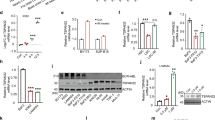
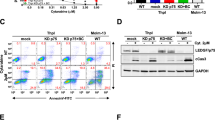
Abstract
BMI1 is a key component of the PRC1 (polycomb repressive complex-1) complex required for maintenance of normal and cancer stem cells. Its aberrant expression is detected in chronic myeloid leukemia and Ph+ acute lymphoblastic leukemia (ALL), but no data exist on BMI1 requirement in ALL cells. We show here that BMI1 expression is important for proliferation and survival of Ph+ ALL cells and for leukemogenesis of Ph+ cells in vivo. Levels of BIM, interferon-α (IFNα)-regulated genes and E2F7 were upregulated in BMI1-silenced cells, suggesting that repressing their expression is important for BMI1 biological effects. Consistent with this hypothesis, we found that: (i) downregulation of BIM or E2F7 abrogated apoptosis or rescued, in part, the reduced proliferation and colony formation of BMI1 silenced BV173 cells; (ii) BIM/E2F7 double silencing further enhanced colony formation and in vivo leukemogenesis of BMI1-silenced cells; (iii) overexpression of BIM and E2F7 mimicked the effect of BMI1 silencing in BV173 and SUP-B15 cells; and (iv) treatment with IFNα suppressed proliferation and colony formation of Ph+ ALL cells. These studies indicate that the growth-promoting effects of BMI1 in Ph+ ALL cells depend on suppression of multiple pathways and support the use of IFNα in the therapy of Ph+ ALL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Melo JV . The diversity of BCR-ABL fusion proteins and their relationship to leukemia phenotype. Blood 1996; 88: 2375–2384.
Goga A, McLaughlin J, Afar DE, Saffran DC, Witte ON . Alternative signals to RAS for hematopoietic transformation by the BCR-ABL oncogene. Cell 1995; 82: 981–988.
Nieborowska-Skorska M, Wasik MA, Slupianek A, Salomoni P, Kitamura T, Calabretta B et al. Signal transducer and activator of transcription (STAT)5 activation by BCR/ABLis dependent on intact Src homology (SH)3 and SH2 domains of BCR/ABL and is required for leukemogenesis. J Exp Med 1999; 189: 1229–1242.
Skorski T, Bellacosa A, Nieborowska-Skorska M, Majewski M, Martinez R, Choi JK et al. Transformation of hematopoietic cells by BCR-ABL requires activation of a PI-3k/Akt-dependent pathway. EMBO J 1997; 16: 6151–6161.
Perrotti D, Calabretta B . The biology of CML blast crisis. Blood 2004; 103: 4010–4022.
Perrotti D, Jamieson C, Goldman J, Skorski T . Chronic myeloid leukemia: mechanisms of blastic transformation. J Clin Invest 2010; 120: 2254–2264.
Lugo TG, Pendergast AM, Muller AJ, Witte ON . Tyrosine kinase activity and transformation potency of bcr-abl oncogene products. Science 1990; 247: 1079–1082.
Kantarjian H, Talpaz M, Dhingre K, Estey E, Keating MJ, Ku S et al. Significance of P210 versus P190 molecular abnormalities in adults with Philadelphia chromosome positive acute leukemia. Blood 1991; 78: 2411–2418.
Cimino G, Pane F, Elia L, Finocezzi E, Fazi P, Annino L et al. The role of BCR/ABL isoforms in the presentation and outcome of patients with Philadelphia-positive acute lymphoblastic leukemia: a seven year update of the GIMENA 0496 trial. Haematologica 2006; 91: 377–380.
Williams RT, Sherr CJ . The INK4-ARF (CDKN2A/B) locus in hematopoiesis and BCR-ABL-induced leukemias. Cold Spring Harb Symp Quant Biol 2008; 73: 461–467.
Mullighan CG, Miller CB, Radtke I, Phillips LA, Dalton J, Ma J et al. BCR-ABL1 lymphoblastic leukemia is characterized by the deletion of Ikaros. Nature 2008; 453: 110–114.
Klemm L, Duy C, Iacobucci I, Kuchen S, von Levetzow G, Feldhahn N et al. The B cell mutator AID promotes B lymphoid blast crisis and drug resistance in chronic myeloid leukemia. Cancer Cell 2009; 16: 232–245.
