Abstract
Cancer stem cells (CSCs) have been hypothesized to represent the driving force behind tumour progression and metastasis, making them attractive cancer targets. However, conclusive experimental evidence for their functional relevance is still lacking for most malignancies. Here we show that the leucine-rich repeat-containing G-protein-coupled receptor 5 (Lgr5) identifies intestinal CSCs in mouse tumours engineered to recapitulate the clinical progression of human colorectal cancer. We demonstrate that selective Lgr5+ cell ablation restricts primary tumour growth, but does not result in tumour regression. Instead, tumours are maintained by proliferative Lgr5− cells that continuously attempt to replenish the Lgr5+ CSC pool, leading to rapid re-initiation of tumour growth upon treatment cessation. Notably, CSCs are critical for the formation and maintenance of liver metastasis derived from colorectal cancers. Together, our data highlight distinct CSC dependencies for primary versus metastasic tumour growth, and suggest that targeting CSCs may represent a therapeutic opportunity for managing metastatic disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Siegel, R., Desantis, C. & Jemal, A. Colorectal cancer statistics, 2014. CA Cancer J. Clin. 64, 104–117 (2014)
Fearon, E. R. & Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 61, 759–767 (1990)
O’Brien, C. A., Pollett, A., Gallinger, S. & Dick, J. E. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 445, 106–110 (2007)
Ricci-Vitiani, L. et al. Identification and expansion of human colon-cancer-initiating cells. Nature 445, 111–115 (2007)
Barker, N. et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457, 608–611 (2009)
Junttila, M. R. et al. Targeting LGR5+ cells with an antibody-drug conjugate for the treatment of colon cancer. Sci. Transl. Med. 7, 314ra186 (2015)
Kemper, K. et al. Monoclonal antibodies against LGR5 identify human colorectal cancer stem cells. Stem Cells 30, 2378–2386 (2012)
Schepers, A. G. et al. Lineage tracing reveals Lgr5+ stem cell activity in mouse intestinal adenomas. Science 337, 730–735 (2012)
Vermeulen, L ., de Sousa e Melo, F ., Richel, D. J . & Medema, J. P. The developing cancer stem-cell model: clinical challenges and opportunities. Lancet Oncol. 13, e83–e89 (2012)
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J. & Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl Acad. Sci. USA 100, 3983–3988 (2003)
Singh, S. K. et al. Identification of human brain tumour initiating cells. Nature 432, 396–401 (2004)
Chen, J. et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 488, 522–526 (2012)
Boumahdi, S. et al. SOX2 controls tumour initiation and cancer stem-cell functions in squamous-cell carcinoma. Nature 511, 246–250 (2014)
Enquist, I. B. et al. Lymph node-independent liver metastasis in a model of metastatic colorectal cancer. Nat. Commun. 5, 3530 (2014)
Tian, H. et al. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478, 255–259 (2011)
Huch, M. et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature 494, 247–250 (2013)
Drost, J. et al. Sequential cancer mutations in cultured human intestinal stem cells. Nature 521, 43–47 (2015)
Matano, M. et al. Modeling colorectal cancer using CRISPR–Cas9-mediated engineering of human intestinal organoids. Nat. Med. 21, 256–262 (2015)
Schwank, G. et al. Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell 13, 653–658 (2013)
Clarke, M. F. et al. Cancer stem cells—perspectives on current status and future directions: AACR workshop on cancer stem cells. Cancer Res. 66, 9339–9344 (2006)
Sheffer, M . et al. Association of survival and disease progression with chromosomal instability: a genomic exploration of colorectal cancer. Proc. Natl Acad. Sci. USA 106, 7131–7136 (2009)
Walther, A., Houlston, R. & Tomlinson, I. Association between chromosomal instability and prognosis in colorectal cancer: a meta-analysis. Gut 57, 941–950 (2008)
Muñoz, J. et al. The Lgr5 intestinal stem cell signature: robust expression of proposed quiescent ‘+4’ cell markers. EMBO J. 31, 3079–3091 (2012)
Kim, T. H. et al. Single-cell transcript profiles reveal multilineage priming in early progenitors derived from Lgr5+ intestinal stem cells. Cell Reports 16, 2053–2060 (2016)
Vermeulen, L. et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat. Cell Biol. 12, 468–476 (2010)
Kaiser, J. The cancer stem cell gamble. Science 347, 226–229 (2015)
