, H., Dudchenko, P., Wood, E., Shapiro, M. & Tanila, H. The hippocampus, memory, and place cells: is it spatial memory or a memory space? Neuron 23, 209–226 (1999).
Lever, C., Wills, T., Cacucci, F., Burgess, N. & O'Keefe, J. Long-term plasticity in hippocampal place-cell representation of environmental geometry. Nature 416, 90–94 (2002).
Hasselmo, M.E. How We Remember: Brain Mechanisms of Episodic Memory (MIT Press, Cambridge, Massachusetts, USA, 2012).
McClelland, J.L., McNaughton, B.L. & O'Reilly, R.C. Why there are complementary learning systems in the hippocampus and neocortex: insights from the successes and failures of connectionist models of learning and memory. Psychol. Rev. 102, 419–457 (1995).
Nadel, L. & Moscovitch, M. Memory consolidation, retrograde amnesia and the hippocampal complex. Curr. Opin. Neurobiol. 7, 217–227 (1997).
Manns, J.R., Hopkins, R.O., Reed, J.M., Kitchener, E.G. & Squire, L.R. Recognition memory and the human hippocampus. Neuron 37, 171–180 (2003).
Moser, E.I., Kropff, E. & Moser, M.B. Place cells, grid cells, and the brain's spatial representation system. Annu. Rev. Neurosci. 31, 69–89 (2008).
Hafting, T., Fyhn, M., Molden, S., Moser, M.B. & Moser, E.I. Microstructure of a spatial map in the entorhinal cortex. Nature 436, 801–806 (2005).
Boccara, C.N. et al. Grid cells in pre- and parasubiculum. Nat. Neurosci. 13, 987–994 (2010).
Sargolini, F. et al. Conjunctive representation of position, direction, and velocity in entorhinal cortex. Science 312, 758–762 (2006).
Krupic, J., Burgess, N. & O'Keefe, J. Neural representations of location composed of spatially periodic bands. Science 337, 853–857 (2012).
Solstad, T., Boccara, C.N., Kropff, E., Moser, M.B. & Moser, E.I. Representation of geometric borders in the entorhinal cortex. Science 322, 1865–1868 (2008).
Yartsev, M.M., Witter, M.P. & Ulanovsky, N. Grid cells without theta oscillations in the entorhinal cortex of bats. Nature 479, 103–107 (2011).
Ulanovsky, N. & Moss, C.F. Hippocampal cellular and network activity in freely moving echolocating bats. Nat. Neurosci. 10, 224–233 (2007).
Ekstrom, A.D. et al. Human hippocampal theta activity during virtual navigation. Hippocampus 15, 881–889 (2005).
Muller, R.U. & Kubie, J.L. The effects of changes in the environment on the spatial firing of hippocampal complex-spike cells. J. Neurosci. 7, 1951–1968 (1987).
Stensola, H. et al. The entorhinal grid map is discretized. Nature 492, 72–78 (2012).
Fyhn, M., Hafting, T., Treves, A., Moser, M.B. & Moser, E.I. Hippocampal remapping and grid realignment in entorhinal cortex. Nature 446, 190–194 (2007).
Leutgeb, S., Leutgeb, J.K., Treves, A., Moser, M.B. & Moser, E.I. Distinct ensemble codes in hippocampal areas CA3 and CA1. Science 305, 1295–1298 (2004).
Mizuseki, K., Royer, S., Diba, K. & Buzsáki, G. Activity dynamics and behavioral correlates of CA3 and CA1 hippocampal pyramidal neurons. Hippocampus 22, 1659–1680 (2012).
Li, X.G., Somogyi, P., Ylinen, A. & Buzsáki, G. The hippocampal CA3 network: an in vivo intracellular labeling study. J. Comp. Neurol. 339, 181–208 (1994).
Treves, A. & Rolls, E.T. Computational analysis of the role of the hippocampus in memory. Hippocampus 4, 374–391 (1994).
Leutgeb, J.K., Leutgeb, S., Moser, M.B. & Moser, E.I. Pattern separation in the dentate gyrus and CA3 of the hippocampus. Science 315, 961–966 (2007).
