Abstract
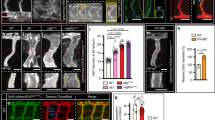
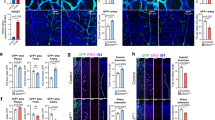
Angiogenesis, the formation of new blood vessels from pre-existing vessels, is critical to most physiological processes and many pathological conditions. During zebrafish development, angiogenesis expands the axial vessels into a complex vascular network that is necessary for efficient oxygen delivery. Although the dorsal aorta and the axial vein are spatially juxtaposed, the initial angiogenic sprouts from these vessels extend in opposite directions, indicating that distinct cues may regulate angiogenesis of the axial vessels. We found that angiogenic sprouts from the dorsal aorta are dependent on vascular endothelial growth factor A (Vegf-A) signalling, and do not respond to bone morphogenetic protein (Bmp) signals. In contrast, sprouts from the axial vein are regulated by Bmp signalling independently of Vegf-A signals, indicating that Bmp is a vein-specific angiogenic cue during early vascular development. Our results support a paradigm whereby different signals regulate distinct programmes of sprouting angiogenesis from the axial vein and dorsal aorta, and indicate that signalling heterogeneity contributes to the complexity of vascular networks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Isogai, S., Lawson, N., Torrealday, S., Horiguchi, M. & Weinstein, B. Angiogenic network formation in the developing vertebrate trunk. Development 130, 5281–5290 (2003).
Risau, W. Mechanisms of angiogenesis. Nature 386, 671–674 (1997).
Covassin, L., Villefranc, J., Kacergis, M., Weinstein, B. & Lawson, N. Distinct genetic interactions between multiple Vegf receptors are required for development of different blood vessel types in zebrafish. Proc. Natl Acad. Sci. USA 103, 6554–6559 (2006).
Herbert, S. et al. Arterial-venous segregation by selective cell sprouting: an alternative mode of blood vessel formation. Science 326, 294–298 (2009).
Lee, S. et al. Autocrine VEGF signalling is required for vascular homeostasis. Cell 130, 691–703 (2007).
Monteiro, R. et al. Two novel type II receptors mediate BMP signalling and are required to establish left-right asymmetry in zebrafish. Dev. Biol. 315, 55–71 (2008).
Langenfeld, E. M. & Langenfeld, J. Bone morphogenetic protein-2 stimulates angiogenesis in developing tumours. Mol. Cancer Res. 2, 141–149 (2004).
Sieber, C., Kopf, J., Hiepen, C. & Knaus, P. Recent advances in BMP receptor signalling. Cytokine Growth Factor Rev. 20, 343–355 (2009).
Halloran, M.C. et al. Laser-induced gene expression in specific cells of transgenic zebrafish. Development 127, 1953–1960 (2000).
Fürthauer, M., Thisse, B. & Thisse, C. Three different noggin genes antagonize the activity of bone morphogenetic proteins in the zebrafish embryo. Dev. Biol. 214, 181–196 (1999).
Derynck, R. & Zhang, Y. Smad-dependent and Smad-independent pathways in TGF-B family signalling. Nature 425, 577–584 (2003).
Pi, X. et al. Sequential roles for myosin-X in BMP6-dependent filopodial extension, migration, and activation of BMP receptors. J. Cell Biol. 179, 1569–1582 (2007).
Hao, J. et al. In vivo structure–activity relationship study of dorsomorphin analogues identifies selective VEGF and BMP inhibitors. ACS Chem. Biol. 19, 245–253 (2010).
Jeltsch, M. et al. Hyperplasia of lymphatic vessels in VEGF-C transgenic mice. Science 276, 1423–1425 (1997).
Hellström, M. et al. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 445, 776–780 (2007).
Siekmann, A. F. & Lawson, N. D. Notch signalling limits angiogenic cell behaviour in developing zebrafish arteries. Nature 445, 781–4 (2007).
Reese, D. E., Hall, C. E. & Mikawa, T. Negative regulation of midline vascular development by the notochord. Dev. Cell 6, 699–708 (2004).
Bressan, M., Davis, P., Timmer, J., Herzlinger, D. & Mikawa, T. Notochord-derived BMP antagonists inhibit endothelial cell generation and network formation. Dev. Biol. 326, 101–111 (2009).
Gu, Z. et al. The type I serine/threonine kinase receptor ActRIA (ALK2) is required for gastrulation of the mouse embryo. Development 126, 2551–61 (1999).
Mishina, Y., Crombie, R., Bradley, A. & Behringer, R. R. Multiple roles foractivin-like kinase-2 signalling during mouse embryogenesis. Dev. Biol. 213, 314–26 (1999).
Beppu, H. et al. BMP type II receptor is required for gastrulation and early development of mouse embryos. Dev. Biol. 221, 49–58 (2000).
Hong, K. H. et al. Genetic ablation of the BMPR2 gene in pulmonary endothelium is sufficient to predispose to pulmonary arterial hypertension. Circulation 118, 722–730 (2008).
