Abstract
Purpose
To evaluate the impact of traditional French summer vacation on visual acuity and spectral domain-optical coherence tomography (SD-OCT) of Wet AMD patients being treated with intravitreal Ranibizumab.
Methods
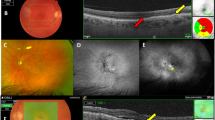
This was a consecutive, comparative, single-centre, prospective analysis. All patients who were being treated with intravitreal injection of 0.5 mg ranibizumab at Cergy Pontoise Hospital, Department of Ophthalmology between July 2013 and September 2014 were included. Patients were divided into two groups: (A) patients who skipped one ranibizumab intravitreal injection during holidays, and (B) patients who received injection during their holidays. Evaluations occurred prior to traditional holiday (baseline) and 2 months later, consisting of BCVA using ETDRS, and a complete ophthalmic examination that included slit-lamp biomicroscopy, fundus examination, fluorescein angiography (FA), indocyanine green angiography (ICGA), and spectral domain-optical coherence tomography (SD-OCT). All patients were being treated with PRN anti-VEGF regimen and criteria for reinjection included a visual acuity loss >5 ETDRS letters and/or an increase of central retinal thickness, presence of subretinal fluid, intraretinal fluid, or pigment epithelium detachment. If reinjection criteria were not met, patients were advised to return in 4 weeks.
Results
The mean visual acuity change was −0.071±0.149 (LogMAR) in group A and +0.003±0.178 in group B (P=0.041). At the second visit (2 months after preholidays visit), 61.8% of patients in group A had SRF and/or intraretinal cysts, and only 27.6% of patients in group B. There was a significant difference in the persistence of fluid between the two groups (P=0.007, χ2-test).
Conclusion
This cases series demonstrated the detrimental impact of holidays on visual acuity in patients treated with ranibizumab for AMD, which, in spite of their treatment regimen, still leave in vacation. Therefore, it is important to convey the message of treatment adherence to patients, despite their need of holidays.
Similar content being viewed by others
Introduction
Age related macular degeneration (AMD) is the main cause of visual impairment in developed countries among individuals over 50 years of age.1, 2 Recently, the prognosis for neovascular AMD has improved owing to the growing to the availability of intravitreal anti-vascular endothelial growth factor (VEGF) therapy.3, 4 Ranibizumab (Lucentis; Genentech Inc, San Francisco, California, USA) is a recombinant humanized monoclonal antibody fragment that neutralizes all biologically active forms of VEGF-A.5 Treatment with intravitreal ranibizumab for exudative AMD has demonstrated, in two phase III clinical studies, efficacy in prevention of vision loss and improvement in mean visual acuity (VA) in a large proportion of patients.3, 4 These results were obtained using a fixed-dosing regimen of ranibizumab 0.5 mg or 0.3 mg monthly, injected over 24 and 12 months, respectively. Two pivotal clinical studies (The Prospective Optical Coherence Tomography Imaging of Patients with Neovascular AMD Treated with Intra-Ocular Ranibizumab (PrONTO)6 and Comparison of Age-Related Macular Degeneration Treatment Trials (CATT)7 demonstrated that a variable dosing regimen was effective (even though not strictly equal to a fixed-dosing regimen) in improving VA and OCT outcomes, requiring a lower number of injections over a period of 12 and even 24 months. At our facility, we apply the variable dosing regimen to treating our patients with neovascular AMD, with monitoring for treatment typically taking place at four week intervals.
In 1231, Pope Gregory II designated that people should be given holiday for agricultural work. In France, until the First World War, students were only given 1 month of summer vacation until 1939, when a decree standardizes summer holidays to include from 15 July to 30 September and later from 28 June to 16 September in the 1960s. This summer vacation, initially related to agricultural activities in France centuries ago, has evolved into paid leave and tourism activities for much of the country, including patients and physicians. Thus, it is common for patients in France to take extended holidays in the summer, and opt to delay, skip or put their anti-VEGF treatments ‘on hold’ until September, giving priority to their holidays over the progression of their eye disease. The goal of this study was to evaluate VA and OCT in AMD patients before and after the summer holidays and compare those patients that missed a treatment owing to those who did not.
Materials and methods
This was a consecutive, comparative, single-centre, prospective analysis. All patients that were being treated with intravitreal injection of 0.5 mg ranibizumab at Cergy Pontoise Hospital, Department of Ophthalmology between July, 2013 and September, 2014 were included. Patients were divided into two groups: (A) patients who skipped one ranibizumab intravitreal injection during holidays, and (B) patients that received injection during their holidays. This prospective study was performed in agreement with the French bioethical legislation and in agreement with the Declaration of Helsinki for research involving human subjects and approved by The French Society of Ophthalmology’s ethics committee.
