Abstract
The hallmark of atopic asthma is transient airways hyperresponsiveness (AHR) preceded by aeroallergen-induced Th-cell activation. This is preceded by upregulation of CD86 on resident airway dendritic cells (DCs) that normally lack competence in T-cell triggering. Moreover, AHR duration is controlled via T-regulatory (Treg) cells, which can attenuate CD86 upregulation on DC. We show that airway mucosal Treg/DC interaction represents an accessible therapeutic target for asthma control. Notably, baseline airway Treg activity in sensitized rats can be boosted by microbe-derived stimulation of the gut, resulting in enhanced capacity to control CD86 expression on airway DC triggered by aeroallergen and accelerated resolution of AHR.
Similar content being viewed by others
Introduction
The hallmark of atopic asthma is chronic low–moderate inflammation of the airway mucosa punctuated by transient episodes of severe inflammation and an ensuing state of airways hyperresponsiveness (AHR).1, 2 Cytokines produced by aeroallergen-specific Th helper 2 (Th2) cells are key components of such exacerbations,3, 4, 5 but the development of effective drugs to control their responses has not been achieved.
Recent experimental studies in our rat models suggest that triggering mucosal Th2 cells following aeroallergen inhalation is a two-stage process commencing with activation of initially quiescent airway mucosal dendritic cell (AMDC) through interactions with Th memory cells, leading to upregulation of CD86 expression and acquisition of potent T-cell-stimulatory activity by the AMDC in situ before translocation to regional airway draining lymph node (ADLN).3, 4 Subsequent interactions between these AMDCs and transiting Th memory cells result in a full-scale local Th-cell activation response,3, 4 the duration of which is determined by T-regulatory (Treg) activity.5 The importance of Treg in controlling AHR and associated allergic disease is strongly suggested by several recent studies (reviewed in Ryanna et al.6 and Xystrakis et al.7). We accordingly reasoned that therapeutic boosting of Treg activity in airway mucosal tissues of atopic asthmatics might represent a valid route to development of novel asthma treatments.
In addressing this possibility experimentally, we were drawn to recent studies indicating that probiotic treatment through the gastrointestinal mucosa can attenuate systemic as well as local immunoinflammatory functions (reviewed in Borchers et al.8). The concepts underpinning this emerging area of therapeutics draw on the principles of the common mucosal immune system, notably that lymphoid cells “programmed” in the gut subsequently home to other mucosal sites, to thus modulate local disease.9 Attempts to exploit this concept therapeutically are still in their infancy, nonetheless there is a growing list of reports suggesting significant probiotic effects on gastrointestinal10 and systemic diseases including eczema,11, 12 allergy,13 infection,14 and asthma.15, 16, 17, 18, 19 Of direct relevance to this study are data showing probiotic-induced increases in murine splenic Treg levels and ensuing resistance to development of AHR.17, 18, 19, 20 In addition, other studies have shown that bacterial agents can induce Treg in vitro17, 20, 21 and in vivo can confer protection against allergic airways inflammation.22
Although live probiotic organisms clearly have therapeutic potential, their biochemical complexity poses significant challenges relating to drug regulation/standardization. Following a slightly different approach, in this study we have tested the activity of a different class of microbial stimuli, which are currently being used in other disease settings, for capacity to modulate immune functions in the respiratory tract. The principal agent is the orally administered bacterial preparation OM-85BV, which has been in clinical use for many years and has shown some efficacy in adult COPD23, 24 and in children susceptible to repeated respiratory infections25, 26 and in experimental models of asthma.27 It is a multicomponent extract of respiratory-tract-derived bacteria comprising principally a mixture of acidic proteins, peptides, and amino acids, with minor components of detoxified lipopolysaccharides and lipoteichoic acids28 that express various immunomodulatory activities in model systems.29 We show that oral treatment of sensitized rats with OM-85BV markedly boosts baseline levels of CD4+CD25+Foxp3+ Tregs in airway mucosal tissues, and that animals with boosted mucosal Treg defenses show markedly attenuated airways inflammation, AHR, and associated AMDC responses following aeroallergen challenge. We have additionally used a single triacylated derivative from the extract (OM-174, a triacylated lipid A purified from the lysates of Gram-negative bacteria, Escherichia coli),30, 31 which recapitulates the key activities of the mixed OM-85 extract.
Results
OM-85BV pretreatment reduces allergic airways inflammation in bronchoalveolar lavage
Typically, a single ovalbumin (OVA) aerosol challenge of OVA-sensitized animals induces inflammation, which in bronchoalveolar lavage (BAL) fluid manifests as an increase in both the total number of cells and in the number of eosinophils, neutrophils, and lymphocytes. Figure 1a–d illustrates cell recoveries from individual baseline control rats and from sensitized rats, untreated vs. OM-85BV treated, exposed to a single OVA aerosol. OVA aerosol exposure of untreated sensitized rats induced a significant increase in total BAL, eosinophil, neutrophil, and lymphocyte cell numbers relative to baseline control rats (Figure 1a–d). The untreated group, as defined in Methods was made up of nil- and saline-exposed animals, which were not significantly different from each other (Supplementary Figure S1). However, pretreatment of sensitized animals with OM-85BV resulted in a dampened inflammatory response to OVA aerosol with reduced cell numbers, particularly eosinophils but also reduced neutrophils and lymphocytes (Figure 1a–d). In contrast to the reduced BAL inflammatory response following aeroallergen challenge, pretreatment of sensitized animals with OM-85BV did not modify serum IgE titers (Figure 1e).