Muller, R.U., Stead, M. & Pach, J. The hippocampus as a cognitive graph. J. Gen. Physiol. 107, 663–694 (1996).
Menzel, R., Geiger, K., Joerges, J., Muller, U. & Chittka, L. Bees travel novel homeward routes by integrating separately acquired vector memories. Anim. Behav. 55, 139–152 (1998).
Wilson, M.A. & McNaughton, B.L. Dynamics of the hippocampal ensemble code for space. Science 261, 1055–1058 (1993).
Hampson, R.E., Byrd, D.R., Konstantopoulos, J.K., Bunn, T. & Deadwyler, S.A. Hippocampal place fields: relationship between degree of field overlap and cross-correlations within ensembles of hippocampal neurons. Hippocampus 6, 281–293 (1996).
Miyashita, Y. Cognitive memory: cellular and network machineries and their top-down control. Science 306, 435–440 (2004).
Suzuki, W.A., Miller, E.K. & Desimone, R. Object and place memory in the macaque entorhinal cortex. J. Neurophysiol. 78, 1062–1081 (1997).
Wood, E.R., Dudchenko, P.A., Robitsek, R.J. & Eichenbaum, H. Hippocampal neurons encode information about different types of memory episodes occurring in the same location. Neuron 27, 623–633 (2000).
Kumaran, D. & McClelland, J.L. Generalization through the recurrent interaction of episodic memories: a model of the hippocampal system. Psychol. Rev. 119, 573–616 (2012).
Heit, G., Smith, M.E. & Halgren, E. Neural encoding of individual words and faces by the human hippocampus and amygdala. Nature 333, 773–775 (1988).
Quiroga, R.Q., Reddy, L., Kreiman, G., Koch, C. & Fried, I. Invariant visual representation by single neurons in the human brain. Nature 435, 1102–1107 (2005).
Trope, Y. & Liberman, N. Construal-level theory of psychological distance. Psychol. Rev. 117, 440–463 (2010).
Navigli, R. & Lapata, M. An experimental study of graph connectivity for unsupervised word sense disambiguation. IEEE Trans. Pattern Anal. Mach. Intell. 32, 678–692 (2010).
Nakashiba, T. et al. Young dentate granule cells mediate pattern separation, whereas old granule cells facilitate pattern completion. Cell 149, 188–201 (2012).
Buckner, R.L. & Carroll, D.C. Self-projection and the brain. Trends Cogn. Sci. 11, 49–57 (2007).
Burgess, N., Maguire, E.A. & O'Keefe, J. The human hippocampus and spatial and episodic memory. Neuron 35, 625–641 (2002).
Jensen, O. & Lisman, J.E. Hippocampal sequence-encoding driven by a cortical multi-item working memory buffer. Trends Neurosci. 28, 67–72 (2005).
Lisman, J.E. Relating hippocampal circuitry to function: recall of memory sequences by reciprocal dentate-CA3 interactions. Neuron 22, 233–242 (1999).
Howard, M.W. & Kahana, M.J. Contextual variability and serial position effects in free recall. J. Exp. Psychol. Learn. Mem. Cogn. 25, 923–941 (1999).
Shrager, Y., Kirwan, C.B. & Squire, L.R. Neural basis of the cognitive map: path integration does not require hippocampus or entorhinal cortex. Proc. Natl. Acad. Sci. USA 105, 12034–12038 (2008).
Frank, L.M., Brown, E.N. & Wilson, M. Trajectory encoding in the hippocampus and entorhinal cortex. Neuron 27, 169–178 (2000).
Pastalkova, E., Itskov, V., Amarasingham, A. & Buzsáki, G. Internally generated cell assembly sequences in the rat hippocampus. Science 321, 1322–1327 (2008).
Buzsáki, G. Neural syntax: cell assemblies, synapsembles, and readers. Neuron 68, 362–385 (2010).
Mizuseki, K., Sirota, A., Pastalkova, E. & Buzsáki, G. Theta oscillations provide temporal windows for local circuit computation in the entorhinal-hippocampal loop. Neuron 64, 267–280 (2009).
Muller, R.U., Bostock, E., Taube, J.S. & Kubie, J.L. On the directional firing properties of hippocampal place cells. J. Neurosci. 14, 7235–7251 (1994).
