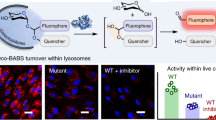
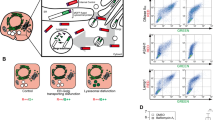
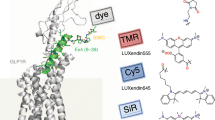
Abstract
Deficiency of glucocerebrosidase (GBA) underlies Gaucher disease, a common lysosomal storage disorder. Carriership for Gaucher disease has recently been identified as major risk for parkinsonism. Presently, no method exists to visualize active GBA molecules in situ. We here report the design, synthesis and application of two fluorescent activity-based probes allowing highly specific labeling of active GBA molecules in vitro and in cultured cells and mice in vivo. Detection of in vitro labeled recombinant GBA on slab gels after electrophoresis is in the low attomolar range. Using cell or tissue lysates, we obtained exclusive labeling of GBA molecules. We present evidence from fluorescence-activated cell sorting analysis, fluorescence microscopy and pulse-chase experiments of highly efficient labeling of GBA molecules in intact cells as well as tissues of mice. In addition, we illustrate the use of the fluorescent probes to study inhibitors and tentative chaperones in living cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Brady, R.O., Kanfer, J.N., Bradley, R.M. & Shapiro, D. Demonstration of a deficiency of glucocerebroside-cleaving enzyme in Gaucher's disease. J. Clin. Invest. 45, 1112–1115 (1966).
Grabowski, G.A. Phenotype, diagnosis, and treatment of Gaucher's disease. Lancet 372, 1263–1271 (2008).
Goker-Alpan, O. et al. The spectrum of Parkinsonian manifestations associated with glucocerebrosidase mutations. Arch. Neurol. 65, 1353–1357 (2008).
Van Weely, S. et al. Clinical genotype of Gaucher disease in relation to properties of mutant glucocerebrosidase in cultured fibroblasts. Biochim. Biophys. Acta 1096, 301–311 (1991).
Lachmann, R.H., Grant, I.R., Halsall, D. & Cox, T.M. Twin pairs showing discordance of phenotype in adult Gaucher's disease. QJM 97, 199–204 (2004).
Barton, N.W. et al. Replacement therapy for inherited enzyme deficiency–macrophage-targeted glucocerebrosidase for Gaucher's disease. N. Engl. J. Med. 324, 1464–1470 (1991).
Grabowski, G.A. et al. Enzyme therapy in type 1 Gaucher disease: comparative efficacy of mannose-terminated glucocerebrosidase from natural and recombinant sources. Ann. Intern. Med. 122, 33–39 (1995).
Aerts, J.M., Hollak, C.E., Boot, R.G., Groener, J.E. & Maas, M. Substrate reduction therapy of glycosphingolipid storage disorders. J. Inherit. Metab. Dis. 29, 449–456 (2006).
Platt, F.M., Neises, G.R., Dwek, R.A. & Butters, T.D. N-butyldeoxynojirimycin is a novel inhibitor of glycolipid biosynthesis. J. Biol. Chem. 269, 8362–8365 (1994).
Jonsson, L.M.V. et al. Biosynthesis and maturation of glucocerebrosidase in Gaucher fibroblasts. Eur. J. Biochem. 164, 171–179 (1987).
Ohashi, T. et al. Characterization of human glucocerebrosidase from different mutant alleles. J. Biol. Chem. 266, 3661–3667 (1991).
Sawkar, A.R. et al. Gaucher disease-associated glucocerebrosidases show mutation-dependent chemical chaperoning profiles. Chem. Biol. 12, 1235–1244 (2005).
Sawkar, A.R. et al. Chemical chaperones increase the cellular activity of N370S beta-glucosidase: a therapeutic strategy for Gaucher disease. Proc. Natl. Acad. Sci. USA 99, 15428–15433 (2002).