In order to be included, patients had to be >50 years old, have a presence of subfoveal CNV secondary to AMD as evaluated by fundus biomicroscopy, fluorescein angiography (FA) and infracyanine green angiography (ICGA), and a presence of serous retinal detachment, pigment epithelium detachment detected on SD-OCT. All patients had previous treatment for exudative AMD. Patients were excluded if they had a refractive error >−6 diopters, macular hemorrhage, adult-onset foveomacular vitelliform dystrophy or other pattern dystrophies, fibroglial scar, CNV attributable to causes other than AMD, or active intraocular inflammation in the study eye.
The baseline evaluation for all patients before holiday (June) and consisted of BCVA using ETDRS, and a complete ophthalmic examination that included slit-lamp biomicroscopy, fundus examination, fluorescein angiography (FA), indocyanine green angiography (ICG) and spectral domain-optical coherence tomography (SD-OCT). All patients were being treated with PRN Anti-VEGF regimen and criteria for reinjection included a VA loss >5 ETDRS letters and/or an increase of central retinal thickness, presence of subretinal fluid, intraretinal fluid, or pigment epithelium detachment. If reinjection criteria were not met, patients were advised to return in 4 weeks.
At two months (final visit) from baseline, each patient underwent an ophthalmic examination, including of BCVA using ETDRS, and a complete ophthalmic examination that included slit-lamp biomicroscopy, fundus examination, FA, ICGA, and SD-OCT. For this analysis only BCVA and the presence of subretinal fluid were included, although all other parameters are part of usual care.
Data was collected and recorded on a pre-designed data collection sheet and managed with an excel spread sheet. Snellen VA measurements were converted to logMAR for analysis. The arithmetic means and standard deviations were calculated for all the descriptive parameters using Stata-11 software (StataCorp LP, College Station, TX, USA). The Chi-square test was used to determine the significance of the association between categorical variables and comparison of interval variables was made using the Mann–Whitney test. P<0.05 was considered to be statistically significant.
Results
Clinical characteristics of the study population at baseline
Sixty three eyes of 63 consecutive patients (28 male, aged 79–81 years [mean 80.2±7.6]) were included in the analysis (Table 1). At baseline, mean BCVA was 0.86±0.35 logMAR. CNV was classified as classic in 9 eyes (14.2%), occult in 48 eyes (76.2%), and retinal angiomatous proliferation (RAP) in 6 eyes (9.5%). All patients had previously received intravitreal injections of ranibizumab for exudative AMD, there were no treatment-naïve patients. The mean number of previous intravitreal injections was 9.5±4.7 injections. All eyes included in this study showed evidence of exudation on OCT (subretinal or intraretinal fluid) during the visit preceding vacation. Patients were divided into two groups: Group A including patients who opted to skip one ranibizumab intravitreal injection during their holidays, and Group B patients adhered to their treatment regimen and received injection during their holidays. Comparison between the two groups showed that there were no differences at baseline (before holidays) in mean age, gender, mean BCVA, and CNV subtype.
Functional and morphologic changes 2 months after first visit
In group A, no eyes gained 5 or more ETDRS letters, 67.6% of eyes (23 eyes) lost 5 or more ETDRS letters, and in 32.4% of eyes (11 eyes) BCVA remained stable (gain or loss of<5 ETDRS letters). In group B, no eyes gained 5 or more ETDRS letters, 75.9% of eyes (22 eyes) lost 5 or ETDRS letters, and in 24.1% of eyes (7 eyes) BCVA remained stable (gain or loss <5 ETDRS letters; P=0.580).
Initial mean VA in group A (0.83±0.33 logMAR) was not different from mean initial VA in group B vs (0.89±0.37 logMAR) P=0.141). After 2 months (post holidays), the mean VA changed by −0.071±0.149 for group A and +0.003±0.178 in group B (P=0.041, Mann–Whitney test), a significant difference.
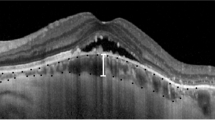
All eyes had subretinal fluid at baseline. After 2 months, 61.8% of patients in group A had SRF and/or intraretinal cysts, and only 27.6% of patients in group B had persistence of SRF and/or intraretinal cysts. There was a significant difference in the persistence of fluid between the two groups (P=0.007, χ2-test). For patients with serous central detachment, 72.4% demonstrated a decrease of fluid after holidays in group B compared with only 38.2% in group A. (P=0.007).
