Abstract
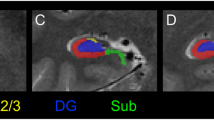
Accumulation of neurobiological knowledge points to neurodevelopmental origins for certain psychotic and mood disorders. Recent landmark postmortem reports implicate Reelin, a secretory glycoprotein responsible for normal lamination of brain, in the pathology of schizophrenia and bipolar disorders. We employed quantitative immunocytochemistry to measure levels of Reelin protein in various compartments of hippocampal formation in subjects diagnosed with schizophrenia, bipolar disorder and major depression compared to normal controls. Significant reductions were observed in Reelin-positive adjusted cell densities in the dentate molecular layer (ANOVA, P < 0.001), CA4 area (ANOVA, P < 0.001), total hippocampal area (ANOVA, P < 0.038) and in Reelin-positive cell counts in CA4 (ANOVA, P < 0.042) of schizophrenics vs controls. Adjusted Reelin-positive cell densities were also reduced in CA4 areas of subjects with bipolar disorder (ANOVA, P < 0.001) and nonsignificantly in those with major depression. CA4 areas were also significantly reduced in schizophrenic (ANOVA, P < 0.009) patients. No significant effects of confounding variables were found. The exception was that family history of psychiatric illness correlated strongly with Reelin reductions in several areas of hippocampus (CA4, adjusted cell density, F = 13.77, P = 0.001). We present new immunocytochemical evidence showing reductions in Reelin expression in hippocampus of subjects with schizophrenia, bipolar disorder and major depression and confirm recent reports documenting a similar deficit involving Reelin expression in brains of subjects with schizophrenia and bipolar disorder.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
D'Arcangelo G, Nakajima K, Miyata T, Ogawa M, Mikoshiba K, Curran T . Reelin is a secreted glycoprotein recognized by the CR-50 monoclonal antibody J Neurosci 1997; 17: 23–31
Bar I, Lambert de Rouvroit C, Royaux I, Krizman DB, Dernancourt C, Ruelle D et al. A YAC contig containing the reeler locus with preliminary characterization of candidate gene fragments Genomics 1995; 26: 543–549
D'Arcangelo G, Miao GG, Chen S-C, Soares HD, Morgan JI, Curran T . A protein related to extracellular matrix proteins deleted in the mouse mutant reeler Nature 1995; 374: 719–723
Klar A, Baldassare M, Jessell TM . F-Spondin: a gene expressed at high levels in the floor plate encodes a secreted protein that promotes neural cell adhesion and neurite extension Cell 1992; 69: 95–110
Vrucinic N, Chiquet-Ehrismann RR . Tenascin function and regulation of expression Symp Soc Exp Biol 1993; 47: 155–162
Goffinet AM . An early developmental defect in the cerebral cortex of the Reeler mouse Anat Embryol 1979; 157: 205–216
Ogawa M, Miyata T, Nakajima K, Yagyu K, Seike M, Ikenaka K et al. The reeler gene-associated antigen on Cajal–Retzius neurons is a crucial molecule for laminar organization of cortical neurons Neuron 1995; 14: 899–912
Borrell V, Del Rio JA, Alcantra S, Derer M, Martinez A, D'Arcangelo G et al. Reelin regulates entorhino-hippocampal connections J Neurosci 1999; 19: 1345–1358
Rodriguez MA, Pesold C, Liu WS, Kriho V, Guidotti A, Pappas GD et al. Colocalization of integrin receptors and Reelin in dendritic spine post-synaptic densities of adult non-human primate cortex Proc Natl Acad Sci 2000; 97: 3550–3555
Impagnatiello F, Guidotti A, Pesold C, Dwlivedi Y, Caruncho H, Pisu MG et al. A decrease of Reelin expression as a putative vulnerability factor in schizophrenia Proc Natl Acad Sci 1998; 95: 15718–15723
Smallheiser NR, Costa E, Guidotti A, Impagnatiello F, Auta J, Lacor P et al. Expression of Reelin in adult mammalian blood, liver, pituitary pars intermedia, and adrenal chromaffin cells Proc Natl Acad Sci 2000; 97: 1281–1286
Guidotti A, Auta J, Davis J, Dwivedi Y, Grayson D, Impagnatiello F et al. Decrease in Reelin and glutamic acid decarboxylase 67 (GAD67) expression in schizophrenia and bipolar disorder: a postmortem brain study Arch Gen Psychiatry 2000; (in press
Knable MB, Fuller Torrey E, Webster MJ, Bartko JJ . Multivariate analysis of prefrontal cortical data from the Stanley Foundation Neuropathology Consortium Brain Res Bull 2000; (in press
Akbarian S, Bunney Jr WE, Potkin SG, Wigal SB, Hagman JO, Sandman ET et al. Altered distribution of nicotinamide-adenine dinucleotide phosphate-diaphorase cells in frontal lobe of schizophrenics implies disturbances of cortical development Arch Gen Psychiatry 1993; 50: 169–177
Akbarian S, Vinuela A, Kim JJ, Potkin SG, Bunney WE, Jones EG . Distorted distribution of nicotinamide-adenine dinucleotide phosphate-diaphorase neurons in temporal lobe of schizophrenics implies anomalous cortical development Arch Gen Psychiatry 1993; 50: 178–187
Fuller Torrey E, Webster M, Knable M, Johnston N, Yolken RH . The Stanley Foundation brain collection and Neuropathology Consortium Schizophr Res 2000; 44: 151–155
