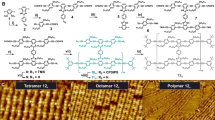
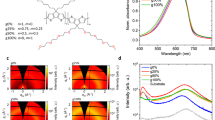
Abstract
Conjugated polymers offer potential for many diverse applications, but we still lack a fundamental microscopic understanding of their electronic structure. Elementary photoexcitations (excitons) span only a few nanometres of a molecule, which itself can extend over microns, and how their behaviour is affected by molecular dimensions is not immediately obvious. For example, where is the exciton formed within a conjugated segment and is it always situated on the same repeat units? Here, we introduce structurally rigid molecular spoked wheels, 6 nm in diameter, as a model of extended π conjugation. Single-molecule fluorescence reveals random exciton localization, which leads to temporally varying emission polarization. Initially, this random localization arises after every photon absorption event because of temperature-independent spontaneous symmetry breaking. These fast fluctuations are slowed to millisecond timescales after prolonged illumination. Intramolecular heterogeneity is revealed in cryogenic spectroscopy by jumps in transition energy, but emission polarization can also switch without a spectral jump occurring, which implies long-range homogeneity in the local dielectric environment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Scholes, G. D., Fleming, G. R., Olaya-Castro, A. & van Grondelle, R. Lessons from nature about solar light harvesting. Nature Chem. 3, 763–774 (2011).
Hwang, I. & Scholes, G. D. Electronic energy transfer and quantum-coherence in π-conjugated polymers. Chem. Mater. 23, 610–620 (2011).
Lupton, J. M. Chromophores in conjugated polymers—all straight? Chem. Phys. Chem. 13, 901–907 (2012).
Hu, D. H. et al. Collapse of stiff conjugated polymers with chemical defects into ordered, cylindrical conformations. Nature 405, 1030–1033 (2000).
Schwartz, B. J. Conjugated polymers: what makes a chromophore? Nature Mater. 7, 427–428 (2008).
Schwartz, B. J. Conjugated polymers as molecular materials: how chain conformation and film morphology influence energy transfer and interchain interactions. Annu. Rev. Phys. Chem. 54, 141–172 (2003).
Bässler, H. & Schweitzer, B. Site-selective fluorescence spectroscopy of conjugated polymers and oligomers. Acc. Chem. Res. 32, 173–182 (1999).
Tempelaar, R., Stradomska, A., Knoester, J. & Spano, F. C. Anatomy of an exciton: vibrational distortion and exciton coherence in H- and J-aggregates. J. Phys. Chem. B 117, 457–466 (2013).
Lippitz, M. et al. Coherent electronic coupling versus localization in individual molecular dimers. Phys. Rev. Lett. 92, 103001 (2004).
Hernando, J. et al. Single molecule photobleaching probes the exciton wave function in a multichromophoric system. Phys. Rev. Lett. 93, 236404 (2004).
Tretiak, S., Saxenea, A., Martin, R. L. & Bishop, A. R. Conformational dynamics of photoexcited conjugated molecules. Phys. Rev. Lett. 89, 097402 (2002).
Ruseckas, A. et al. Ultrafast depolarization of the fluorescence in a conjugated polymer. Phys. Rev. B 72, 115214 (2005).
Mössinger, D., Hornung, J., Lei, S., De Feyter, S. & Höger, S. Molecularly defined shape-persistent 2D oligomers: the covalent-template approach to molecular spoked wheels. Angew. Chem. Int. Ed. 46, 6802–6806 (2007).
Mayor, M. & Didschies, C. A giant conjugated molecular ring. Angew. Chem. Int. Ed. 42, 3176–3179 (2003).
Mössinger, D. et al. Large all-hydrocarbon spoked wheels of high symmetry: modular synthesis, photophysical properties, and surface assembly. J. Am. Chem. Soc. 132, 1410–1423 (2010).
Jung, S. H. et al. A conjugated polycarbazole ring around a porphyrin. Angew. Chem. Int. Ed. 45, 4685–4690 (2006).
Simon, S. C., Schmaltz, B., Rouhanipour, A., Räder, H. J., & Müllen, K. A macrocyclic model dodecamer for polyfluorenes. Adv. Mater. 21, 83–85 (2009).