Gruber TA, Chang MS, Sposto R, Müshen M . Activation-induced cytidine deaminase accelerates clonal evolution in BCR-ABL1-driven B cell lineage acute lymphoblastic leukemia. Cancer Res 2010; 70: 7411–7420.
van Lohuizen M, Frasch M, Wientjens E, Berns A . Sequence similarity between the mammalian bmi-1 proto-oncogene and the Drosophila regulatory genes Psc and Su(z)2. Nature 1991; 353: 353–355.
Waldron T, De Dominici M, Soliera AR, Audia A, Iacobucci I, Lonetti A et al. c-Myb and its target Bmi1 are required for p190BCR/ABL leukemogenesis in mouse and human cells. Leukemia 2012; 26: 644–653.
Cao R, Tsukada Y, Zhang Y . Role of Bmi-1 and Ring1A in H2A ubiquitylation and Hox gene silencing. Mol Cell 2005; 20: 845–854.
Lessard J, Sauvageau G . Bmi-1 determines the proliferative capacity of normal and leukaemic hematopoietic stem cells. Nature 2003; 423: 255–260.
Park IK, Qian D, Kiel M, Becker MW, Pihalja M, Weissman IL et al. Bmi-1 is required for maintenance of adult self-renewing hematopoietic stem cells. Nature 2003; 423: 302–305.
Molofsky AV, Pardal R, Iwashita T, Park IK, Clarke MF, Morrison SJ . Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature 2003; 425: 962–967.
Sparmann A, van Lohuizen M . Polycomb silencers control cell fate,development and cancer. Nat Rev Cancer 2006; 6: 846–856.
Sauvageau M, Sauvageau G . Polycomb group proteins: multi-facet regulators of somatic stem cells and cancer. Cell Stem Cell 2010; 7: 299–313.
Jiang L, Li J, Song L . Bmi-1, stem cells and cancer. Acta Biochim Biophys Sin (Shanghai) 2009; 41: 527–534.
Cao L, Bombard J, Cintron K, Sheedy J, Weatall ML, Davis TW . BMI1 as a novel target for drug discovery in cancer. J Cell Biochem 2011; 112: 2729–2741.
Glinsky GV . Death-from-cancer signatures and stem cell contribution to metastatic cancer. Cell Cycle 2005; 4: 1171–1175.
Glinsky GV, Berezovska O, Glinskii AB . Microarray analysis identifies a death-from-cancer signature predicting therapy failure in patients with multiple types of cancer. J Clin Invest 2005; 115: 1503–1521.
Mohty M, Yong AS, Szydlo RM, Apperley JF, Melo JV . The polycomb group BMI1 gene is a molecular marker for predicting prognosis of chronic myeloid leukemia. Blood 2007; 110: 380–383.
Bhattacharyya J, Mihara K, Yasunaga S, Tanaka H, Hoshi M, Takihara Y et al. BMI-1 expression is enhanced through transcriptional and posttranscriptional regulation during the progression of chronic myeloid leukemia. Ann Hematol 2009; 88: 333–340.
Rizo A, Horton SJ, Olthef S, Dontje B, Aurema A, van Os R et al. BMI1 collaborates with BCR-ABL in leukemic transformation of human CD34+ cells. Blood 2010; 116: 4621–4630.
Sengupta A, Ficker AM, Dunn SK, Madhu M, Cancelas JA . Bmi-1 reprograms CML B-lymphoid progenitors to become B-ALL-initiating cells. Blood 2012; 119: 494–502.
Jacobs JJ, Kieboom K, Marino S, DePinho RA, van Lohuizen M . The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature 1999; 397: 164–168.
Jacobs JJ, Scheijen B, Vonchen JW, Kieboom K, Berns A, van Lohuizen M . Bmi-1 collaborates with c-Myc in tumorigenesis by inhibiting c-Myc-induced apoptosis via INK4a/ARF. Genes Dev 1999; 13: 2678–2690.
Douglas D, Hsu JH, Hung L, Cooper A, Abdueva D, van Doorninck J et al. BMI-1 promotes Ewing sarcoma tumorigenicity independent of CDKN2A repression. Cancer Res 2008; 68: 6507–6511.