Modulation of peripheral lung inflammatory response to aeroallergen challenge by OM-85BV pretreatment of sensitized animals. Results are shown here as individual data points or as box plots (mean, interquartile range, and minimum/maximum values). (a) Total BALF cells in baseline control rats or sensitized rats repeatedly treated with OM-85BV or untreated as per description in Methods. BAL was performed 24 h after a single aerosol challenge. (b) Total eosinophils in BALF as per (a). (c) Total neutrophils in BALF as per (a). (d) Total lymphocytes in BALF as per (a). (e) Serum OVA-specific IgE (log2 PCA titer; n=6 animals per group). Statistical significance was determined using Mann–Whitney test.
In situ activation of AMDC is inhibited by OM-85BV pretreatment
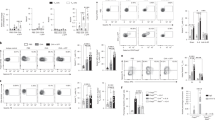
We next sought to determine if the responses of other cell types associated with allergic airways inflammation, specifically AMDC, were modified by pretreatment with OM-85BV vs. untreated groups of animals. Figure 2a shows representative fluorescence-activated cell sorting (FACS) plots of tracheal digests illustrating the gating strategy used to identify major histocompatibility complex (MHC) class II+ AMDC expressing CD4 and CD86 following repeated daily aerosol exposures. As shown in Figure 2b, the proportion of CD4+ MHC class II+ AMDCs in tracheal digests started increasing within 2 h following OVA aerosol exposure compared with baseline control rats, and by the third exposure this population had increased 2.5- to 3-fold above baseline. With continued daily exposure however, consistent with our previous findings,5 the numbers of AMDC declined, returning to baseline levels by day 6. As also shown in Figure 2b, treatment of sensitized animals with OM-85BV before challenge did not alter the kinetics of the transient CD4+ AMDC response and no significant differences were detected between the treatment groups at any time point. Central to the development of allergic airways inflammation is the in situ activation of AMDC,32 and in accordance with our earlier findings,5 data in Figure 2a, c show that 2 h after a single aerosol challenge of sensitized rats, the surface expression of CD86 is markedly upregulated on CD4+ AMDC. This activation of CD4+ AMDC is maintained over a 3-day exposure period (Figure 2c), and analogous to the overall pattern observed with AMDC numbers, the CD86 response was transient and resolved spontaneously by day 6 of a daily aeroallergen exposure regimen. OM-85BV treatment promoted acceleration of this normal resolution process, such that by day 3 CD86 expression was no longer different from resting levels in baseline control animals (Figure 2a,c). These findings suggest that the effects of OM-85BV treatment appear to be directed toward regulation of the CD86 response of AMDC, as opposed to their trafficking, and likely involve local mechanisms that modulate activation of AMDC in the OM-85BV-treated rats. We also determined the surface expression of CD172α, which has recently been described to be involved in trafficking of airway DC during allergic airways inflammation,33 on AMDC and ADLN populations and found that this was unaffected by treatment with OM-85BV. It is noteworthy that OM-85BV-induced accelerated resolution of the CD86 response on AMDC during ongoing aeroallergen exposure is restricted to the populations within the airway mucosa, i.e., CD86 expression on CD4+ MHC class II+ DC in ADLN remained high on day 3 (representative FACS plots in Figure 2d) whereas it essentially returned to baseline in upstream mucosal tissues (Figure 2c).
Modulation of airway mucosal dendritic cell (DC) responses to aeroallergen by OM-85BV. (a) Representative fluorescence-activated cell sorting (FACS) plots of surface expression of major histocompatibility complex (MHC) class II and CD4 or CD86 on tracheal digest cells (macrophage and B-cell depleted) from sensitized untreated (as described in Methods) vs. OM-85BV-pretreated rats after aerosol exposure as indicated. Data are representative of three independent experiments for each series using n=4 animals per group. (b) MHC class II+ CD4+ AMDC in tracheal digest preparations, in baseline control rats and in sensitized aerosol-exposed animals untreated vs. with OM-85BV treatment. (c) MHC class II+ CD4+ AMDC expressing surface CD86 in groups as per panel (b). (d) Representative FACS plots illustrating surface expression of CD86 on MHC class II+ CD4+ ADLN DC, gated as shown, prepared from sensitized/aerosol-challenged animals as indicated. Mean data derived from three independent experiments with n=4 animals per group, or two independent experiments for OVA × 6 aerosol exposures. Tracheal digests were prepared 2 h following the final aerosol exposure. Statistical significance was determined using Student's t-test.
