Abstract
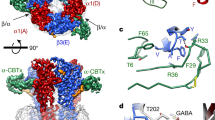
Inhibitory neurotransmission is mediated primarily by GABA. The metabotropic GABAB receptor is a G protein–coupled receptor central to mammalian brain function. Malfunction of GABAB receptor has been implicated in several neurological disorders. GABAB receptor functions as a heterodimeric assembly of GBR1 and GBR2 subunits, where GBR1 is responsible for ligand-binding and GBR2 is responsible for G protein coupling. Here we demonstrate that the GBR2 ectodomain directly interacts with the GBR1 ectodomain to increase agonist affinity by selectively stabilizing the agonist-bound conformation of GBR1. We present the crystal structure of the GBR2 ectodomain, which reveals a polar heterodimeric interface. We also identify specific heterodimer contacts from both subunits, and GBR1 residues involved in ligand recognition. Lastly, our structural and functional data indicate that the GBR2 ectodomain adopts a constitutively open conformation, suggesting a structural asymmetry in the active state of GABAB receptor that is unique to the GABAergic system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Macdonald, R.L. & Olsen, R.W. GABAA receptor channels. Annu. Rev. Neurosci. 17, 569–602 (1994).
Bettler, B., Kaupmann, K., Mosbacher, J. & Gassmann, M. Molecular structure and physiological functions of GABAB receptors. Physiol. Rev. 84, 835–867 (2004).
Bowery, N.G. et al. International Union of Pharmacology. XXXIII. Mammalian gamma-aminobutyric acidB receptors: structure and function. Pharmacol. Rev. 54, 247–264 (2002).
Pin, J.P. et al. The activation mechanism of class-C G protein coupled receptors. Biol. Cell 96, 335–342 (2004).
Kunishima, N. et al. Structural basis of glutamate recognition by a dimeric metabotropic glutamate receptor. Nature 407, 971–977 (2000).
Tsuji, Y. et al. Cryptic dimer interface and domain organization of the extracellular region of metabotropic glutamate receptor subtype 1. J. Biol. Chem. 275, 28144–28151 (2000).
Kniazeff, J. et al. Closed state of both binding domains of homodimeric mGlu receptors is required for full activity. Nat. Struct. Mol. Biol. 11, 706–713 (2004).
Muto, T., Tsuchiya, D., Morikawa, K. & Jingami, H. Structures of the extracellular regions of the group II/III metabotropic glutamate receptors. Proc. Natl. Acad. Sci. USA 104, 3759–3764 (2007).
Tsuchiya, D., Kunishima, N., Kamiya, N., Jingami, H. & Morikawa, K. Structural views of the ligand-binding cores of a metabotropic glutamate receptor complexed with an antagonist and both glutamate and Gd3+. Proc. Natl. Acad. Sci. USA 99, 2660–2665 (2002).
Jones, K.A. et al. GABAB receptors function as a heteromeric assembly of the subunits GABABR1 and GABABR2. Nature 396, 674–679 (1998).
Kaupmann, K. et al. GABAB-receptor subtypes assemble into functional heteromeric complexes. Nature 396, 683–687 (1998).
Kuner, R. et al. Role of heteromer formation in GABAB receptor function. Science 283, 74–77 (1999).
Ng, G.Y. et al. Identification of a GABAB receptor subunit, gb2, required for functional GABAB receptor activity. J. Biol. Chem. 274, 7607–7610 (1999).
White, J.H. et al. Heterodimerization is required for the formation of a functional GABAB receptor. Nature 396, 679–682 (1998).
Milligan, G. G protein-coupled receptor hetero-dimerization: contribution to pharmacology and function. Br. J. Pharmacol. 158, 5–14 (2009).
Margeta-Mitrovic, M., Jan, Y.N. & Jan, L.Y. A trafficking checkpoint controls GABA(B) receptor heterodimerization. Neuron 27, 97–106 (2000).
Duthey, B. et al. A single subunit (GB2) is required for G protein activation by the heterodimeric GABAB receptor. J. Biol. Chem. 277, 3236–3241 (2002).
Galvez, T. et al. Allosteric interactions between GB1 and GB2 subunits are required for optimal GABAB receptor function. EMBO J. 20, 2152–2159 (2001).
Havlickova, M. et al. The intracellular loops of the GB2 subunit are crucial for G protein coupling of the heteromeric gamma-aminobutyrate B receptor. Mol. Pharmacol. 62, 343–350 (2002).