Ashton, G. H. et al. Focal adhesion kinase is required for intestinal regeneration and tumorigenesis downstream of Wnt/c-Myc signaling. Dev. Cell 19, 259–269 (2010)
Ritsma, L. et al. Intestinal crypt homeostasis revealed at single-stem-cell level by in vivo live imaging. Nature 507, 362–365 (2014)
Asfaha, S. et al. Krt19+/Lgr5− cells are radioresistant cancer-initiating stem cells in the colon and intestine. Cell Stem Cell 16, 627–638 (2015)
Tetteh, P. W. et al. Replacement of lost Lgr5-positive stem cells through plasticity of their enterocyte-lineage daughters. Cell Stem Cell 18, 203–213 (2016)
Storm, E. E. et al. Targeting PTPRK-RSPO3 colon tumours promotes differentiation and loss of stem-cell function. Nature 529, 97–100 (2016)
Beumer, J. & Clevers, H. Regulation and plasticity of intestinal stem cells during homeostasis and regeneration. Development 143, 3639–3649 (2016)
Metcalfe, C., Kljavin, N. M., Ybarra, R. & de Sauvage, F. J. Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration. Cell Stem Cell 14, 149–159 (2014)
Pascual, G. et al. Targeting metastasis-initiating cells through the fatty acid receptor CD36. Nature 541, 41–45 (2017)
Thalheimer, A. et al. The intraportal injection model: a practical animal model for hepatic metastases and tumor cell dissemination in human colon cancer. BMC Cancer 9, 29 (2009)
Sharpe, H. J. et al. Genomic analysis of smoothened inhibitor resistance in basal cell carcinoma. Cancer Cell 27, 327–341 (2015)
Wu, T. D. & Nacu, S. Fast and SNP-tolerant detection of complex variants and splicing in short reads. Bioinformatics 26, 873–881 (2010)
Law, C. W., Chen, Y., Shi, W. & Smyth, G. K. voom: precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol. 15, R29 (2014)
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015)
Srinivasan, K. et al. Untangling the brain’s neuroinflammatory and neurodegenerative transcriptional responses. Nat. Commun. 7, 11295 (2016)
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079 (2009)
McKenna, A. et al. The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010)
Wilm, A. et al. LoFreq: a sequence-quality aware, ultra-sensitive variant caller for uncovering cell-population heterogeneity from high-throughput sequencing datasets. Nucleic Acids Res. 40, 11189–11201 (2012)
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011)
Boeva, V. et al. Control-FREEC: a tool for assessing copy number and allelic content using next-generation sequencing data. Bioinformatics 28, 423–425 (2012)
Yaari, G., Bolen, C. R., Thakar, J. & Kleinstein, S. H. Quantitative set analysis for gene expression: a method to quantify gene set differential expression including gene–gene correlations. Nucleic Acids Res. 41, e170 (2013)
Liberzon, A. et al. Molecular signatures database (MSigDB) 3.0. Bioinformatics 27, 1739–1740 (2011)
Hu, Y. & Smyth, G. K. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J. Immunol. Methods 347, 70–78 (2009)
Acknowledgements
We thank M. Junttila and K. Leong for generation of the AKVL mouse model, A. Bruce for help designing schematic figures, J. Jian for tissue processing, J. Stinson and the NGS laboratory for RNA-sequencing, J. Paw, C. Poon, T. Ho and J. Borneo for flow cytometry support, members of Laboratory Animal Resources for animal care and de Sauvage laboratory members for comments on the manuscript.
Author information
Authors and Affiliations
Contributions
F.d.S.M. and F.J.d.S. conceptualized the project and designed experiments. F.d.S.M. engineered and characterized all mutant organoid lines, produced concentrated lentivirus, optimized the orthotopic injection methodology and conducted bioluminescent imaging. F.d.S.M., A.V.K. and N.K. performed subcutaneous transplants, endoscopic imaging, DT treatment, tumour measurements and collected tissues. F.d.S.M. and A.V.K. conducted flow cytometry experiments. J.M.H. performed portal vein injections. G.J.P.D. modified pLKO-SHC201, cloned sgRNAs and designed pGEIL-tRFP. F.d.S.M., J.D.H., J.H., J.E.A. and E.E.S. either acquired or evaluated immunohistochemistry and/or immunofluorescence data. H.K. performed histopathological evaluation of tumour and liver tissues. Z.M. contributed next-generation sequencing data. R.P. provided bioinformatics support. F.d.S.M., G.J.P.D. and F.J.d.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
All authors are employees of Genentech and own Roche shares.