McNaughton, B.L., Barnes, C.A. & O'Keefe, J. The contributions of position, direction, and velocity to single unit activity in the hippocampus of freely-moving rats. Exp. Brain Res. 52, 41–49 (1983).
Royer, S., Sirota, A., Patel, J. & Buzsáki, G. Distinct representations and theta dynamics in dorsal and ventral hippocampus. J. Neurosci. 30, 1777–1787 (2010).
Itskov, V., Pastalkova, E., Mizuseki, K., Buzsáki, G. & Harris, K.D. Theta-mediated dynamics of spatial information in hippocampus. J. Neurosci. 28, 5959–5964 (2008).
Wickelgren, W.A. Webs, cell assemblies, and chunking in neural nets: introduction. Can. J. Exp. Psychol. 53, 118–131 (1999).
Buzsáki, G. & Draguhn, A. Neuronal oscillations in cortical networks. Science 304, 1926–1929 (2004).
Bragin, A. et al. Gamma (40–100 Hz) oscillation in the hippocampus of the behaving rat. J. Neurosci. 15, 47–60 (1995).
Colgin, L.L. et al. Frequency of gamma oscillations routes flow of information in the hippocampus. Nature 462, 353–357 (2009).
Tort, A.B., Komorowski, R.W., Manns, J.R., Kopell, N.J. & Eichenbaum, H. Theta-gamma coupling increases during the learning of item-context associations. Proc. Natl. Acad. Sci. USA 106, 20942–20947 (2009).
Axmacher, N. et al. Cross-frequency coupling supports multi-item working memory in the human hippocampus. Proc. Natl. Acad. Sci. USA 107, 3228–3233 (2010).
Canolty, R.T. et al. High gamma power is phase-locked to theta oscillations in human neocortex. Science 313, 1626–1628 (2006).
Griesmayr, B., Gruber, W.R., Klimesch, W. & Sauseng, P. Human frontal midline theta and its synchronization to gamma during a verbal delayed match to sample task. Neurobiol. Learn. Mem. 93, 208–215 (2010).
Harris, K.D., Csicsvari, J., Hirase, H., Dragoi, G. & Buzsáki, G. Organization of cell assemblies in the hippocampus. Nature 424, 552–556 (2003).
Dragoi, G. & Buzsáki, G. Temporal encoding of place sequences by hippocampal cell assemblies. Neuron 50, 145–157 (2006).
Koch, C., Rapp, M. & Segev, I. A brief history of time (constants). Cereb. Cortex 6, 93–101 (1996).
Whittington, M.A., Traub, R.D., Kopell, N., Ermentrout, B. & Buhl, E.H. Inhibition-based rhythms: experimental and mathematical observations on network dynamics. Int. J. Psychophysiol. 38, 315–336 (2000).
Buzsáki, G. & Wang, X.J. Mechanisms of gamma oscillations. Annu. Rev. Neurosci. 35, 203–225 (2012).
Maurer, A.P., Cowen, S.L., Burke, S.N., Barnes, C.A. & McNaughton, B.L. Phase precession in hippocampal interneurons showing strong functional coupling to individual pyramidal cells. J. Neurosci. 26, 13485–13492 (2006).
Geisler, C., Robbe, D., Zugaro, M., Sirota, A. & Buzsáki, G. Hippocampal place cell assemblies are speed-controlled oscillators. Proc. Natl. Acad. Sci. USA 104, 8149–8154 (2007).
Royer, S. et al. Control of timing, rate and bursts of hippocampal place cells by dendritic and somatic inhibition. Nat. Neurosci. 15, 769–775 (2012).
Skaggs, W.E., McNaughton, B.L., Wilson, M.A. & Barnes, C.A. Theta phase precession in hippocampal neuronal populations and the compression of temporal sequences. Hippocampus 6, 149–172 (1996).
O'Keefe, J. & Recce, M.L. Phase relationship between hippocampal place units and the EEG theta rhythm. Hippocampus 3, 317–330 (1993).
Geisler, C. et al. Temporal delays among place cells determine the frequency of population theta oscillations in the hippocampus. Proc. Natl. Acad. Sci. USA 107, 7957–7962 (2010).
