Abstract
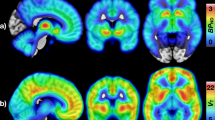
Positron emission tomography (PET) studies suggest opioidergic system dysfunction in morbid obesity, while evidence for the role of the dopaminergic system is less consistent. Whether opioid dysfunction represents a state or trait in obesity remains unresolved, but could be assessed in obese subjects undergoing weight loss. Here we measured brain μ-opioid receptor (MOR) and dopamine D2 receptor (D2R) availability in 16 morbidly obese women twice—before and 6 months after bariatric surgery—using PET with [11C]carfentanil and [11C]raclopride. Data were compared with those from 14 lean control subjects. Receptor-binding potentials (BPND) were compared between the groups and between the pre- and postoperative scans among the obese subjects. Brain MOR availability was initially lower among obese subjects, but weight loss (mean=26.1 kg, s.d.=7.6 kg) reversed this and resulted in ~23% higher MOR availability in the postoperative versus preoperative scan. Changes were observed in areas implicated in reward processing, including ventral striatum, insula, amygdala and thalamus (P's<0.005). Weight loss did not influence D2R availability in any brain region. Taken together, the endogenous opioid system plays an important role in the pathophysiology of human obesity. Because bariatric surgery and concomitant weight loss recover downregulated MOR availability, lowered MOR availability is associated with an obese phenotype and may mediate excessive energy uptake. Our results highlight that understanding the opioidergic contribution to overeating is critical for developing new treatments for obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Volkow ND, Wang GJ, Tomasi D, Baler RD . Obesity and addiction: neurobiological overlaps. Obes Rev 2013; 14: 2–18.
Baik JH . Dopamine signaling in reward-related behaviors. Front Neural Circuits 2013; 7: 152.
Ikemoto S, Bonci A . Neurocircuitry of drug reward. Neuropharmacology 2014; 76 (Pt B): 329–341.
Volkow ND, Wang GJ, Fowler JS, Logan J, Hitzemann R, Ding YS et al. Decreases in dopamine receptors but not in dopamine transporters in alcoholics. Alcohol Clin Exp Res 1996; 20: 1594–1598.
Volkow ND, Chang L, Wang GJ, Fowler JS, Ding YS, Sedler M et al. Low level of brain dopamine D2 receptors in methamphetamine abusers: association with metabolism in the orbitofrontal cortex. Am J Psychiatry 2001; 158: 2015–2021.
Martinez D, Saccone PA, Liu F, Slifstein M, Orlowska D, Grassetti A et al. Deficits in dopamine D(2) receptors and presynaptic dopamine in heroin dependence: commonalities and differences with other types of addiction. Biol Psychiatry 2012; 71: 192–198.
Wang GJ, Volkow ND, Logan J, Pappas NR, Wong CT, Zhu W et al. Brain dopamine and obesity. Lancet 2001; 357: 354–357.
de Weijer BA, van de Giessen E, van Amelsvoort TA, Boot E, Braak B, Janssen IM et al. Lower striatal dopamine D2/3 receptor availability in obese compared with non-obese subjects. EJNMMI Res 2011; 1: 37.
Haltia LT, Rinne JO, Merisaari H, Maguire RP, Savontaus E, Helin S et al. Effects of intravenous glucose on dopaminergic function in the human brain in vivo. Synapse 2007; 61: 748–756.
Steele KE, Prokopowicz GP, Schweitzer MA, Magunsuon TH, Lidor AO, Kuwabawa H et al. Alterations of central dopamine receptors before and after gastric bypass surgery. Obes Surg 2010; 20: 369–374.
Eisenstein SA, Antenor-Dorsey JA, Gredysa DM, Koller JM, Bihun EC, Ranck SA et al. A comparison of D2 receptor specific binding in obese and normal-weight individuals using PET with (N-[(11)C]methyl)benperidol. Synapse 2013; 67: 748–756.
Karlsson HK, Tuominen L, Tuulari JJ, Hirvonen J, Parkkola R, Helin S et al. Obesity is associated with decreased mu-opioid but unaltered dopamine D2 receptor availability in the brain. J Neurosci 2015; 35: 3959–3965.