Pesold C, Impagnatiello F, Pisu MG, Uzunov DP, Costa E, Guidotti A et al. Reelin is preferentially expressed in neurons synthesizing gamma-amniobutyric acid in cortex and hippocampus of adult rats Proc Natl Acad Sci 1998; 95: 3221–3226
De Bergeyck V, Nakajima K, Lambert de Roubroit C, Naerhuyzen B, Goffinet AM, Miyata T et al. A truncated Reelin protein is produced but not secreted in the ‘Orleans’ reeler mutation (Reln rl-crl) Mol Brain Res 1997; 50: 85–90
Weinberger DR . Cell biology of the hippocampal formation in schizophrenia Biol Psychiatry 1999; 45: 395–402
Nakajima K, Mikoshiba K, Miyata T, Kudo C, Ogawa M . Disruption of hippocampal development in vivo by CR-50 mAb against Reelin Proc Natl Acad Sci 1997; 94: 8196–8201
Selemon LD, Goldman-Rakic PS . The reduced neuropil hypothesis: a circuit based model of schizophrenia Biol Psychiatry 1999; 45: 17–25
Fatemi SH, Emamian ES, Kist D, Sidwell RW, Nakajima K, Akhter P et al. Defective corticogenesis and reduction in Reelin immunoreactivity in cortex and hippocampus of prenatally infected neonatal mice Mol Psychiatry 1999; 4: 145–154
Tueting P, Costa E, Dwivedi Y, Guidotti A, Impagnatiello F, Manev R et al. The phenotypic characteristics of heterozygous reeler mouse Neuroreport 1999; 10: 1329–1334
Suddath RL, Christison GW, Torrey EF, Casanova MF, Weinberger DR . Anatomical abnormalities in the brains of monozygotic twins discordant for schizophrenia N Engl J Med 1990; 322: 789–794
Velakoulis D, Pantelis C, McGorry PD, Dudgeon P, Brewer W, Cook M et al. Hippocampal volume in first episode psychosis and chronic schizophrenia Arch Gen Psychiatry 1999; 56: 133–140
Hoge EA, Friedman L, Schulz SC . Meta-analysis of brain size in bipolar disorder Schizophr Res 1999; 37: 177–181
Mohn AR, Gainetdinov RR, Caron MG, Koller BH . Mice with reduced NMDA receptor expression display behaviors related to schizophrenia Cell 1999; 98: 427–436
Trommsdorff M, Gotthardt M, Hiesberger T, Shelton J, Stockinger W, Nimpf J et al. Reeler/disabled-like disruption of neuronal migration in knockout mice lacking the VLDL receptor and ApoE receptor 2 Cell 1999; 97: 689–701
Anton ES, Kreidberg JA, Rakic P . Distinct functions of α3 and αν integrin receptors in neuronal migration and laminar organization of the cerebral cortex Neuron 1999; 22: 277–289
Lacor P, Grayson DR, Auta J, Sugaya I, Costa E, Guidotti A . Reelin secretion from glutamatergic neurons in culture is independent from neurotransmitter regulation Proc Natl Acad Sci 2000; 97: 3556–3561
Akbarian S, Kim JJ, Potkin SG, Hagman JO, Tafazzoli A, Bunney WE et al. Gene expression for glutamic acid decarboxylase is reduced without loss of neurons in prefrontal cortex of schizophrenics Arch Gen Psychiatry 1995; 52: 258–266
Volk DW, Austin MC, Pierri JN, Sampson AR, Lewis DA . Decreased glutamic acid decarboxylase 67 messenger RNA expression in a subset of prefrontal cortical gamma-aminobutyric acid neurons in subjects with schizophrenia Arch Gen Psychiatry 2000; 57: 237–245
Acknowledgements
Supported by Minnesota Medical Foundation and Stanley Foundation grants (SHF). We are grateful to Dr Andre M Goffinet for his generous gifts of G10 and 142 antibodies and to the Stanley Foundation Neuropathology Consortium Brain Bank for provision of brain samples courtesy of Drs LB Bigelow, J Cervenak, MM Herman, TN Hyde, JE Kleinman, M Knable, JD Paltan, RM Post, EF Torrey, MJ Webster and RH Yolken. We are indebted to Dr Joel Hektner for his astute statistical help with reanalysis of data. We appreciate the helpful comments provided by Drs E Costa, A Guidotti, C Pesold, SC Schulz and M Knable regarding this manuscript. We appreciate the secretarial assistance of Ms Janet Holland and Ms Heather Winstead, and careful review of data by Mr Joel Stary.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fatemi, S., Earle, J. & McMenomy, T. Reduction in Reelin immunoreactivity in hippocampus of subjects with schizophrenia, bipolar disorder and major depression. Mol Psychiatry 5, 654–663 (2000). https://doi.org/10.1038/sj.mp.4000783
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4000783
Keywords
This article is cited by
-
Formulating treatment of major psychiatric disorders: algorithm targets the dominantly affected brain cell-types
Discover Mental Health (2023)
-
Schizophrenia in the genetic era: a review from development history, clinical features and genomic research approaches to insights of susceptibility genes
Metabolic Brain Disease (2023)
-
Hippocampal volume and hippocampal neuron density, number and size in schizophrenia: a systematic review and meta-analysis of postmortem studies
Molecular Psychiatry (2021)
-
Fast-acting antidepressant-like effects of Reelin evaluated in the repeated-corticosterone chronic stress paradigm
Neuropsychopharmacology (2020)
-
The neuropathology of bipolar disorder: systematic review and meta-analysis
Molecular Psychiatry (2020)