Margeta-Mitrovic, M., Jan, Y.N. & Jan, L.Y. Function of GB1 and GB2 subunits in G protein coupling of GABAB receptors. Proc. Natl. Acad. Sci. USA 98, 14649–14654 (2001).
Robbins, M.J. et al. GABAB2 is essential for G protein coupling of the GABAB receptor heterodimer. J. Neurosci. 21, 8043–8052 (2001).
Monnier, C. et al. Trans-activation between 7transmembrane domains: implication in heterodimeric GABAB receptor activation. EMBO J. 30, 32–42 (2011).
Malitschek, B. et al. The N-terminal domain of gamma-aminobutyric acidB receptors is sufficient to specify agonist and antagonist binding. Mol. Pharmacol. 56, 448–454 (1999).
Kniazeff, J., Galvez, T., Labesse, G. & Pin, J.P. No ligand binding in the GB2 subunit of the GABAB receptor is required for activation and allosteric interaction between the subunits. J. Neurosci. 22, 7352–7361 (2002).
Liu, J. et al. Molecular determinants involved in the allosteric control of agonist affinity in the GABAB receptor by the GABAB2 subunit. J. Biol. Chem. 279, 15824–15830 (2004).
Nomura, R., Suzuki, Y., Kakizuka, A. & Jingami, H. Direct detection of the interaction between recombinant soluble extracellular regions in the heterodimeric metabotropic gamma-aminobutyric acid receptor. J. Biol. Chem. 283, 4665–4673 (2008).
Quick, M. & Javitch, J.A. Monitoring the function of membrane transport proteins in detergent-solubilized form. Proc. Natl. Acad. Sci. USA 104, 3603–3608 (2007).
Kaupmann, K. et al. Expression cloning of GABAB receptors uncovers similarity to metabotropic glutamate receptors. Nature 386, 239–246 (1997).
He, X., Chow, D., Martick, M.M. & Garcia, K.C. Allosteric activation of a spring-loaded natriuretic peptide receptor dimer by hormone. Science 293, 1657–1662 (2001).
He, X.L., Dukkipati, A. & Garcia, K.C. Structural determinants of natriuretic peptide receptor specificity and degeneracy. J. Mol. Biol. 361, 698–714 (2006).
Ogawa, H., Qiu, Y., Ogata, C.M. & Misono, K.S. Crystal structure of hormone-bound atrial natriuretic peptide receptor extracellular domain: rotation mechanism for transmembrane signal transduction. J. Biol. Chem. 279, 28625–28631 (2004).
van den Akker, F. et al. Structure of the dimerized hormone-binding domain of a guanylyl-cyclase-coupled receptor. Nature 406, 101–104 (2000).
Armstrong, N., Sun, Y., Chen, G.Q. & Gouaux, E. Structure of a glutamate-receptor ligand-binding core in complex with kainate. Nature 395, 913–917 (1998).
Sack, J.S., Saper, M.A. & Quiocho, F.A. Periplasmic binding protein structure and function. Refined X-ray structures of the leucine/isoleucine/valine-binding protein and its complex with leucine. J. Mol. Biol. 206, 171–191 (1989).
Rondard, P. et al. Functioning of the dimeric GABAB receptor extracellular domain revealed by glycan wedge scanning. EMBO J. 27, 1321–1332 (2008).
Galvez, T. et al. Mutagenesis and modeling of the GABAB receptor extracellular domain support a Venus flytrap mechanism for ligand binding. J. Biol. Chem. 274, 13362–13369 (1999).
Galvez, T. et al. Mapping the agonist-binding site of GABAB type 1 subunit sheds light on the activation process of GABAB receptors. J. Biol. Chem. 275, 41166–41174 (2000).
Matsushita, S., Nakata, H., Kubo, Y. & Tateyama, M. Ligand-induced rearrangements of the GABAB receptor revealed by fluorescence resonance energy transfer. J. Biol. Chem. 285, 10291–10299 (2010).
Rovira, X., Pin, J.P. & Giraldo, J. The asymmetric/symmetric activation of GPCR dimers as a possible mechanistic rationale for multiple signalling pathways. Trends Pharmacol. Sci. 31, 15–21 (2010).
Han, Y., Moreira, I.S., Urizar, E., Weinstein, H. & Javitch, J.A. Allosteric communication between protomers of dopamine class A GPCR dimers modulates activation. Nat. Chem. Biol. 5, 688–695 (2009).