OM-85BV pretreatment induces selective recruitment of Treg to airways
Next we determined the effect of OM-85BV treatment on the T-cell compartment during the allergic airways inflammatory response. The total TCR+ population in the tracheal mucosa of sensitized and OVA-aerosol-exposed rats was found to rapidly expand compared with baseline control rats (Figure 3a), peaking at day 3. Reduction in total T-cell numbers commenced thereafter despite continuing exposure, and OM-85BV treatment did not significantly alter this. Expression of CD25 on tracheal CD4+ T cells increased markedly following aerosol exposure but did not vary between the treatment groups (data not shown; note in Supplementary Figure S2 that expression levels of CD25 on Foxp3+CD4+ cells between untreated and OM-85BV-treated groups is similar). However, significant differences were observed with CD4+CD25+Foxp3+ cells. In particular, in sensitized animals given OM-85BV pretreatment, the total number of Foxp3+ cells in tracheal digests was already significantly elevated relative to sensitized untreated groups before aerosol challenge, as shown in the representative FACS plots (Figure 3b). This effect can also be observed in baseline control rats that also show a significant increase in basal numbers of Foxp3+ cells in airway mucosal tissue after OM-85BV feeding (Figure 3c). Following aerosol challenge of sensitized animals a sharp rise was observed in tracheal CD4+Foxp3+ numbers particularly in the OM-85BV-treated group (Figure 3d). Numbers remained elevated in the OM-85BV treatment group until at least day 3 into the exposure regimen, and this gap eventually narrowed by day 6 exposure, as CD4+CD25+Foxp3+ cells progressively accumulated in the mucosae of treatment control rats, as expected, in response to the continuing daily challenge (Figure 3e). We5 and others34 have shown that Treg can function to downregulate DC activation, and the increased number of Foxp3 cells within the airway mucosae of OM-85BV-treated animals at this earlier time point therefore potentially explains the reduced time frame over which their AMDCs remain activated during exposure (Figure 2c). This accumulation of Treg was not attributable to an overall increase in the proportion of proliferating Foxp3+ cells in the airways as although the number of Treg in the airways expressing the proliferation antigen Ki-67 increases in response to aerosol challenge, we did not detect differences between treatment groups at any stage of the time course (Supplementary Figure S2). In addition, the effects of OM-85BV pretreatment in the airway mucosa were reflected by corresponding changes in baseline numbers of Foxp3+ cells in ADLN, however following aerosol exposure of sensitized animals any such effects of OM-85BV treatment were no longer significant (Figure 3f).
Modulation of airway mucosal T-cell responses by OM-85BV. (a) TCRαβ+ cells in tracheal digests of baseline control rats and from sensitized untreated (as described in Methods) vs. OM-85BV-pretreated rats after aerosol exposure as indicated. (b) Representative plots showing side scatter (SSC) vs. Foxp3 staining in tracheal digest cells of baseline control animals, untreated vs. OM-85BV treatment. (c) Foxp3+ cells in tracheal digests of baseline control animals untreated or following OM-85BV treatment, normalized against cell numbers in baseline control group. (d) Representative plots illustrating Foxp3 and CD4 coexpression in tracheal digest cells from sensitized/challenged (× 3) animals untreated vs. OM-85BV treatment. (e) Percent Foxp3+ cells in tracheal digests in baseline control animals and in sensitized/challenged animals untreated vs. OM-85BV treatment; data shown are representative and derived from paired observations from three independent experiments. (f) Foxp3+ cells in airway draining lymph node (ADLN) digests from animals as per panel (e). Data are derived from three independent experiments using pools of n=4 animals per group, except 6 × aerosol exposures, which is representative of two data sets. All tissue digests prepared 2 h following the final aerosol exposure. Numerical values shown in representative plots indicate percent within selected gate. *P<0.05 by paired Student's t-test.
AHR is attenuated by pretreatment with OM-85BV
The findings mentioned above show that pretreatment with OM-85BV can significantly attenuate aspects of the cellular response in the airways of sensitized animals triggered by aeroallergen exposure. We next questioned whether these effects have relevance to aeroallergen-induced changes in lung function. We thus compared the development of AHR in sensitized OM-85BV pretreated vs. untreated animals following OVA aerosol challenge. In sensitized rats of all treatment groups mean reciprocal EC200 responses to methacholine challenge increased equivalently following a single OVA aerosol, indicating onset of AHR. Responsiveness increased further in the untreated group by the third aerosol exposure and had returned to baseline levels as expected by exposure day 6 in accordance with the capacity to self-regulate the response. Importantly, resolution of AHR was accelerated in the OM-85BV pretreatment group, and had already returned to baseline equivalence by exposure day 3 (Figure 4). The suppressive capacity of sorted CD4+CD25+ cells from ADLN was examined in vitro as reported previously,5 however no marked difference was detected in the ability to suppress OVA-specific T-cell proliferation on a per cell basis between OM-85BV-treated vs. -untreated groups (Supplementary Figure S3a). Levels of Foxp3 expression, as measured by qRT-pcr (see Strickland et al.5) or assessed by flow cytometry, were also comparable between different treatment groups (Supplementary Figure S3b).
Airways hyperresponsiveness (AHR) in OM-85BV-treated animals. The EC200Raw is presented as the reciprocal normalized to baseline control rats, thus a higher value represents increased AHR. Statistical significance was determined using one-way analysis of variance.
OM-85BV treatment of naive animals also alters baseline cellularity within gut tissue, blood, and BAL
Cell populations resident on the airway lumenal surface were sampled by BAL in untreated baseline control rats vs. following treatment with OM-85BV. As illustrated in Figure 5, OM-85BV treatment reduced total cell numbers (Figure 5a), in particular alveolar macrophages (Figure 5b). A reciprocal increase in total Foxp3+ cells in BAL of OM-85BV treated rats was detected and is shown relative to untreated baseline control rats (Figure 5c). Similarly, OM85-BV treatment of baseline control rats results in elevated numbers of total mononuclear cells per ml of blood (Figure 5d) and Foxp3 cells (Figure 5e) relative to untreated groups. The same trend was also observed in gut lamina propria following OM-85BV treatment (Figure 5f). Conversely, we detected reduced numbers of Foxp3 cells in mesenteric lymph node of OM-85BV-treated baseline control rats vs. untreated groups, despite similar total CD4 cell number (Figure 5g, h).