Bruggeman SW, Hulsman T, Tanger E, Buckle T, Blom M, Zevenhoven J et al. Bmi1 controls tumor development in an Ink4a/arf-independent manner in a mouse model for glioma. Cancer Cell 2007; 12: 328–341.
Jagani Z, Wiederschain D, Loo A, He D, Mosher R, Fordjour P et al. The Polycomb group protein Bmi-1 is essential for the growth of multiple myeloma cells. Cancer Res 2010; 70: 5528–5538.
Feldhahn N, Henke N, Melchior K, Duy C, Soh BN, Klein F et al. Activation-induced cytidine deaminase acts as a mutator in BCR-ABL1-transformed acute lymphoblastic leukemia cells. J Exp Med 2007; 204: 1157–1166.
Westendorp B, Mokry M, Groot Koerkamp MJ, Holstege FC, Cuppen E, de Bruin A . E2F7 represses a network of oscillating cell cycle genes to control S-phase progression. Nucleic Acids Res 2012; 40: 3511–3523.
Liu B, Shats I, Angus SP, Gatza ML, Nevins JR . Interaction of E2F7 transcription factor with E2F1 and C-terminal binding protein (CtBP) provides a mechanism for E2F7-dependent transcription repression. J Biol Chem 2013; 288: 24581–24589.
Kreso A, van Galen P, Pedley NM, Lima-Fernandes E, Frelin C, Davis T et al. Self-renewal as a therapeutic target in human colorectal cancer. Nat Med 2014; 20: 29–38.
Ottmann OG, Pfeifer H . First-line treatment of Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL) in adults. Curr Opin Oncol 2009; Suppl 1: S43–S46.
Branford S, Rudzki Z, Walsh S, Grigg A, Arthur C, Taylor K et al. High frequency of point mutations clustered within the adenosine triphosphate-binding region of BCR/ABL in patients with chronic myeloid leukemia or Ph-positive acute lymphoblastic leukemia (Ph+ ALL) who develop imatinib (STI571) resistance. Blood 2002; 99: 3472–3475.
Pfeifer H, Wassmann B, Pavlova A, Wunderle L, Oldenburg J, Binckebanck A et al. Kinase domain mutations of BCR-ABL frequently precede imatinib-based therapy and give rise to relapse in patients with de novo Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood 2007; 110: 727–734.
Visani G, Isidori A, Malagola M, Alberti D, Capdeville R, Martinelli G et al. Efficacy of imatinib mesylate (STI571) in conjunction with alpha-interferon: long-term quantitative molecular remission in relapsed P-190BCR-ABL-positive acute lymphoblastic leukemia. Leukemia 2002; 16: 2159–2161.
Wassmann B, Scheuring U, Pfeifer H, Binckebanck A, Kabish A, Lübbert M et al. Efficacy and safety of imatinib mesylate (Glivec) in combination with interferon-alpha (IFN-alpha) in Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL). Leukemia 2003; 17: 1919–1924.
Piccaluga PP, Martinelli G, Isidori A, Malagola M, Rondoni M, Paolini S et al. long-term molecular complete remission with IFN-α in Ph+ adult acute lymphoid leukemia patients. Leukemia 2008; 22: 1617–1618.
Mourgues L, Imbert V, Nebout M, Colosetti P, Neffati Z, Lagadec P et al. The BMI1 polycomb protein represses cyclin G2-induced autophagy to support proliferation in chronic myeloid leukemia cells. Leukemia 2015; 29: 1993–2002.
Acknowledgements
This work was supported, in part, by the NCI grant RO1 CA167169 (to BC) and by the SKCC support grant P30 CA056036. Dr Samanta A Mariani was supported, in part, by an AIRC-Marie Curie international fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Mariani, S., Minieri, V., De Dominici, M. et al. CDKN2A-independent role of BMI1 in promoting growth and survival of Ph+ acute lymphoblastic leukemia. Leukemia 30, 1682–1690 (2016). https://doi.org/10.1038/leu.2016.70
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.70
This article is cited by
-
Nuclear Vav3 is required for polycomb repression complex-1 activity in B-cell lymphoblastic leukemogenesis
Nature Communications (2022)