Additional information
Reviewer Information Nature thanks F. Greten, R. Shivdasani and the anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Validation of CRISPR–Cas9-mediated gene targeting.
a, b, Gene-targeting strategy for Trp53 (a) and Smad4 (b). The protospacer adjacent motif (PAM) is underlined on the target sequence and the allelic fraction of each haplotype is indicated. c, d, Sequence alignment of exome-sequencing reads at a representative predicted off-target site for one of the sgRNAs used to mutate either Trp53 (c) or Smad4 (d). Of note, neither single-nucleotide variants nor copy-number changes could be detected. e, Relative p21 (also known as Cdkn1a) expression in AKVL and AKVPL organoids cultured in either the presence or absence of Nutlin-3 (qPCR data was normalized to Actb; n = 2 independent AKVL clones; n = 3 independent AKVPL clones generated by 2 different sgRNAs targeting Trp53). f, Relative expression of Bax (a canonical Trp53-target gene) and Id3 (a canonical Smad4-target gene) in organoids with indicated genotype (RNA-seq expression analysis was quantile normalized and represented as log2 counts per million).
Extended Data Figure 2 Genotype affects CSC content and tumorigenic capacity.
a, Per cent tumour-initiating cells (TIC) in subcutaneous AKVPL and AKVPSL tumours estimated with the extreme limiting dilution analysis (ELDA48; n = 6 allografts per dilution; error bars represent the 95% confidence interval). b, Representative images of Lgr5-GFP+ staining of n = 4 tumours per indicated genotype. DAPI nuclear staining is in blue; Lgr5 in green. Scale bar, 20 μm. c, Per cent Lgr5-GFP+ cells in subcutaneous AKVPL and AKVPSL tumours collected three weeks after implantation (FACS data represented as mean ± s.d., n = 5 tumours per genotype). d, Per cent clonogenic cells in AKVPL and AKVPSL organoid cultures estimated with ELDA48 (n = 2 independent experiment per genotype; error bars represent the 95% confidence interval). e, Lgr5-GFP+ cell count and fluorescence intensity of in vitro organoid cultures with indicated genotype as determined by FACS. *P < 0.05, **P < 0.01, ***P < 0.001; n represents the number of independent animals.
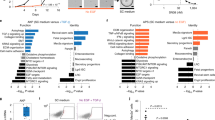
Extended Data Figure 3 Gene expression analysis and tumorigenic potential of Lgr5+ and Lgr5− cells across organoid tumours.
a, FACS plots for GFP expression in dissociated AKV, AKVPL and AKVPSL tumours. The green and grey gates represent the sorted Lgr5+ and Lgr5− populations, respectively. b, Heatmap of Lgr5-GFP+ and Lgr5-GFP− cells from n = 4 AKVPSL tumours for expression of intestinal stem cell (ISC) and canonical Wnt target genes (data are represented as mean-centred RPKM). c, QuSAGE of indicated gene signatures in Lgr5-GFP+ versus Lgr5-GFP− cells from AKVPSL and AKVPL tumours (n = 4 tumours per genotype). d, Tumour-engraftment efficiency of Lgr5+ and Lgr5− cells from AKVPL and AKVPSL tumours. e, Per cent tumour-initiating cells (TICs) in Lgr5-GFP+ and Lgr5-GFP− cell populations from AKVPL and AKVPSL tumours estimated with ELDA48 (n ≥ 5 grafts per dilution; error bars represent the 95% confidence interval). f, Per cent GFP+ cells in secondary tumours that originated from either 2,500 Lgr5-GFP+ or 2,500 Lgr5-GFP− AKVPSL tumour cells (n = 5 grafts per cell population). *P < 0.05; **P < 0.01; ***P < 0.001.
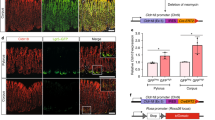
Extended Data Figure 4 Effects of Lgr5+ cell depletion on tumour growth.
a, b, Representative sections of n = 3 AKVPL tumours per condition stained for GFP (indicating Lgr5, green), cleaved caspase 3 (CC3, red) and nuclei (DAPI, blue). White arrows indicate yellow double positive cells that are detectable as early as 6 h after initiation of DT treatment, confirming specific depletion of the Lgr5-GFP+ cell population. Scale bar, 20 μm. c, d, Dosing schedule (c) and tumour growth (d) of mice with subcutaneous AKVPL allografts in response to intermittent DT treatment (n = 8 for the saline- or n = 7 for DT-treated group). e, f, Dosing schedule (e) and tumour growth (f) of mice with subcutaneous AKVPSL allografts in response to continuous DT treatment (n = 7 for each treatment group). n represents the number of independent animals.
Extended Data Figure 5 Gene expression profiling of DT-treated tumours.
a, Gene-expression analysis of bulk AKVPL tumour cells isolated by FACS at different time points after a single dose of either saline or DT (n = 4 tumours per time point and condition). b, Venn diagram depicting the number of differentially expressed genes for the samples in a at various time points after treatment. Number of genes with increasing or decreasing expression patterns are depicted. c, QuSAGE of indicated gene signatures in DT- versus saline-treated AKVPL tumours 24 h after treatment. d, Representative images of n = 3 AKVPL tumours per condition stained for BrdU (green), cleaved caspase 3 (CC3, red) and nuclei (DAPI), revealing dynamic changes in proliferation and cell death after DT treatment for indicated length of time. Scale bar, 50 μm. n represents the number of independent animals.