Modulation of cell profiles in tissues of baseline control animals by OM-85BV treatment. (a) Total BALF cells in individual baseline control rats untreated vs. with OM-85BV treatment as per protocol in Methods. BAL was performed 24 h after the last treatment. (b) Total macrophages in BALF as per (a). (c) Percent Foxp3+ cells as determined by intracellular staining in BALF, data shown are paired observations normalized against untreated baseline control animals and show two independent experiments. (d) Total cells per ml isolated from peripheral blood of untreated baseline control vs. OM-85BV-treated rats, data shown are paired observations. (e) Percent Foxp3+ cells in isolated PBMC, data shown as per (c). (f) Percent Foxp3+ cells in gut LP, data shown as per (c). (g) Percent Foxp3+ cells in mesenteric lymph node, data shown as per (c) for three independent experiments. (h) Percent total CD4+ cells in mesenteric lymph node of OM-85 BV-treated rats relative to untreated baseline control rats. Statistical significance was determined using Mann-Whitney test.
A single bacterial-derived molecular species OM-174 mimics the effects of OM-85BV
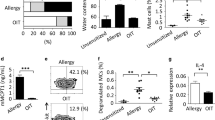
We also examined the extent to which the immunomodulatory effects of the mixed bacterial lysate preparation OM-85BV could be replicated by a single molecular species derived from the lysate (OM-174). Similar to that observed for OM-85BV treatment above, OM-174 pretreatment of baseline control rats resulted in significantly reduced total and macrophage cell counts in BAL (Figure 6a,b) compared with untreated groups of animals. In parallel, the basal number of tracheal Foxp3+ cells was significantly elevated in OM-174-treated vs. -untreated baseline control rats (Figure 6c). The cellular inflammatory response to OVA challenge of sensitized rats was also significantly dampened in OM-174 pretreated rats (Figure 6d), in particular the eosinophil component (Figure 6e) and also the neutrophil and lymphocyte responses (Figure 6f, g). Consistent with the pattern observed for OM-85BV (Figure 3), pretreatment of animals with OM-174 expanded the baseline Foxp3+ population in the tracheal mucosa and this remained elevated during the crucial early stages of the response to aeroallergen (Figure 6h). Treatment with OM-174 did not alter the IgE response (Supplementary Figure S4).
Modulation of respiratory Treg populations by OM-174 treatment. (a) Cell numbers in BALF of individual untreated baseline control rats vs. OM-174 treatment. (b) Macrophage numbers in BALF as per (a). (c) Foxp3+ cells in tracheal digests as per (a). (d) Total BALF cells in individual baseline control rats or sensitized animals untreated or repeatedly treated with OM-174 as per protocol in Methods. BAL was performed 24 h after a single aerosol challenge. (e) Total eosinophils in BALF as determined by differential cell counting as per (d). (f) Total neutrophils in BALF as per (d). (g) Total lymphocytes as per (d). Statistical significance was determined for a, b and d–g using Mann–Whitney test. (h) Foxp3+ cells in tracheal digest cells, as determined by intracellular staining; data are paired observations from representative experiments showing baseline control animals and sensitized animals untreated or treated with OM-174 and exposed to 1, 3, or 6 consecutive daily aerosols. Indicated statistics were performed on paired observations from three independent experiments, n=4 animals pooled per group. *P<0.05, by paired t-test.
Discussion
The development of persistent atopic asthma is a multifactorial process involving a range of innate and adaptive immune cell populations. A key component of pathogenesis involves cyclical waves of Th2 memory cell activation and accompanying cytokine release in the airway mucosa,35, 36 which is associated with the development of AHR and the chronic wheezing manifestations characteristic of the atopic asthmatic phenotype.2, 35, 36, 37 We have described how local activation of sensitized Th2 memory cells in airway tissue is normally constrained by the immature functional status of resident AMDC.32 Moreover, using an experimental model that mimics the essential features of human asthma,3, 5 we have also shown that the triggering step in this response involves cognate interaction between T memory cells and allergen-bearing AMDCs leading to transient upregulation of CD86 expression and antigen-presenting cell activity.3 This process and the ensuing AHR, which develops as a result of local cytokine release, are terminated by Treg cells that accumulate in the inflamed airway mucosa during continuous allergen exposure.5
In asthma in humans, persistence of AHR following exacerbation can be up to several weeks and the length of this period largely determines the severity of short- and long-term clinical sequelae. Although it is not possible to stepwise track Tregs in airway tissues in humans with a comparable degree of precision, there is indirect evidence that deficiencies in the number of Tregs underlie susceptibility to both the Th2 sensitization/reactivation and AHR components of asthma in humans.38, 39, 40 This conclusion is consistent with current perceptions of the important role of Tregs in maintenance of immunological homeostasis throughout the body.38, 41, 42, 43 Consequently, there is growing interest in Tregs as potential targets for development of anti-inflammatory therapeutics, in particular the general notion that “boosting” Treg activity at sites of ongoing inflammatory disease may control symptoms.