Kleeff, J. et al. Bone morphogenetic protein 2 exerts diverse effects on cell growth in vitro and is expressed in human pancreatic cancer in vivo. Gastroenterology 116, 1202–1216 (1999).
Jin, Y. et al. Overexpression of BMP-2/4, -5 and BMPR-IA associated with malignancy of oral epithelium. Oral Oncol. 37, 225–233 (2001).
Langenfeld, E. M. et al. The mature bone morphogenetic protein-2 is aberrantly expressed in non-small cell lung carcinomas and stimulates tumour growth of A549 cells. Carcinogenesis 24, 1445–1454 (2003).
Ferrara, N. & Kerbel, R. S. Angiogenesis as a therapeutic target. Nature 438, 967–974 (2005).
Westerfield, M. The Zebrafish Book, a Guide for the Laboratory Use of Zebrafish (Danio Rerio) 4th edn (Univ. Oregon Press, 2000).
Roman, B. L. et al. Disruption of acvrl1 increases endothelial cell number in zebrafish cranial vessels. Development 129, 3009–3019 (2002).
Jin, S. W., Beis, D., Mitchell, T., Chen, J. N. & Stainier, D. Y. Cellular and molecular analyses of vascular tube and lumen formation in zebrafish. Development 132, 5199–5209 (2005).
Chi, N. C. et al. Genetic and physiologic dissection of the vertebrate cardiac conduction system. PLoS Biol. 6, 1006–1019 (2008).
Chocron, S., Verhoeven, M. C., Rentzsch, F., Hammerschmidt, M. & Bakkers, J. Zebrafish Bmp4 regulates left-right asymmetry at two distinct developmental time points. Dev. Biol. 305, 577–588 (2007).
Pyati, U. J., Webb, A. E. & Kimelman, D. Transgenic zebrafish reveal stage-specific roles for Bmp signalling in ventral and posterior mesoderm development. Development 132, 2333–2343 (2005).
Thisse, B. et al. Expression of the zebrafish genome during embryogenesis. ZFIN Direct Data Submission (http://zfin.org/cgi-bin/webdriver?MIval=aa-pubview2.apg&OID=ZDB-PUB-010810-1) (2001).
Thisse, C. & Thisse, B. Zebrafish Science Monitor, Vol. 5. (Univ. Oregon Press, 1998).
Nasevicius, A. & Ekker, S. C. Effective targeted gene ‘knockdown’ in zebrafish. Nat. Genet. 26, 216–220 (2000).
Kwan, et al. The Tol2kit: a multisite gateway-based construction kit for Tol2 transposon transgenesis constructs. Dev. Dyn. 236, 3088–3099 (2007).
Acknowledgements
The authors thank E. Flynn for excellent fish care; the members of the Jin and Bautch laboratories for discussions; the UNC Histology Facility; Michael Hooker Microscopy Facility; W. Comb and M. Aleman for technical assistance; and M. Hammerschmidt and N. Chi for providing transgenic lines, and C-B. Chien for the kdrl 5′ entry gateway vector. The authors also thank F. Conlon, M. Majesky, C. Patterson and J. Rawls for discussion and critical reading of the manuscript. This study was supported by grants from the NIH to S-W.J. (HL090960) and V.L.B. (HL43174 and HL86564), and the UNC Integrative Vascular Biology Training Grant (T32HL69768) and an American Heart Association Pre-doctoral Fellowship to D.M.W.
Author information
Authors and Affiliations
Contributions
D.M.W., V.L.B. and S-W.J. designed the experiments, D.M.W. carried out the experiments, J-D.K. helped with in situ hybridization, J.H. and C.C.H. provided key reagents and D.M.W., V.L.B. and S-W.J. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1179 kb)
Supplementary Information
Supplementary Movie 1 (AVI 1355 kb)
Supplementary Information
Supplementary Movie 2 (AVI 980 kb)
Supplementary Information
Supplementary Movie 3 (AVI 1645 kb)
Supplementary Information
Supplementary Movie 4 (AVI 2610 kb)
Supplementary Information
Supplementary Movie 5 (AVI 1972 kb)
Rights and permissions
About this article
Cite this article
Wiley, D., Kim, JD., Hao, J. et al. Distinct signalling pathways regulate sprouting angiogenesis from the dorsal aorta and the axial vein. Nat Cell Biol 13, 686–692 (2011). https://doi.org/10.1038/ncb2232
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2232
This article is cited by
-
The cytoskeleton adaptor protein Sorbs1 controls the development of lymphatic and venous vessels in zebrafish
BMC Biology (2024)
-
A defined clathrin-mediated trafficking pathway regulates sFLT1/VEGFR1 secretion from endothelial cells
Angiogenesis (2023)
-
Single-cell transcriptomic analysis of vascular endothelial cells in zebrafish embryos
Scientific Reports (2022)
-
Environmental and intrinsic modulations of venous differentiation
Cellular and Molecular Life Sciences (2022)
-
The versatility and paradox of BMP signaling in endothelial cell behaviors and blood vessel function
Cellular and Molecular Life Sciences (2022)