Discussion
Patterns of nonadherence to treatment regimens is a challenge in many disease states. Several studies have investigated the negative impact of missed doses or missed appointments while patients were on holiday and raised concerns in many areas, including diabetes care, hemodialysis, and antihypertensive care.8, 9, 10 The goal of this cases series was to demonstrate the impact of the absence of treatment in patients with active exudative AMD, whatever the reason for not adhering to prescribed monitoring and treatment. In some regions and health systems around the World, patients may experience delay in access for AMD-related care due to clinical or nonclinical circumstances. An example of such circumstances has been described by Real et al11 in Argentina, where funding approval for ranibizumab is typically delayed for 160 days in order to verify clinical need, leading to significant visual decline. Similar delays to treatment, as a consequence of clinical verification of need have also been reported in Germany.12
The treatment of patients in our cohort were not restricted by bureaucratic barriers, but rather a delay due to patient choice. We did focus the summer holiday period in France because we have found it to be one of the most pronounced periods of absenteeism for treatment and monitoring of AMD in the French population. Summer holidays were specifically chosen owing to their longer duration, as compared with the typical 2-week period of Christmas and New Year’s. In addition, periods of annual fasting or Ramadan in some populations was considered, although despite the observances, patients often continue to receive their intravitreal injection during that time.
As ranibizumab has been shown to be a highly effective in the treatment of neovascular AMD and has become the reference treatment and recognized standard of care, patients now have an elevated treatment expectation beyond the slowing of vision loss, and now anticipate stabilization or improvement of their vision.13
The initial dosing regimen used in our routine practice was pro re nata (PRN (as needed)) basis. The PrONTO study was an open-label prospective, single-centre, nonrandomized clinical study which employed a protocol of three initial monthly injections followed by an as needed (PRN) decision to retreat or not based on the evolution of VA and the presence or absence of subfoveal fluid as observed by OCT.6 All eyes in our cohort benefited from this protocol, as measured by OCT. The mean number of injections performed for each eye during the 2-month period in our series was 9.5±4.7 injections, similar to those with atrophy at 24 months in the PrONTO protocol, 9.9 and 8.6, respectively.
In group A, BCVA significantly decreased in 48.5% patients, maintained in 40.1%, whereas 11.4% of patients demonstrated increased VA. In comparison, in group B; only 17.8% had decrease of VA, 32.1% maintained their VA and half of patients (50.1%) in group B improved. Our findings for group A appear to align with the natural history and prognosis of neovascular age-related macular degeneration. Wong et al14 performed a meta-analysis of 53 studies and demonstrated mean baseline VA of 0.64 logMAR that lost 0.1logMAR after 3 months. This is comparable to our group A, which was 0.83logMAR at baseline and declined by 0.071 after 2 months. In addition, the study from Muether et al12 found that for their cohort of German patients delaying anti-VEGF retreatment, a regression analysis revealed a VA loss of one ETDRS line within 43 days.
Though a significant percentage of our patients treated with ranibizumab had an increase in BCVA after holidays. We could not confirm the exact role of ranibizumab in the increase of VA of 11.4% of patients that skipped a treatment in group A. Overall there was insufficient information to assign causality.
As expected, ranibizumab has demonstrated its effectiveness in the reabsorption of the subretinal fluid, particularly in those patients who received treatment during the holiday months; however, some patients had a persistence of fluid, which can be attributed to reasons including the chronicity of the disease, the presence of fibrosis or effect of tachyphylaxis. Furthermore, we observed the reabsorption of subretinal fluid in a few patients who did not receive their scheduled ranibizumab injection, which may be owing to some patients being good responders, genetic influence or the possibility of previous ranibizumab being maintained in the vitreous.
We recognize that our study had several limitations, particularly that it is a small series and not representative of all French population. Although our patient groups were quite evenly matched for age, gender, CNV type, and duration of neovascular AMD, they were not matched for health factors, such as smoking status. As well, we did not investigate the resources available to the patients in order to return for, or seek out treatment during their holidays. All of our patients were treated with a PRN regimen and the study’s monocentric nature of data collection and imaging analysis during follow-up and the somewhat short follow-up period are recognized limitation. As well, changes in the measurements of central macular thickness and epithelium pigment detachment were not evaluated because in their regular monitoring, all patients do not undergo CMT measurements. Further studies with larger sample sizes and longer follow-up periods are needed for a more precise evaluation.