The tissue in which this appears to show most immediate relevance is the gastrointestinal tract (GIT). Proof-of-concept evidence is already available indicating that adoptive transfer of Tregs that home to GIT tissues can ameliorate local autoimmune inflammation.44 Significant progress has been made in elucidation of the underlying mechanisms of mucosal Treg generation, particularly the role of TGF-β and retinoic acid-dependent CD103+ DCs that appear uniquely active in GIT tissue in Treg generation.45, 46, 47 Moreover, there is evidence to suggest that the local generation of Tregs can be stimulated by exposure to lumenal antigens, particularly from microbial flora.44, 48 Accordingly, this microenvironment would appear readily accessible for therapeutic boosting of local Treg activity. This represents an area of increasingly active research, particularly involving the use of controlled microbial exposure through oral administration of probiotics to treat gastrointestinal disorders.
However the applicability of the “Treg-boosting” paradigm to inflammatory diseases in other tissues such as the asthmatic lung has not been explored in detail and appears considerably more challenging. In particular, the fragility of airway mucosal surfaces (relative to the GIT mucosa) and the hyperresponsiveness of these tissues to irritant stimuli in asthma mitigate against direct local stimulation with Treg-inductive agents, which suggests that a more viable approach may be by indirect systemic stimulation. This study addresses this possibility and uses as its starting point recent observations suggesting that Tregs that are stimulated by the GIT can influence immunoinflammatory processes in distal tissues. Of particular relevance are studies showing that Tregs stimulated in the GIT can downregulate AHR in murine asthma models.17, 20, 49
We show here that oral pretreatment of sensitized rats with the bacteria-derived immunostimulant OM-85BV strikingly accelerates the resolution of AHR that is triggered by aeroallergen exposure. Moreover, the kinetics of the AHR response in treated animals closely mirrors associated CD86 expression on AMDC, which we have shown to be controlled by Tregs.5 Furthermore, the principal feature distinguishing OM-85BV-treated rats from controls was their markedly increased (2 × ) baseline numbers of airway mucosal Tregs. Current evidence suggests that efficient expression of Treg activity requires that Treg numbers exceed a critical “threshold” in the microenvironment of their targets to achieve control,6, 7, 38, 45 and we surmise that the enhanced baseline activity in the airway mucosa of OM-85BV-treated sensitized rats enables them to reach this threshold faster following aeroallergen challenge. It is pertinent to note that in a recently reported probiotic model attenuation of aerosol-induced AHR in sensitized/treated mice was associated with an expanded splenic Treg population,17 and in the present model we also noted increased numbers of Tregs in the ADLN of OM-85BV-treated rats. However this is the first evidence that Treg stimulation through the GIT results in increased Treg trafficking to resting peripheral mucosal tissues, potentially providing an endogenous mechanism for prevention of inflammation rather than a purely reactive mechanism that relies on local Treg triggering after tissue damage is initiated. The fact that in nonsensitized rats, immunostimulation by the gut can give rise to increased populations of Treg cells in other mucosal sites, such as the respiratory mucosa, makes the use of this type of model biologically relevant for studies on control of human disease. Also noteworthy is the finding that in immunostimulant-treated rats, resistance to eosinophilia/AHR after aerosol challenge was independent of changes in specific IgE titers, which is reminiscent of findings in successful immunotherapy in humans and reinforces the view that a significant component of aeroallergen-induced airways inflammation is independent of IgE-mediated mast cell responses.
Various issues require resolution to achieve a more comprehensive understanding of the underlying mechanisms operative in this system. For example it is unclear as to precisely where in the GIT these Tregs are programmed in response to oral dosing, and clarification of this question may be a prerequisite for development of standardizable therapeutic strategies targeting this pathway. Our initial observations reported here indicate increases in the range of 30–40% in CD4+CD25+Foxp3+ numbers in the upper intestinal mucosa and blood after OM-85BV feeding, in the absence of corresponding increases in the mesenteric lymph node; this may indicate mobilization of locally activated Tregs directly into the peripheral circulation, but more detailed follow-up studies are required. Of equivalent importance is the nature of the receptor–ligand interactions underlying generation of Tregs that can traffic to distal tissues following GIT stimulation. A wide range of GIT stimuli have been associated with alterations in inflammatory (in particular atopy-related) processes in other tissues beyond the present bacterial extracts and conventional probiotics, including helminths,50 CPG,51 Cryptosporidium-related parasites,52 and various enteric pathogens associated with fecal contamination of water supplies.53 The breadth of this range suggests that multiple microbial pattern recognition receptors are capable of mediating what may be an essentially generic process through which the GIT mucosal immune system titrates local (and indirectly systemic) Treg activity in response to the intensity of incoming antigenic stimulation from the microbial environment.
In summary, our current findings, taken together with those from other recently reported models, suggest that it may be possible to harness this readily accessible mechanism for systemic Treg boosting for therapeutic purposes, in this case in the context of reducing susceptibility to aeroallergen-induced persistent AHR.
