Abstract
When mammals fast, glucose homeostasis is achieved by triggering expression of gluconeogenic genes in response to glucagon and glucocorticoids. The pathways act synergistically to induce gluconeogenesis (glucose synthesis), although the underlying mechanism has not been determined1,2,3,4. Here we show that mice carrying a targeted disruption of the cyclic AMP (cAMP) response element binding (CREB) protein gene, or overexpressing a dominant-negative CREB inhibitor, exhibit fasting hypoglycaemia and reduced expression of gluconeogenic enzymes. CREB was found to induce expression of the gluconeogenic programme through the nuclear receptor coactivator PGC-1, which is shown here to be a direct target for CREB regulation in vivo. Overexpression of PGC-1 in CREB-deficient mice restored glucose homeostasis and rescued expression of gluconeogenic genes. In transient assays, PGC-1 potentiated glucocorticoid induction of the gene for phosphoenolpyruvate carboxykinase (PEPCK), the rate-limiting enzyme in gluconeogenesis. PGC-1 promotes cooperativity between cyclic AMP and glucocorticoid signalling pathways during hepatic gluconeogenesis. Fasting hyperglycaemia* is strongly correlated with type II diabetes, so our results suggest that the activation of PGC-1 by CREB in liver contributes importantly to the pathogenesis of this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Notes
* Correction: In the printed version, "hypoglycaemia" incorrectly appeared as "hyperglycaemia". A Correction will shortly be published.
References
Hanson, R. W. & Reshef, L. Regulation of phosphoenolpyruvate carboxykinase (GTP) gene expression. Annu. Rev. Biochem. 66, 581–611 (1997).
Lemaigre, F. P. & Rousseau, G. G. Transcriptional control of genes that regulate glycolysis and gluconeogenesis in adult liver. Biochem. J. 303, 1–14 (1994).
Angrand, P. O., Coffinier, C. & Weiss, M. C. Response of the phosphoenolpyruvate carboxykinase gene to glucocorticoids depends on the integrity of the cAMP pathway. Cell Growth Differ. 5, 957–966 (1994).
Imai, E., Miner, J. N., Mitchell, J. A., Yamamoto, K. R. & Granner, D. K. Glucocorticoid receptor–cAMP response element-binding protein interaction and the response of the phosphoenolpyruvate carboxykinase gene to glucocorticoids. J. Biol. Chem. 268, 5353–5356 (1993).
Gonzalez, G. A. & Montminy, M. R. Cyclic AMP stimulates somatostatin gene transcription by phosphorylation of CREB at Serine 133. Cell 59, 675–680 (1989).
Liu, J. S., Park, E. A., Gurney, A. L., Roesler, W. J. & Hanson, R. W. Cyclic AMP induction of phosphoenolpyruvate carboxykinase (GTP) gene transcription is mediated by multiple promoter elements. J. Biol. Chem. 266, 19095–19102 (1991).
Quinn, P. G. & Granner, D. K. Cyclic AMP-dependent protein kinase regulates transcription of the phosphoenolpyruvate carboxykinase gene but not binding of nuclear factors to the cyclic AMP regulatory element. Mol. Cell. Biol. 10, 3357–3364 (1990).
Liu, J. K., Bergman, Y. & Zaret, K. S. The mouse albumin promoter and a distal upstream site are simultaneously DNase I hypersensitive in liver chromatin and bind similar liver-abundant factors in vitro. Genes Dev. 2, 528–541 (1988).
Ahn, S. et al. A dominant negative inhibitor of CREB reveals that it is a general mediator stimulus-dependent transcription of c-fos. Mol. Cell. Biol. 18, 967–977 (1998).
Long, F., Schipani, E., Asahara, H., Kronenberg, H. & Montminy, M. The CREB family of activators is required for endochondral bone development. Development 128, 541–550 (2001).
Consoli, A. Role of liver in pathophysiology of NIDDM. Diabetes Care 15, 430–441 (1992).
Lee, G. H. et al. Abnormal splicing of the leptin receptor in diabetic mice. Nature 379, 632–635 (1996).
Chen, H. et al. Evidence that the diabetes gene encodes the leptin receptor: identification of a mutation in the leptin receptor gene in db/db mice. Cell 84, 491–495 (1996).
Wexler, I. D. et al. Molecular characterization of pyruvate carboxylase deficiency in two consanguineous families. Pediatr. Res. 43, 579–584 (1998).
Lei, K. J. et al. Glucose-6-phosphatase dependent substrate transport in the glycogen storage disease type-1a mouse. Nature Genet. 13, 203–209 (1996).
Schmoll, D. et al. Identification of a cAMP response element within the glucose-6-phosphatase hydrolytic subunit gene promoter which is involved in the transcriptional regulation by cAMP and glucocorticoids in H4IIE hepatoma cells. Biochem. J. 338, 457–463 (1999).
Jitrapakdee, S., Booker, G. W., Cassady, A. I. & Wallace, J. C. The rat pyruvate carboxylase gene structure. Alternate promoters generate multiple transcripts with the 5′-end heterogeneity. J. Biol. Chem. 272, 20522–20530 (1997).
Yoon, J. C. et al. Control of hepatic gluconeogenesis through the transcriptional coactivator PGC-1. Nature 413, 131–138 (2001).
Rudolph, D. et al. Impaired fetal T cell development and perinatal lethality in mice lacking the cAMP response element binding protein. Proc. Natl Acad. Sci. USA 95, 4481–4486 (1998).
Short, J. M., Wynshaw-Boris, A., Short, H. P. & Hanson, R. W. Characterization of the phosphoenolpyruvate carboxykinase (GTP) promoter-regulatory region. II. Identification of cAMP and glucocorticoid regulatory domains. J. Biol. Chem. 261, 9721–9726 (1986).
Imai, E. et al. Characterization of a complex glucocorticoid response unit in the phosphoenolpyruvate carboxykinase gene. Mol. Cell. Biol. 10, 4712–4719 (1990).
Friedman, J. E. et al. Phosphoenolpyruvate carboxykinase (GTP) gene transcription and hyperglycemia are regulated by glucocorticoids in genetically obese db/db transgenic mice. J. Biol. Chem. 272, 31475–31481 (1997).
Knutti, D., Kaul, A. & Kralli, A. A tissue-specific coactivator of steroid receptors, identified in a functional genetic screen. Mol. Cell. Biol. 20, 2411–2422 (2000).
He, T. et al. A simplified system for generating recombinant adenoviruses. Proc. Natl Acad. Sci. USA 95, 2509–2514 (1998).
Michael, L. F., Asahara, H., Shulman, A., Kraus, W. & Montminy, M. The phosphorylation status of a cyclic AMP-responsive activator is modulated via a chromatin-dependent mechanism. Mol. Cell. Biol. 20, 1596–1603 (2000).
Becker, T. et al. Use of recombinant adenovirus for metabolic engineering of mammalian cells. Methods Cell Biol. 43, 161–189 (1994).
Shang, Y., Hu, X., DiRenzo, J., Lazar, M. & Brown, M. Cofactor dynamics and sufficiency in estrogen receptor-regulated transcription. Cell 103, 843–852 (2000).
Puigserver, P. et al. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92, 829–839 (1998).
Nakajima, T. et al. RNA helicase A mediates association of CBP with RNA polymerase II. Cell 90, 1107–1112 (1997).
Acknowledgements
We thank J. Frangioni for help with adenovirus constructs, C. Arias for help with histology, and K. Suter for performing injections. This work was supported by grants from the National Institutes for Health to M.M. and from the DFG to S.H.
Author information
Authors and Affiliations
Corresponding author
Supplementary information