Ron, I. & Horowitz, M. ER retention and degradation as the molecular basis underlying Gaucher disease heterogeneity. Hum. Mol. Genet. 14, 2387–2398 (2005).
Mu, T.W. et al. Chemical and biological approaches synergize to ameliorate protein-folding diseases. Cell 134, 769–781 (2008).
Steet, R.A. et al. The iminosugar isofagomine increases the activity of N370S mutant acid beta-glucosidase in Gaucher fibroblasts by several mechanisms. Proc. Natl. Acad. Sci. USA 103, 13813–13818 (2006).
Yu, Z., Sawkar, A.R., Whalen, L.J., Wong, C.H. & Kelly, J.W. Isofagomine- and 2,5-anhydro-2,5-imino-D-glucitol-based glucocerebrosidase pharmacological chaperones for Gaucher disease intervention. J. Med. Chem. 50, 94–100 (2007).
Lieberman, R.L. et al. Structure of acid beta-glucosidase with pharmacological chaperone provides insight into Gaucher disease. Nat. Chem. Biol. 3, 101–107 (2007).
Kornhaber, G.J. et al. Isofagomine induced stabilization of glucocerebrosidase. ChemBioChem 9, 2643–2649 (2008).
Shen, J.S., Edwards, N.J., Hong, Y.B. & Murray, G.J. Isofagomine increases lysosomal delivery of exogenous glucocerebrosidase. Biochem. Biophys. Res. Commun. 369, 1071–1075 (2008).
Liou, B. & Grabowski, G.A. Participation of asparagine 370 and glutamine 235 in the catalysis by acid beta-glucosidase: the enzyme deficient in Gaucher disease. Mol. Genet. Metab. 97, 65–74 (2009).
Rempel, B.P. & Withers, S.G. Covalent inhibitors of glycosidases and their applications in biochemistry and biology. Glycobiology 18, 570–586 (2008).
Atsumi, S., Nosaka, C., Iinuma, H. & Umezawa, K. Accumulation of tissue glucosylsphingosine in Gaucher-like mouse induced by the glucosylceramidase inhibitor cyclophellitol. Arch. Biochem. Biophys. 304, 302–304 (1993).
Hansen, F.G., Bundgaard, E. & Madsen, R. A short synthesis of (+)-cyclophellitol. J. Org. Chem. 70, 10139 (2005).
Brumshtein, B. et al. Crystal structures of complexes of N-butyl- and N-nonyl-deoxynojirimycin bound to acid beta-glucosidase: insights into the mechanisms of chemical chaperone action in Gaucher disease. J. Biol. Chem. 282, 29052 (2007).
Kacher, Y. et al. Acid beta-glucosidase: insights from structural analysis and relevance to Gaucher disease therapy. Biol. Chem. 389, 1361–1369 (2008).
Overkleeft, H.S. et al. Generation of specific deoxynojirimycin-type inhibitors of the non-lysosomal glucosylceramidase. J. Biol. Chem. 273, 26522–26527 (1998).
Arribas, J.C. et al. Differential mechanism-based labeling and unequivocal activity assignment of the two active sites of intestinal lactase/phlorizin hydrolase. Eur. J. Biochem. 267, 6996–7005 (2000).
Aerts, J.M. et al. Glucocerebrosidase, a lysosomal enzyme that does not undergo oligosaccharide phosphorylation. Biochim. Biophys. Acta 964, 303–308 (1988).
van Weely, S. et al. Role of pH in determining the cell-type-specific residual activity of glucocerebrosidase in type 1 Gaucher disease. J. Clin. Invest. 91, 1167–1175 (1993).
Chang, H.H., Asano, N., Ishii, S., Ichikawa, Y. & Fan, J.Q. Hydrophilic iminosugar active-sitespecific chaperones increase residual glucocerebrosidase activity in fibroblasts from Gaucher patients. FEBS J. 273, 4082–4092 (2006).