Methods
Animals, treatments, and allergen exposures. Inbred specified-pathogen-free PVG rats of both sexes were used in these studies, which were approved by our institutional animal ethics committee. Sensitization to OVA was performed by IP inoculation of 100 μg OVA in 200 μl aluminum hydroxide. Treatment with OM-85BV or OM-174 was for 7 consecutive days from day 10 after sensitization. Rats were used for aerosol challenge experiments on the day following final treatment. Aerosol challenge was carried out over a 60 min period (Tri R Airborne Infection Apparatus; Tri-R Instruments, Rockville Centre, NY) using OVA (Grade V; Sigma-Aldrich, NSW, Australia) at 1% in phosphate-buffered saline.4, 5 Responses to OVA aerosol exposure in sensitized animals treated with saline (as placebo) for 7 consecutive days did not differ from those left untreated, and data from untreated sensitized controls are presented throughout. Where shown, baseline controls indicate nonsensitized/non-treated/non-aerosol-exposed rats. OVA aerosol challenge of nonsensitized animals did not elicit detectable cellular responses.
Media and reagents. Tissue culture medium and isolation reagents including monoclonal antibodies and immunostaining reagents are as previously reported.3, 4, 5 For intracellular staining of Foxp3, an anti-mouse/rat Foxp3FLR staining kit from eBioscience (San Diego, CA) was used. Ki-67 to detect cell proliferation antigen was from BD Pharmingen (Perth, Western Australia) and was used in combination with Foxp3. Data were acquired on an LSRII flow cytometer (BD Biosciences, Eugene, OR) and analyzed using FlowJo software (version 4.6.1; Tree Star, Ashland, OR).
OM-85 is a mixed bacterial lyophylizate, which has been used various clinical settings to modulate airways inflammation associated mainly with infection,24, 27, 31, 32 and was used here at a dose rate of 400 mg kg−1 body weight per day. OM-174 is a triacylated lipid A purified from lysate of E. coli.31 OM-174 was administered orally at a dose of 1 mg per day (10 mg kg−1). Both extracts were delivered through the oral route in liquid form. Access to food and water during the treatment period was ad libitum. All experiments for each of the bacterial extracts were performed using a single batch, supplied by OM Pharma (Geneva, Switzerland).
Cell preparations. Single cell suspensions of LNs and trachea digests were prepared and depleted of macrophages and B cells as described previously.4, 5 We adopted the same protocol to obtain digested cell preparations from flushed sections of dissected gut lamina propria. Blood mononuclear cells were collected over Lymphoprep density gradient (density 1.077; Axis, Oslo, Norway). Purification of cell populations was by standard Dynabead depletion method, MACS bead enrichment, high-speed cell sorting, or combinations of these techniques.7, 8 BALF cells were obtained by standard methods and stained using Diff-Quik (Lab Aids, Narrabeen, NSW, Australia) for differential cell counting.
Lung function and methacholine hyperresponsiveness. The techniques used were as described previously.5 Briefly, respiratory impedance (Zrs) was measured by forced oscillation between 0.5. and 20 Hz using a computer-controlled piston ventilator (flexiventâ; Scireq, Montreal, Quebec, Canada). The constant-phase model was fitted to give estimates of airway resistance (Raw). Following determination of baseline lung function, methacholine challenges were performed by delivery of aerosols (2 min) of saline (control) and methacholine (0.1, 0.3, 1.0, 3.0, 10.0, and 30.0 mg ml−1) during tidal ventilation. Five measurements of Zrs were carried out after each dose and peak responses were reported. A concentration–response curve was constructed and the concentration associated with a doubling of RAW (EC200Raw) was calculated.
Statistical analysis. The experiments were carried out using groups of animals (n as indicated) or pooled tissue using four animals per group, and replicated independently on three occasions. As indicated, two-tailed unpaired Student's t-test was performed to determine statistical significance for difference between mean data or paired t-test was used for experiments between different treatment groups performed simultaneously, a=0.05, b=0.8. Mann–Whitney test was used for nonparametric data, 95% confidence level.
References
Busse, W.W., Banks-Schlegel, S. & Wenzel, S.E. Pathophysiology of severe asthma. J. Allergy Clin. Immunol. 106, 1033–1042 (2000).
Holgate, S.T. Pathogenesis of asthma. Clin. Exp. Allergy 38, 872–897 (2008).
Huh, J.C. et al. Bidirectional interactions between antigen-bearing respiratory tract dendritic cells (DCs) and T cells precede the late phase reaction in experimental asthma: DC activation occurs in the airway mucosa but not in the lung parenchyma. J. Exp. Med. 198, 19–30 (2003).
Jahnsen, F.L. et al. Accelerated antigen sampling and transport by airway mucosal dendritic cells following inhalation of a bacterial stimulus. J. Immunol. 177, 5861–5867 (2006).
Strickland, D.H. et al. Reversal of airway hyperresponsiveness by induction of airway mucosal CD4+CD25+ regulatory T cells. J. Exp. Med. 203, 2649–2660 (2006).
Ryanna, K., Stratigou, V., Safinia, N. & Hawrylowicz, C.M. Regulatory T cells in bronchial asthma. Allergy 64, 335–347 (2009).
Xystrakis, E., Urry, Z. & Hawrylowicz, C.M. Regulatory T cell therapy as individualized medicine for asthma and allergy. Curr. Opin. Allergy Clin. Immunol. 7, 535–541 (2007).
Borchers, A.T., Selmi, C., Meyers, F.J., Keen, C.L. & Gershwin, M.E. Probiotics and immunity. J. Gastroenterol. 44, 26–46 (2009).
Mestecky, J. The common mucosal immune system and current strategies for induction of immune responses in external secretions. J. Clin. Immunol. 7, 265–276 (1987).