Heinz A, Reimold M, Wrase J, Hermann D, Croissant B, Mundle G et al. Correlation of stable elevations in striatal mu-opioid receptor availability in detoxified alcoholic patients with alcohol craving: a positron emission tomography study using carbon 11-labeled carfentanil. Arch Gen Psychiatry 2005; 62: 57–64.
Gorelick DA, Kim YK, Bencherif B, Boyd SJ, Nelson R, Copersino M et al. Imaging brain mu-opioid receptors in abstinent cocaine users: time course and relation to cocaine craving. Biol Psychiatry 2005; 57: 1573–1582.
Weerts EM, Wand GS, Kuwabara H, Munro CA, Dannals RF, Hilton J et al. Positron emission tomography imaging of mu- and delta-opioid receptor binding in alcohol-dependent and healthy control subjects. Alcohol Clin Exp Res 2011; 35: 2162–2173.
Whistler JL . Examining the role of mu opioid receptor endocytosis in the beneficial and side-effects of prolonged opioid use: from a symposium on new concepts in mu-opioid pharmacology. Drug Alcohol Depend 2012; 121: 189–204.
Koch T, Hollt V . Role of receptor internalization in opioid tolerance and dependence. Pharmacol Ther 2008; 117: 199–206.
Gosnell BA, Levine AS . Reward systems and food intake: role of opioids. Int J Obes (Lond) 2009; 33: S54–S58.
Gloy VL, Briel M, Bhatt DL, Kashyap SR, Schauer PR, Mingrone G et al. Bariatric surgery versus non-surgical treatment for obesity: a systematic review and meta-analysis of randomised controlled trials. BMJ 2013; 347: f5934.
Dunn JP, Cowan RL, Volkow ND, Feurer ID, Li R, Williams DB et al. Decreased dopamine type 2 receptor availability after bariatric surgery: preliminary findings. Brain Res 2010; 1350: 123–130.
de Weijer BA, van de Giessen E, Janssen I, Berends FJ, van de Laar A, Ackermans MT et al. Striatal dopamine receptor binding in morbidly obese women before and after gastric bypass surgery and its relationship with insulin sensitivity. Diabetologia 2014; 57: 1078–1080.
Morinigo R, Moize V, Musri M, Lacy AM, Navarro S, Marin JL et al. Glucagon-like peptide-1, peptide YY, hunger, and satiety after gastric bypass surgery in morbidly obese subjects. J Clin Endocrinol Metab 2006; 91: 1735–1740.
Karamanakos SN, Vagenas K, Kalfarentzos F, Alexandrides TK . Weight loss, appetite suppression, and changes in fasting and postprandial ghrelin and peptide-YY levels after Roux-en-Y gastric bypass and sleeve gastrectomy: a prospective, double blind study. Ann Surg 2008; 247: 401–407.
Zubieta J, Greenwald MK, Lombardi U, Woods JH, Kilbourn MR, Jewett DM et al. Buprenorphine-induced changes in mu-opioid receptor availability in male heroin-dependent volunteers: a preliminary study. Neuropsychopharmacology 2000; 23: 326–334.
Frost JJ, Wagner HN Jr, Dannals RF, Ravert HT, Links JM, Wilson AA et al. Imaging opiate receptors in the human brain by positron tomography. J Comput Assist Tomogr 1985; 9: 231–236.
Farde L, Hall H, Ehrin E, Sedvall G . Quantitative analysis of D2 dopamine receptor binding in the living human brain by PET. Science 1986; 231: 258–261.
Gunn RN, Lammertsma AA, Hume SP, Cunningham VJ . Parametric imaging of ligand-receptor binding in PET using a simplified reference region model. NeuroImage 1997; 6: 279–287.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage 2002; 15: 273–289.
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K et al. A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. NeuroImage 2005; 25: 1325–1335.
Tellez LA, Medina S, Han WF, Ferreira JG, Licona-Limon P, Ren XY et al. A gut lipid messenger links excess dietary fat to dopamine deficiency. Science 2013; 341: 800–802.
Hardman CA, Herbert VM, Brunstrom JM, Munafo MR, Rogers PJ . Dopamine and food reward: effects of acute tyrosine/phenylalanine depletion on appetite. Physiol Behav 2012; 105: 1202–1207.
Small DM, Jones-Gotman M, Dagher A . Feeding-induced dopamine release in dorsal striatum correlates with meal pleasantness ratings in healthy human volunteers. NeuroImage 2003; 19: 1709–1715.