Burgess, N. & O'Keefe, J. Models of place and grid cell firing and theta rhythmicity. Curr. Opin. Neurobiol. 21, 734–744 (2011).
Diba, K. & Buzsáki, G. Hippocampal network dynamics constrain the time lag between pyramidal cells across modified environments. J. Neurosci. 28, 13448–13456 (2008).
Lisman, J.E. & Idiart, M.A. Storage of 7 +/− 2 short-term memories in oscillatory subcycles. Science 267, 1512–1515 (1995).
Jezek, K., Henriksen, E.J., Treves, A., Moser, E.I. & Moser, M.B. Theta-paced flickering between place-cell maps in the hippocampus. Nature 478, 246–249 (2011).
Johnson, A. & Redish, A.D. Neural ensembles in CA3 transiently encode paths forward of the animal at a decision point. J. Neurosci. 27, 12176–12189 (2007).
Gupta, A.S., van der Meer, M.A., Touretzky, D.S. & Redish, A.D. Segmentation of spatial experience by hippocampal theta sequences. Nat. Neurosci. 15, 1032–1039 (2012).
Kjelstrup, K.B. et al. Finite scale of spatial representation in the hippocampus. Science 321, 140–143 (2008).
Dusek, J.A. & Eichenbaum, H. The hippocampus and memory for orderly stimulus relations. Proc. Natl. Acad. Sci. USA 94, 7109–7114 (1997).
MacDonald, C.J., Lepage, K.Q., Eden, U.T. & Eichenbaum, H. Hippocampal “time cells” bridge the gap in memory for discontiguous events. Neuron 71, 737–749 (2011).
Hasselmo, M.E. Arc length coding by interference of theta frequency oscillations may underlie context-dependent hippocampal unit data and episodic memory function. Learn. Mem. 14, 782–794 (2007).
Fujisawa, S., Amarasingham, A., Harrison, M.T. & Buzsáki, G. Behavior-dependent short-term assembly dynamics in the medial prefrontal cortex. Nat. Neurosci. 11, 823–833 (2008).
Harvey, C.D., Coen, P. & Tank, D.W. Choice-specific sequences in parietal cortex during a virtual-navigation decision task. Nature 484, 62–68 (2012).
Mankin, E.A. et al. Neuronal code for extended time in the hippocampus. Proc. Natl. Acad. Sci. USA 109, 19462–19467 (2012).
Naya, Y. & Suzuki, W.A. Integrating what and when across the primate medial temporal lobe. Science 333, 773–776 (2011).
Koenig, J., Linder, A.N., Leutgeb, J.K. & Leutgeb, S. The spatial periodicity of grid cells is not sustained during reduced theta oscillations. Science 332, 592–595 (2011).
Fell, J., Klaver, P., Elger, C.E. & Fernandez, G. The interaction of rhinal cortex and hippocampus in human declarative memory formation. Rev. Neurosci. 13, 299–312 (2002).
Acknowledgements
We thank G. Fishell for comments, Heather McKellar for assistance, and the US National Institutes of Health (NS34994; MH54671; NS 074015), International Human Frontiers Science Program Organization (RGP0032/2011), James S. McDonnell Foundation, Global Institute for Scientific Thinking, European Research Council ('CIRCUIT' Advanced Investigator Grant, Grant Agreement 232608), Louis-Jeantet Prize for Medicine, Kavli Foundation and Centre of Excellence scheme of the Research Council of Norway for support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Buzsáki, G., Moser, E. Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat Neurosci 16, 130–138 (2013). https://doi.org/10.1038/nn.3304
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3304
This article is cited by
-
Minute-scale oscillatory sequences in medial entorhinal cortex
Nature (2024)
-
Subfield-specific interneuron circuits govern the hippocampal response to novelty in male mice
Nature Communications (2024)
-
Increases in pre-stimulus theta and alpha oscillations precede successful encoding of crossmodal associations
Scientific Reports (2024)
-
Reinforcement Learning Navigation for Robots Based on Hippocampus Episode Cognition
Journal of Bionic Engineering (2024)
-
Systematic characterization of a non-transgenic Aβ1–42 amyloidosis model: synaptic plasticity and memory deficits in female and male mice
Biology of Sex Differences (2023)