Di Giacinto, C., Marinaro, M., Sanchez, M., Strober, W. & Boirivant, M. Probiotics ameliorate recurrent Th1-mediated murine colitis by inducing IL-10 and IL-10-dependent TGF-beta-bearing regulatory cells. J. Immunol. 174, 3237–3246 (2005).
Kalliomaki, M. et al. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. [see comment]. Lancet 357, 1076–1079 (2001).
Kalliomaki, M., Salminen, S., Poussa, T., Arvilommi, H. & Isolauri, E. Probiotics and prevention of atopic disease: 4-year follow-up of a randomised placebo-controlled trial. [see comment]. Lancet 361, 1869–1871 (2003).
Pohjavuori, E. et al. Lactobacillus GG effect in increasing IFN-gamma production in infants with cow's milk allergy. [see comment]. J. Allergy Clin. Immunol. 114, 131–136 (2004).
de Vrese, M. et al. Probiotic bacteria reduced duration and severity but not the incidence of common cold episodes in a double blind, randomized, controlled trial. Vaccine 24, 6670–6674 (2006).
Blumer, N. et al. Perinatal maternal application of Lactobacillus rhamnosus GG suppresses allergic airway inflammation in mouse offspring. Clin. Exp. Allergy 37, 348–357 (2007).
Feleszko, W. et al. Probiotic-induced suppression of allergic sensitization and airway inflammation is associated with an increase of T regulatory-dependent mechanisms in a murine model of asthma. Clin. Exp. Allergy 37, 498–505 (2007).
Karimi, K., Inman, M.D., Bienenstock, J. & Forsythe, P. Lactobacillus reuteri-induced regulatory T cells protect against an allergic airway response in mice. Am. J. Respir. Crit. Care Med. 179, 186–193 (2009).
Pochard, P. et al. Lactic acid bacteria inhibit TH2 cytokine production by mononuclear cells from allergic patients. J. Allergy Clin. Immunol. 110, 617–623 (2002).
Yoo, J., Tcheurekdjian, H., Lynch, S.V., Cabana, M. & Boushey, H.A. Microbial manipulation of immune function for asthma prevention: inferences from clinical trials. Proc. Am. Thorac. Soc. 4, 277–282 (2007).
Forsythe, P., Inman, M.D. & Bienenstock, J. Oral treatment with live Lactobacillus reuteri inhibits the allergic airway response in mice. Am. J. Respir. Crit. Care Med. 175, 561–569 (2007).
Smits, H.H. et al. Selective probiotic bacteria induce IL-10-producing regulatory T cells in vitro by modulating dendritic cell function through dendritic cell-specific intercellular adhesion molecule 3-grabbing nonintegrin. J. Allergy Clin. Immunol. 115, 1260–1267 (2005).
Zuany-Amorim, C. et al. Suppression of airway eosinophilia by killed Mycobacterium vaccae-induced allergen-specific regulatory T-cells. Nat. Med. 8, 625–629 (2002).
Cazzola, M., Rogliani, P. & Curradi, G. Bacterial extracts for the prevention of acute exacerbations in chronic obstructive pulmonary disease: a point of view. Respir. Med. 102, 321–327 (2008).
Soler, M., Mutterlein, R., Cozma, G. & Swiss-German, O.M.S.G. Double-blind study of OM-85 in patients with chronic bronchitis or mild chronic obstructive pulmonary disease. Respiration 74, 26–32 (2007).
Schaad, U.B., Mutterlein, R., Goffin, H. & Group, B.V.-C.S. Immunostimulation with OM-85 in children with recurrent infections of the upper respiratory tract: a double-blind, placebo-controlled multicenter study. Chest 122, 2042–2049 (2002).
Steurer-Stey, C., Lagler, L., Straub, D.A., Steurer, J. & Bachmann, L.M. Oral purified bacterial extracts in acute respiratory tract infections in childhood: a systematic quantitative review. [see comment]. Eur. J. Pediatr. 166, 365–376 (2007).
Mascarell, L. et al. A synthetic triacylated pseudo-dipeptide molecule promotes Th1/TReg immune responses and enhances tolerance induction via the sublingual route. Vaccine 26, 108–118 (2007).
Alyanakian, M.A. et al. Transforming growth factor-beta and natural killer T-cells are involved in the protective effect of a bacterial extract on type 1 diabetes. Diabetes 55, 179–185 (2006).
Rozy, A. & Chorostowska-Wynimko, J. Bacterial immunostimulants—mechanism of action and clinical application in respiratory diseases. Pneumonol. Alergol. Pol. 76, 353–359 (2008).
Clements, A. et al. Secondary acylation of Klebsiella pneumoniae lipopolysaccharide contributes to sensitivity to antibacterial peptides. J. Biol. Chem. 282, 15569–15577 (2007).
OM-PHARMA. Glucosamine disaccharides, method for their preparation, pharmaceutical composition comprising same, and their use. in patent WO/1995/014026 (ed. PHARMA, O.) (1995).
Holt, P.G., Strickland, D.H., Wikstrom, M.E. & Jahnsen, F.L. Regulation of immunological homeostasis in the respiratory tract. Nat. Rev. Immunol. 8, 142–152 (2008).
Raymond, M. et al. Selective control of SIRP-alpha-positive airway dendritic cell trafficking through CD47 is critical for the development of T(H)2-mediated allergic inflammation. J. Allergy Clin. Immunol. 124, 1333–1342. e1331 (2009).