This cases series demonstrated the detrimental impact of holidays on VA in patients treated with ranibizumab for AMD, which, in spite of their treatment regimen, still leave in vacation. Therefore, it is important to convey the message of treatment adherence to patients, despite their need of holidays. The intention is certainly not to deprive AMD patients of holidays, but rather to increase awareness to consult and pursue their treatment on vacation in order to preserve their VA. Although we are not aware of any specific policies in place to recommend seeking clinical care while on holiday, in order to ensure continuity of patients care, it would be beneficial to provide patients with a list available of Ophthalmologists in various locations to seek treatment during their holidays, or prearrange treatment on patient’s behalf. Hence, alternative or emergency service to maintain AMD treatment during the summer holiday period may be considered.
Further prospective investigations are needed to improve our knowledge regarding the worsening signs in some exudative AMD patients and the stability in other patients to improve monitoring and treatment strategies during vacation periods.

References
Bressler NM . Age-related macular degeneration is the leading cause of blindness. JAMA 2004; 291 (15): 1900–1901.
Wong TY, Wong T, Chakravarthy U, Klein R, Mitchell P, Zlateva G et al. The natural history and prognosis of neovascular age-related macular degeneration: a systematic review of the literature and meta-analysis. Ophthalmology 2008; 115 (1): 116–126.
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 2006; 355 (14): 1419–1431.
Brown DM, Kaiser PK, Michels M, Soubrane G, Heier JS, Kim RY et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 2006; 355 (14): 1432–1444.
Fung AE, Lalwani GA, Rosenfeld PJ, Dubovy SR, Michels S, Feuer WJ et al. An optical coherence tomography-guided, variable dosing regimen with intravitreal ranibizumab (Lucentis) for neovascular age-related macular degeneration. Am J Ophthalmol 2007; 143 (4): 566–583.
Lalwani GA, Rosenfeld PJ, Fung AE, Dubovy SR, Michels S, Feuer W et al. A variable-dosing regimen with intravitreal ranibizumab for neovascular age- related macular degeneration: year 2 of the PrONTO Study. Am J Ophthalmol 2009; 148 (1): 43–58.
CATT Research Group, Martin DF, Maguire MG, Ying G, Grunwald JE, Fine SL et al. Ranibizumab and bevacizumab for neovascular age- related macular degeneration. N Engl J Med 2011; 364 (20): 1897–1908.
Coyle ME, Francis K, Chapman Y . Self-management activities in diabetes care: a systematic review. Aust Health Rev 2013; 37 (4): 513–522.
Chan KE, Thadhani RI, Maddux FW . Adherence barriers to chronic dialysis in the United States. J Am Soc Nephrol 2014; 25 (11): 2642–2648.
Grigoryan L, Pavlik VN, Hyman DJ . Patterns of nonadherence to antihypertensive therapy in primary care. J Clin Hypertens (Greenwich) 2013; 15 (2): 107–111.
Real JP, Granero GE, De Santis MO, Juarez CP, Palma SD, Kelly SP, Luna JD . Rate of vision loss in neovascular age-related macular degeneration explored. Graefes Arch Clin Exp Ophthalmol 2014; e-pub ahead of print 11 December 2014.
Muether PS, Hermann MM, Koch K, Fauser S . Delay between medical indication to anti-VEGF treatment in age-related macular degeneration can result in a loss of visual acuity. Graefes Arch Clin Exp Ophthalmol 2011; 249 (5): 633–637.
Kaiser PK . Ranibizumab: the evidence of its therapeutic value in neovascular age-related macular degeneration. Core Evid 2008; 2 (4): 273–294.
Wong TY, Chakravarthy U, Klein R, Mitchell P, Zlateva G, Buggage R et al. The natural history and prognosis of neovascular age-related macular degeneration: a systematic review of the literature and metaanalysis. Ophthalmology 2008; 115: 116–126.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Massamba, N., Dirani, A., Knoeri, J. et al. Evaluating the impact of summer vacation on the visual acuity of AMD patients treated with ranibizumab. Eye 29, 1453–1457 (2015). https://doi.org/10.1038/eye.2015.128
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2015.128
This article is cited by
-
Non-adherence and non-persistence to intravitreal anti-vascular endothelial growth factor (anti-VEGF) therapy: a systematic review and meta-analysis
Systematic Reviews (2023)
-
Reasons for Delayed Anti-VEGF Treatment During COVID-19 Lockdown and Clinical Impact in Neovascular Age-Related Macular Degeneration
Ophthalmology and Therapy (2023)
-
Adhärenz bei der Anti-VEGF-Therapie – Überlegungen und praktische Empfehlungen
Der Ophthalmologe (2021)
-
The effect of COVID-19 pandemic restrictions on neovascular AMD patients treated with treat-and-extend protocol
International Ophthalmology (2021)
-
A 12-month, prospective, observational study of ranibizumab in treatment-naïve Taiwanese patients with neovascular age-related macular degeneration: the RACER study
BMC Ophthalmology (2020)