Rudensky, B. et al. Fluorescent flow cytometric assay: a new diagnostic tool for measuring beta-glucocerebrosidase activity in Gaucher disease. Blood Cells Mol. Dis. 30, 97–99 (2003).
Gloster, T.M., Madsen, R. & Davies, G.J. Structural basis for cyclophellitol inhibition of a betaglucosidase. Org. Biomol. Chem. 5, 444–446 (2007).
Zechel, D.L. et al. Iminosugar glycosidase inhibitors: structural and thermodynamic dissection of the binding of isofagomine and 1-deoxynojirimycin to beta-glucosidases. J. Am. Chem. Soc. 125, 14313–14323 (2003).
Phenix, C.P. et al. Imaging of enzyme replacement therapy using PET. Proc. Natl. Acad. Sci. USA 107, 10842–10847 (2010).
Aerts, J.M.F.G. et al. A procedure for the rapid purification in high yield of human glucocerebrosidase using immunoaffinity chromatography with monoclonal antibodies. Anal. Biochem. 154, 655 (1986).
Boot, R.G. et al. Glucocerebrosidase genotype of Gaucher patients in The Netherlands: Limitations in prognostic value. Hum. Mutat. 10, 348 (1997).
Andersson, U., Butters, T.D., Dwek, R.A. & Platt, F.M. N-butyldeoxygalactonojirimycin: a more selective inhibitor of glycosphingolipid biosynthesis than N-butyldeoxynojirimycin, in vitro and in vivo. Biochem. Pharmacol. 59, 821 (2000).
Acknowledgements
Funding from The Netherlands Organization for Scientific Research (NWO-CW, to M.D.W., W.W.K., R.G.B., G.v.d.M., H.S.O. and J.M.F.G.A.) and The Netherlands Proteomics Centre (to B.I.F. and H.S.O.) is acknowledged.
Author information
Authors and Affiliations
Contributions
M.D.W. and W.W.K. designed the experiments. M.D.W., K.-Y.L. and A.M.C.H.v.d.N. conducted the synthesis, and W.W.K. conducted the kinetic experiments and the cell assays. J.A. conducted the microscopic analysis. A.S. conducted the in situ assays. W.E.D.-K. conducted the in vitro assays. B.B. conducted the structural modeling. G.K. and B.I.F. conducted the proteomic experiments. B.H. conducted the FACS analysis. C.E.M.H. conducted the tissue culture. R.O. conducted the animal experiments. R.G.B. supervised the cell assays. G.v.d.M. supervised the synthesis. H.S.O. and J.M.F.G.A. conceived of the idea and supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14, Supplementary Methods and Supplementary Results (PDF 10099 kb)
Supplementary Video
Time-lapse microscopy of GBA labeling in intact fibroblasts. Cells incubated with 5 nM MDW941 6 were imaged every 5 minutes for 2 hours simultaneously using fluorescence microscopy and phase-contrast bright field microscopy. (AVI 180003 kb)
Rights and permissions
About this article
Cite this article
Witte, M., Kallemeijn, W., Aten, J. et al. Ultrasensitive in situ visualization of active glucocerebrosidase molecules. Nat Chem Biol 6, 907–913 (2010). https://doi.org/10.1038/nchembio.466
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.466
This article is cited by
-
Activity-based protein profiling reveals dynamic substrate-specific cellulase secretion by saprotrophic basidiomycetes
Biotechnology for Biofuels and Bioproducts (2022)
-
Quantifying lysosomal glycosidase activity within cells using bis-acetal substrates
Nature Chemical Biology (2022)
-
Lysosomal integral membrane protein-2 as a phospholipid receptor revealed by biophysical and cellular studies
Nature Communications (2017)
-
Activity-based probes for functional interrogation of retaining β-glucuronidases
Nature Chemical Biology (2017)
-
Small molecules as therapeutic agents for inborn errors of metabolism
Journal of Inherited Metabolic Disease (2017)