Arnold, L., Norouzi-Arasi, H., Wagner, M., Enkelmann, V. & Müllen, K. A porphyrin-related macrocycle from carbazole and pyridine building blocks: synthesis and metal coordination. Chem. Commun. 47, 970–972 (2011).
Cho, H. S. et al. Excitation energy transport processes of porphyrin monomer, dimer, cyclic trimer, and hexamer probed by ultrafast fluorescence anisotropy decay. J. Am. Chem. Soc. 125, 5849–5860 (2003).
Hori, T. et al. Giant porphyrin wheels with large electronic coupling as models of light-harvesting photosynthetic antenna. Chem. Eur. J. 12, 1319–1327 (2006).
Sprafke, J. K. et al. Belt-shaped π-systems: relating geometry to electronic structure in a six-porphyrin nanoring. J. Am. Chem. Soc. 133, 17262–17273 (2011).
O'Sullivan, M. C. et al. Vernier templating and synthesis of a 12-porphyrin nano-ring. Nature 469, 72–75 (2011).
Yang, J., Yoon, M. C., Yoo, H., Kim, P., & Kim, D. Excitation energy transfer in multiporphyrin arrays with cyclic architectures: towards artificial light-harvesting antenna complexes. Chem. Soc. Rev. 41, 4808–4826 (2012).
Hoffmann, M. et al. Enhanced π conjugation around a porphyrin[6] nanoring. Angew. Chem. Int. Ed. 47, 4993–4996 (2008).
Iyoda, M., Yamakawa, J. & Rahman, M. J. Conjugated macrocycles: concepts and applications. Angew. Chem. Int. Ed. 50, 10522–10553 (2011).
Kawase, T. & Kurata, H. Ball-, bowl-, and belt-shaped conjugated systems and their complexing abilities: exploration of the concave–convex π−π interaction. Chem. Rev. 106, 5250–5273 (2006).
Kawase, T. et al. Cyclic [5]paraphenyleneacetylene: synthesis, properties, and formation of a ring-in-ring complex showing a considerably large association constant and entropy effect. Angew. Chem. Int. Ed. 46, 1086–1088 (2007).
Höger, S. & Bonrad, K. 3-Cyanopropyl-dimethysilyl acetylene (CPDMS acetylene), a polar analogue of trimethylsilyl acetylene: synthesis and applications in the preparation of monoprotected bisacetylenes. J. Org. Chem. 65, 2243–2245 (2000).
Bednarz, M., Reineker, P., Mena-Osteritz, E. & Bäuerle, P. Optical absorption spectra of linear and cyclic thiophenes—selection rules manifestation. J. Lumin. 110, 225–231 (2004).
Bhaskar, A. et al. Enhancement of two-photon absorption cross-section in macrocyclic thiophenes with cavities in the nanometer regime. J. Phys. Chem. B 111, 946–954 (2007).
Mena-Osteritz, E., Zhang, F., Gotz, G., Reineker, P. & Bäuerle, P. Optical properties of fully conjugated cyclo[n]thiophenes—an experimental and theoretical approach. Beilstein J. Nanotech. 2, 720–726 (2011).
Varnavski, O., Bäuerle, P. & Goodson, T. Strong coupling in macrocyclic thiophene investigated by time-resolved two-photon excited fluorescence. Opt. Lett. 32, 3083–3085 (2007).
Zhang, F., Gotz, G., Winkler, H. D. F., Schalley, C. A. & Bäuerle, P. Giant cyclo[n]thiophenes with extended π conjugation. Angew. Chem. Int. Ed. 48, 6632–6635 (2009).
Kunz, R. et al. Exciton self trapping in photosynthetic pigment–protein complexes studied by single-molecule spectroscopy. J. Phys. Chem. B 116, 11017–11023 (2012).
Tretiak, S. & Mukamel, S. Density matrix analysis and simulation of electronic excitations in conjugated and aggregated molecules. Chem. Rev. 102, 3171–3212 (2002).
Tubasum, S., Cogdell, R. J., Scheblykin, I. G. & Pullerits, T. Excitation–emission polarization spectroscopy of single light harvesting complexes. J. Phys. Chem. B 115, 4963–4970 (2011).