Nguyen, K.D., Vanichsarn, C. & Nadeau, K.C. Impaired IL-10-dependent induction of tolerogenic dendritic cells by CD4+CD25hiCD127lo/− natural regulatory T cells in human allergic asthma. Am. J. Respir. Crit. Care Med. 180, 823–833 (2009).
Larche, M., Robinson, D.S. & Kay, A.B. The role of T lymphocytes in the pathogenesis of asthma. J. Allergy Clin. Immunol. 111, 450–463. quiz 464 (2003).
Medoff, B.D., Thomas, S.Y. & Luster, A.D. T cell trafficking in allergic asthma: the ins and outs. Annu. Rev. Immunol. 26, 205–232 (2008).
Wills-Karp, M. Interleukin-13 in asthma pathogenesis. Curr. Allergy Asthma Rep. 4, 123–131 (2004).
Akdis, M. et al. Immune responses in healthy and allergic individuals are characterized by a fine balance between allergen-specific T regulatory 1 and T helper 2 cells. J. Exp. Med. 199, 1567–1575 (2004).
Hartl, D. et al. Quantitative and functional impairment of pulmonary CD4+CD25hi regulatory T cells in pediatric asthma. J. Allergy Clin. Immunol. 119, 1258–1266 (2007).
Ling, E.M. et al. Relation of CD4+CD25+ regulatory T-cell suppression of allergen-driven T-cell activation to atopic status and expression of allergic disease. Lancet 363, 608–615 (2004).
Belkaid, Y. Regulatory T cells and infection: a dangerous necessity. Nat. Rev. Immunol. 7, 875–888 (2007).
Tang, Q. & Bluestone, J.A. The Foxp3+ regulatory T cell: a jack of all trades, master of regulation. Nat. Immunol. 9, 239–244 (2008).
Wohlfert, E. & Belkaid, Y. Role of endogenous and induced regulatory T cells during infections. J. Clin. Immunol. 28, 707–715 (2008).
Izcue, A., Coombes, J.L. & Powrie, F. Regulatory lymphocytes and intestinal inflammation. Annu. Rev. Immunol. 27, 313–338 (2009).
Chirdo, F.G., Millington, O.R., Beacock-Sharp, H. & Mowat, A.M. Immunomodulatory dendritic cells in intestinal lamina propria. Eur. J. Immunol. 35, 1831–1840 (2005).
Coombes, J.L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-beta and retinoic acid-dependent mechanism. [see comment]. J. Exp. Med. 204, 1757–1764 (2007).
Sun, C.M. et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3T reg cells via retinoic acid. [see comment]. J. Exp. Med. 204, 1775–1785 (2007).
Coombes, J.L. & Maloy, K.J. Control of intestinal homeostasis by regulatory T cells and dendritic cells. Semin. Immunol. 19, 116–126 (2007).
Feleszko, W., Jaworska, J. & Hamelmann, E. Toll-like receptors—novel targets in allergic airway disease (probiotics, friends and relatives). Eur. J. Pharmacol. 533, 308–318 (2006).
Wilson, M.S. et al. Suppression of allergic airway inflammation by helminth-induced regulatory T cells. J. Exp. Med. 202, 1199–1212 (2005).
Kitagaki, K., Businga, T.R. & Kline, J.N. Oral administration of CpG-ODNs suppresses antigen-induced asthma in mice. Clin. Exp. Immunol. 143, 249–259 (2006).
Gibbons, D.L. et al. Suppression of airway inflammation by a natural acute infection of the intestinal epithelium. Mucosal Immunol. 2, 144–155 (2009).
Matricardi, P.M. et al. Exposure to foodborne and orofecal microbes versus airborne viruses in relation to atopy and allergic asthma: epidemiological study. BMJ 320, 412–417 (2000).
Acknowledgements
The National Health and Medical Research Council of Australia and OM PHARMA, Geneva, Switzerland funded this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declared no conflict of interest.
Additional information
Author contributions: DHS and PGH shared responsibility for concept design and interpretation of data and article preparation. DHS was responsible for the initial data analysis and for supervision of technical aspects of the project. SJ and JAT carried out the majority of the basic immunology work. AL and PDS were responsible for lung function aspects of the study.
The funding bodies did not contribute to the initial conceptual design of the experimental work or the analysis and interpretation of the data.
SUPPLEMENTARY MATERIAL is linked to the online version of the paper
Rights and permissions
About this article
Cite this article
Strickland, D., Judd, S., Thomas, J. et al. Boosting airway T-regulatory cells by gastrointestinal stimulation as a strategy for asthma control. Mucosal Immunol 4, 43–52 (2011). https://doi.org/10.1038/mi.2010.43
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mi.2010.43
This article is cited by
-
Viral infections and wheezing–asthma inception in childhood: is there a role for immunomodulation by oral bacterial lysates?
Clinical and Translational Allergy (2020)
-
Notch Ligand DLL4 Alleviates Allergic Airway Inflammation via Induction of a Homeostatic Regulatory Pathway
Scientific Reports (2017)
-
The Immunotherapeutic Role of Bacterial Lysates in a Mouse Model of Asthma
Lung (2017)
-
Bystander immunotherapy as a strategy to control allergen-driven airway inflammation
Mucosal Immunology (2015)
-
Respiratory viral infections in children with asthma: do they matter and can we prevent them?
BMC Pediatrics (2012)