Wang GJ, Tomasi D, Convit A, Logan J, Wong CT, Shumay E et al. BMI modulates calorie-dependent dopamine changes in accumbens from glucose intake. PLoS One 2014; 9: e101585.
Michaelides M, Thanos PK, Kim R, Cho J, Ananth M, Wang GJ et al. PET imaging predicts future body weight and cocaine preference. NeuroImage 2012; 59: 1508–1513.
Volkow ND, Wise RA . How can drug addiction help us understand obesity? Nat Neurosci 2005; 8: 555–560.
Yeomans MR, Gray RW . Opioid peptides and the control of human ingestive behaviour. Neurosci Biobehav Rev 2002; 26: 713–728.
Tuominen L, Tuulari JJ, Karlsson HK, Hirvonen J, Helin S, Salminen P et al. Aberrant mesolimbic dopamine-opiate interaction in obesity. NeuroImage 2015; 122: 80–86.
Zubieta JK, Gorelick DA, Stauffer R, Ravert HT, Dannals RF, Frost JJ . Increased mu opioid receptor binding detected by PET in cocaine-dependent men is associated with cocaine craving. Nat Med 1996; 2: 1225–1229.
Williams TM, Daglish MR, Lingford-Hughes A, Taylor LG, Hammers A, Brooks DJ et al. Brain opioid receptor binding in early abstinence from opioid dependence: positron emission tomography study. Br J Psychiatry 2007; 191: 63–69.
Billes SK, Sinnayah P, Cowley MA . Naltrexone/bupropion for obesity: An investigational combination pharmacotherapy for weight loss. Pharmacol Res 2014; 84c: 1–11.
Caravaggio F, Raitsin S, Gerretsen P, Nakajima S, Wilson A, Graff-Guerrero A . Ventral striatum binding of a dopamine d2/3 receptor agonist but not antagonist predicts normal body mass index. Biol Psychiatry 2015; 77: 196–202.
Cooper TC, Simmons EB, Webb K, Burns JL, Kushner RF . Trends in weight regain following Roux-en-Y gastric bypass (RYGB) bariatric surgery. Obes Surg 2015; 25: 1474–1481.
Acknowledgements
The study was conducted within the Finnish Centre of Excellence in Cardiovascular and Metabolic Diseases supported by the Academy of Finland (grants #251125 and #121031), Sigrid Juselius Foundation, University of Turku, Turku University Hospital, and Åbo Akademi University. HKK was supported by personal grants from The Finnish Diabetes Research Foundation and The National Graduate School of Clinical Investigation. We thank study nurse Mia Koutu for her support in data collection. We thank Robert M Badeau, of Aura Professional English Consulting, for the language content editing and proofreading of this manuscript.
Author contributions
HKK acquired and analyzed PET data and wrote the manuscript, JJT acquired PET data and wrote the manuscript, LT analyzed PET data and wrote the manuscript, JH designed the experiments and wrote the manuscript, HH acquired PET data and edited the manuscript, RP screened MRI data, SH produced the radiotracers and edited the manuscript, PS recruited the study subjects, PN designed the experiments and wrote the manuscript, LN designed the experiments, supervised data analysis and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Karlsson, H., Tuulari, J., Tuominen, L. et al. Weight loss after bariatric surgery normalizes brain opioid receptors in morbid obesity. Mol Psychiatry 21, 1057–1062 (2016). https://doi.org/10.1038/mp.2015.153
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2015.153
This article is cited by
-
[11C]carfentanil PET imaging for studying the peripheral opioid system in vivo: effect of photoperiod on mu-opioid receptor availability in brown adipose tissue
European Journal of Nuclear Medicine and Molecular Imaging (2023)
-
Obesity risk is associated with altered cerebral glucose metabolism and decreased μ-opioid and CB1 receptor availability
International Journal of Obesity (2022)
-
Mesolimbic opioid-dopamine interaction is disrupted in obesity but recovered by weight loss following bariatric surgery
Translational Psychiatry (2021)
-
The Role of Positron Emission Tomography in Bariatric Surgery Research: a Review
Obesity Surgery (2021)
-
Sucrose intake lowers μ-opioid and dopamine D2/3 receptor availability in porcine brain
Scientific Reports (2019)