Tubasum, S., Thomsson, D., Cogdell, R., Scheblykin, I. & Pullerits, T. Polarization single complex imaging of circular photosynthetic antenna. Photosyn. Res. 111, 41–45 (2012).
Bopp, M. A., Sytnik, A., Howard, T. D., Cogdell, R. J. & Hochstrasser, R. M. The dynamics of structural deformations of immobilized single light-harvesting complexes. Proc. Natl Acad. Sci. USA 96, 11271–11276 (1999).
Mirzov, O. et al. Polarization portraits of single multichromophoric systems: visualizing conformation and energy transfer. Small 5, 1877–1888 (2009).
Stangl, T. et al. Temporal switching of homo-FRET pathways in single-chromophore dimer models of π-conjugated polymers. J. Am. Chem. Soc. 135, 78–81 (2013).
Thomasson, D., Sforazzini, G., Anderson, H. L. & Scheblykin, I. G. Excitation polarization provides structural resolution of individual non-blinking nano-objects. Nanoscale 5, 3070–3077 (2013).
Forster, M., Thomsson, D., Hania, P. R. & Scheblykin, I. G. Redistribution of emitting state population in conjugated polymers probed by single-molecule fluorescence polarization spectroscopy. Phys. Chem. Chem. Phys. 9, 761–766 (2007).
Flors, C. et al. Energy and electron transfer in ethynylene bridged perylene diimide multichromophores. J. Phys. Chem. C 111, 4861–4870 (2007).
Feist, F. A., Tommaseo, G. & Basché, T. Observation of very narrow linewidths in the fluorescence excitation spectra of single conjugated polymer chains at 1.2 K. Phys. Rev. Lett. 98, 208301 (2007).
Schindler, F., Lupton, J. M., Feldmann, J. & Scherf, U. A universal picture of chromophores in π-conjugated polymers derived from single-molecule spectroscopy. Proc. Natl Acad. Sci. USA 101, 14695–14700 (2004).
Müller, J. G., Anni, M., Scherf, U., Lupton, J. M. & Feldmann, J. Vibrational fluorescence spectroscopy of single conjugated polymer molecules. Phys. Rev. B 70, 035205 (2004).
Wu, S. L., Liu, F., Shen, Y., Cao, J. S. & Silbey, R. J. Efficient energy transfer in light-harvesting systems, I: optimal temperature, reorganization energy and spatial–temporal correlations. New J. Phys. 12, 105012 (2010).
Higgins, D. A., VandenBout, D. A., Kerimo, J. & Barbara, P. F. Polarization-modulation near-field scanning optical microscopy of mesostructured materials. J. Phys. Chem. 100, 13794–13803 (1996).
Acknowledgements
The authors are indebted to the Volkswagen Foundation for providing collaborative funding. A.V.A. and A.T. acknowledge financial support by the Fonds der Chemischen Industrie. J.M.L. is a David & Lucile Packard Foundation fellow and is grateful for a European Research Council Starting Grant (MolMesON, #305020).
Author information
Authors and Affiliations
Contributions
A.V.A., A.I., D.K. and S.H. designed and synthesized the compounds. A.T., D.W., T.S., F.S., J.V. and J. M. L. conceived, designed and performed the spectroscopy experiments and analysed the data. S-S.J. and S.H. performed and interpreted the STM experiments. A.T., J.V., S.H. and J.M.L. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 4584 kb)
Rights and permissions
About this article
Cite this article
Aggarwal, A., Thiessen, A., Idelson, A. et al. Fluctuating exciton localization in giant π-conjugated spoked-wheel macrocycles. Nature Chem 5, 964–970 (2013). https://doi.org/10.1038/nchem.1758
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1758
This article is cited by
-
Nanoscale π-conjugated ladders
Nature Communications (2021)
-
Solvent tuning of photochemistry upon excited-state symmetry breaking
Nature Communications (2020)
-
Coherent exciton-vibrational dynamics and energy transfer in conjugated organics
Nature Communications (2018)
-
Using coherence to enhance function in chemical and biophysical systems
Nature (2017)
-
Supramolecular nesting of cyclic polymers
Nature Chemistry (2015)