Extended Data Figure 6 Association of AKVPL tumours with intestinal cell signatures during DT treatment.
a, QuSAGE of intestinal cell gene signatures23 in DT- versus saline-treated AKVPL tumours 24 h after treatment. b, Representative images of n = 3 AKVPL tumours per condition stained with Alcian blue to evaluate the number of goblet cells after DT treatment for the indicated length of time (counter stained with nuclear fast red). Scale bar, 20 μm. n represents the number of independent animals.
Extended Data Figure 7 Morphology of orthotopic tumours and liver metastasis after DT treatment.
a, Quantification of bioluminescence signal in orthotopic AKVPSL tumours from saline- (n = 17) and DT-treated (n = 15) animals before liver evaluation. Data represented as mean (lines) and individual animals (circles). b, Representative images of haematoxylin and eosin staining of orthotopic AKVPSL tumours from n = 4 animals per condition after either saline or DT treatment. Boxed areas show invasive front, which is magnified on the right; note that DT treatment did not abrogate tumour invasiveness. c, Gross morphology of representative livers from n = 4 animals with orthotopic AKVPSL tumours treated with either saline (top) or DT (bottom). White arrows indicate macro-metastases that are absent from the DT-treated cohort. d, Related to Fig. 3j; histological evaluation of livers from saline- and DT-treated mice with an orthotopic AKVPSL tumour. Black arrows indicate metastatic foci that are absent from the DT-treated cohort. Representative images are shown from n = 6 per treatment group. Scale bars, 300 μm (b, d), 2 cm (c). n represents the number of independent animals.
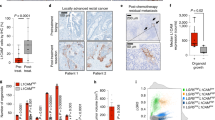
Extended Data Figure 8 Effect of Lgr5+ cell depletion in established liver metastasis.
a, Representative bioluminescent images of mice with established liver metastasis before either saline or DT treatment (n = 7 for saline and n = 8 for DT). Pre-tx, pre-treatment. b, Quantification of the bioluminescence signal in animals with established liver metastasis before DT or saline treatment (n = 7 for saline- and n = 8 for DT-treated group) c, Quantification of the bioluminescence signal in animals with established liver metastasis during (1 w DT) DT treatment. d, Related to Fig. 4d, e; livers from either saline- or DT-treated animals with established liver metastasis were collected one week after treatment. e, Number of RFP+ tumour cells in enzymatically dissociated livers of mice with established metastasis one week after treatment (FACS data represented as mean (lines) and individual animals (circles and squares); n = 6 per treatment group). f, Number of RFP+ tumour cells in enzymatically dissociated livers of mice with established metastasis two weeks after treatment (FACS data represented as mean (lines) and individual animals (circles); n = 7 animals per treatment group). *P < 0.05; **P < 0.01; ***P < 0.001; ns, not significant; n represents the number of independent animals.
Supplementary information
Supplementary Table 1
Off-target prediction for Trp53 and Smad4 sgRNAs. (XLSX 13 kb)
Supplementary Table 2
Time course analysis of transcriptional changes after DT treatment in vivo. (XLSX 126 kb)
Supplementary Table 3
Quantitative gene set analysis of expression changes in tumours after DT treatment. (XLSX 186 kb)
Supplementary Table 4
Quantitative gene set analysis of expression changes of selected gene sets. (XLSX 16 kb)
Source data
Rights and permissions
About this article
Cite this article
de Sousa e Melo, F., Kurtova, A., Harnoss, J. et al. A distinct role for Lgr5+ stem cells in primary and metastatic colon cancer. Nature 543, 676–680 (2017). https://doi.org/10.1038/nature21713
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature21713
This article is cited by
-
Crosstalk between colorectal CSCs and immune cells in tumorigenesis, and strategies for targeting colorectal CSCs
Experimental Hematology & Oncology (2024)
-
The dopamine transporter antagonist vanoxerine inhibits G9a and suppresses cancer stem cell functions in colon tumors
Nature Cancer (2024)
-
Targeted activation of ferroptosis in colorectal cancer via LGR4 targeting overcomes acquired drug resistance
Nature Cancer (2024)
-
Shaping immune landscape of colorectal cancer by cholesterol metabolites
EMBO Molecular Medicine (2024)
-
Circadian regulation of cancer stem cells and the tumor microenvironment during metastasis
Nature Cancer (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.