Goudet, C. et al. Asymmetric functioning of dimeric metabotropic glutamate receptors disclosed by positive allosteric modulators. J. Biol. Chem. 280, 24380–24385 (2005).
Margeta-Mitrovic, M., Jan, Y.N. & Jan, L.Y. Ligand-induced signal transduction within heterodimeric GABAB receptor. Proc. Natl. Acad. Sci. USA 98, 14643–14648 (2001).
Pape, T. & Schneider, T.R. HKL2MAP: a graphical user interface for phasing with SHELX programs. J. Appl. Crystallogr. 37, 843–844 (2004).
De la Fortelle, E. & Bricogne, G. Maximum-likelihood heavy-atom parameter refinement for multiple isomorphous replacement and multiwavelength anomalous diffraction methods. Methods Enzymol. 276, 472–494 (1997).
Roversi, P., Blanc, E., Vonrhein, C., Evans, G. & Bricogne, G. Modelling prior distributions of atoms for macromolecular refinement and completion. Acta Crystallogr. D Biol. Crystallogr. 56, 1316–1323 (2000).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum–likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Chen, V.B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Navaza, J. Implementation of molecular replacement in AMoRe. Acta Crystallogr. D Biol. Crystallogr. 57, 1367–1372 (2001).
Eswar, N., Eramian, D., Webb, B., Shen, M.Y. & Sali, A. Protein structure modeling with MODELLER. Methods Mol. Biol. 426, 145–159 (2008).
Maurel, D. et al. Cell-surface protein-protein interaction analysis with time-resolved FRET and snap-tag technologies: application to GPCR oligomerization. Nat. Methods 5, 561–567 (2008).
Kniazeff, J. et al. Locking the dimeric GABAB G protein-coupled receptor in its active state. J. Neurosci. 24, 370–377 (2004).
Acknowledgements
We thank W.A. Hendrickson for advice and for generously providing lab space at the initial stage of this project, R. Kass for support and advice on electrophysiology experiments, L. Jan (University of California, San Francisco) for the cDNA clones encoding GIRK channels, V. Frasca for isothermal titration calorimetry and DSC measurements, C. Brouillette and I. Protassevitch for DSC measurements, L. Shapiro and J. Hunt for advice, M. Gawinowicz for mass spectrometry and protein sequencing, K. Rajashankar at the Advanced Proton Source and J. Schwanof at the National Synchrotron Light Source for help with data collection, L. Geneve for analyzing the glycosylation mutants, the ARPEGE platform facility at Institute of Functional Genomics (Montpellier, France) for inositol phosphate and Tag-lite (Cisbio) assays, and W.A. Hendrickson and M. Evelyn for critical reading of the manuscript. This work was supported by American Heart Association grant SDG0835183N, US National Institutes of Health grant R01GM088454 (both to Q.R.F.) and Agence Nationale de la Recherche (France) grant ANR-09-BLANC-0272 (to J.-P.P.). Q.R.F. is an Irma Hirschl Career Scientist, Pew Scholar and McKnight Scholar.
Author information
Authors and Affiliations
Contributions
Q.R.F. conceived the study, designed the experiments and wrote the paper; Q.R.F., Y.G., D.X., L.M., D.L.M., Y.C., S.B. and M.B. performed experiments and analyzed data; J.K. and J.-P.P. designed and carried out studies of glycosylation mutants; M.Q. and J.A.J. supervised SPA experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1–5 (PDF 1042 kb)
Rights and permissions
About this article
Cite this article
Geng, Y., Xiong, D., Mosyak, L. et al. Structure and functional interaction of the extracellular domain of human GABAB receptor GBR2. Nat Neurosci 15, 970–978 (2012). https://doi.org/10.1038/nn.3133
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3133
This article is cited by
-
Filamin A organizes γ‑aminobutyric acid type B receptors at the plasma membrane
Nature Communications (2023)
-
Mechanism of sensitivity modulation in the calcium-sensing receptor via electrostatic tuning
Nature Communications (2022)
-
PTH hypersecretion triggered by a GABAB1 and Ca2+-sensing receptor heterocomplex in hyperparathyroidism
Nature Metabolism (2020)
-
Structure of human GABAB receptor in an inactive state
Nature (2020)
-
A genetically encoded fluorescent sensor for in vivo imaging of GABA
Nature Methods (2019)