Figure S1
(GIF 7.38 KB)
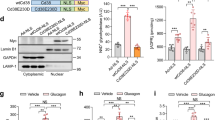
Plasma insulin levels in fed and fasted C57Bl6 (top) and db/db (bottom) mice injected with control (GFP) or A-CREB adenoviruses.

Figure S2
(GIF 4.36 KB)
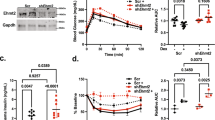
(left). Dose-response curve of insulin effects on PGC-1 mRNA levels in FAO hepatoma cells. Cells were treated with indicated concentrations of insulin for 4 hours, and PGC-1 mRNA levels were measured by quantitative PCR analysis.
(right). Time course analysis of insulin effects on PGC-1 mRNA levels in FAO hepatoma cells. Cells were treated with maximal (100nM) dose of insulin for various times, and PGC-1 mRNA levels were measured by quantitative PCR analysis. Average of two independent experiments ± SEM.

Figure S3
(GIF 3.91 KB)
PGC-1 has no effect on expression of A-CREB in mice co-infected with PGC-1 plus A-CREB adenoviruses. Western blot analysis of liver extracts from mice infected with GFP, A-CREB plus GFP, or A-CREB plus PGC-1 adenoviruses. Equal numbers of adenovirus particles were infected in each set. A-CREB protein was detected with anti-FLAG tag antiserum.
Rights and permissions
About this article
Cite this article
Herzig, S., Long, F., Jhala, U. et al. CREB regulates hepatic gluconeogenesis through the coactivator PGC-1. Nature 413, 179–183 (2001). https://doi.org/10.1038/35093131
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35093131
This article is cited by
-
Peroxisome proliferator-activated receptor gamma coactivator-1 (PGC-1) family in physiological and pathophysiological process and diseases
Signal Transduction and Targeted Therapy (2024)
-
Causality-enriched epigenetic age uncouples damage and adaptation
Nature Aging (2024)
-
Modulation of mitochondrial activity by sugarcane (Saccharum officinarum L.) top extract and its bioactive polyphenols: a comprehensive transcriptomics analysis in C2C12 myotubes and HepG2 hepatocytes
Natural Products and Bioprospecting (2024)
-
Local GHR roles in regulation of mitochondrial function through mitochondrial biogenesis during myoblast differentiation
Cell Communication and Signaling (2023)
-
Crosstalk between mitochondrial biogenesis and mitophagy to maintain mitochondrial homeostasis
Journal of Biomedical Science (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.