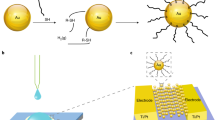
Abstract
Although multiple methods have been developed to detect metal cations, only a few offer sensitivities below 1 pM, and many require complicated procedures and sophisticated equipment. Here, we describe a class of simple solid-state sensors for the ultrasensitive detection of heavy-metal cations (notably, an unprecedented attomolar limit for the detection of CH3Hg+ in both standardized solutions and environmental samples) through changes in the tunnelling current across films of nanoparticles (NPs) protected with striped monolayers of organic ligands. The sensors are also highly selective because of the ligand–shell organization of the NPs. On binding of metal cations, the electronic structure of the molecular bridges between proximal NPs changes, the tunnelling current increases and highly conductive paths ultimately percolate the entire film. The nanoscale heterogeneity of the structure of the film broadens the range of the cation-binding constants, which leads to wide sensitivity ranges (remarkably, over 18 orders of magnitude in CH3Hg+ concentration).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Boening, D. W. Ecological effects, transport, and fate of mercury: A general review. Chemosphere 40, 1335–1351 (2000).
Clarkson, T. W. The toxicology of mercury. Crit. Rev. Clin. Lab. Sci. 34, 369–403 (1997).
Matsumoto, H., Koya, G. & Takeuchi, T. Fetal minamata disease: A neuropathological study of two cases of intrauterine intoxication by a methyl mercury compound. J. Neuropathol. Exp. Neurol. 24, 563–574 (1965).
Harada, M. Minamata disease—methylmercury poisoning in Japan caused by environmental pollution. Crit. Rev. Toxicol. 25, 1–24 (1995).
Inaba, T. et al. Estimation of cumulative cadmium intake causing Itai-itai disease. Toxicol. Lett. 159, 192–201 (2005).
Nomiyama, K. Recent progress and perspectives in cadmium health-effects studies. Sci. Total Environ. 14, 199–232 (1980).
Nriagu, J. O. & Pacyna, J. M. Quantitative assessment of worldwide contamination of air, water and soils by trace-metals. Nature 333, 134–139 (1988).
Lin, S. H. & Juang, R. S. Heavy metal removal from water by sorption using surfactant-modified montmorillonite. J. Hazard. Mater. 92, 315–326 (2002).
Jensen, S. & Jernelov, A. Biological methylation of mercury in aquatic organisms. Nature 223, 753–754 (1969).
Narin, I., Soylak, M., Elci, L. & Dogan, M. Determination of trace metal ions by AAS in natural water samples after preconcentration of pyrocatechol violet complexes on an activated carbon column. Talanta 52, 1041–1046 (2000).
Ghaedi, M., Ahmadi, F. & Shokrollahi, A. Simultaneous preconcentration and determination of copper, nickel, cobalt and lead ions content by flame atomic absorption spectrometry. J. Hazard. Mater. 142, 272–278 (2007).
Caroli, S., Forte, G., Iamiceli, A. L. & Galoppi, B. Determination of essential and potentially toxic trace elements in honey by inductively coupled plasma-based techniques. Talanta 50, 327–336 (1999).
Demuth, N. & Heumann, K. G. Validation of methylmercury determinations in aquatic systems by alkyl derivatization methods for GC analysis using ICP-IDMS. Anal. Chem. 73, 4020–4027 (2001).
Lee, J. S., Han, M. S. & Mirkin, C. A. Colorimetric detection of mercuric ion (Hg2+) in aqueous media using DNA-functionalized gold nanoparticles. Angew. Chem. Int. Ed. 46, 4093–4096 (2007).
Huang, C. C. & Chang, H. T. Parameters for selective colorimetric sensing of mercury(II) in aqueous solutions using mercaptopropionic acid-modified gold nanoparticles. Chem. Commun. 1215–1217 (2007).
Chiang, C. K., Huang, C. C., Liu, C. W. & Chang, H. T. Oligonucleotide-based fluorescence probe for sensitive and selective detection of mercury (II) in aqueous solution. Anal. Chem. 80, 3716–3721 (2008).
Chang, H. Y., Hsiung, T. M., Huang, Y. F. & Huang, C. C. Using rhodamine 6G-modified gold nanoparticles to detect organic mercury species in highly saline solutions. Environ. Sci. Technol. 45, 1534–1539 (2011).
Kang, T., Hong, S. R., Moon, J., Oh, S. & Yi, J. Fabrication of reusable sensor for detection of Cu2+ in an aqueous solution using a self-assembled monolayer with surface plasmon resonance spectroscopy. Chem. Commun. 3721–3723 (2005).
Li, D., Wieckowska, A. & Willner, I. Optical analysis of Hg2+ ions by oligonucleotide-gold-nanoparticle hybrids and DNA-based machines. Angew. Chem. Int. Ed. 47, 3927–3931 (2008).
Wang, G. Q., Wang, Y. Q., Chen, L. X. & Choo, J. Nanomaterial-assisted aptamers for optical sensing. Biosens. Bioelectron. 25, 1859–1868 (2010).
Yin, J. et al. SERS-active nanoparticles for sensitive and selective detection of cadmium ion (Cd2+). Chem. Mater. 23, 4756–4764 (2011).
Lin, Z. Z., Li, X. H. & Kraatz, H. B. Impedimetric immobilized DNA-based sensor for simultaneous detection of Pb2+, Ag+, and Hg2+. Anal. Chem. 83, 6896–6901 (2011).
Zhang, Z., Yu, K., Bai, D. & Zhu, Z. Q. Synthesis and electrochemical sensing toward heavy metals of bunch-like bismuth nanostructures. Nanoscale Res. Lett. 5, 398–402 (2010).
Thompson, R. B., Maliwal, B. P., Feliccia, V. L., Fierke, C. A. & McCall, K. Determination of picomolar concentrations of metal lens using fluorescence anisotropy: Biosensing with a ‘reagentless’ enzyme transducer. Anal. Chem. 70, 4717–4723 (1998).
Forzani, E. S., Zhang, H. Q., Chen, W. & Tao, N. J. Detection of heavy metal ions in drinking water using a high-resolution differential surface plasmon resonance sensor. Environ. Sci. Technol. 39, 1257–1262 (2005).
Kumar, M. & Zhang, P. Highly sensitive and selective label-free optical detection of mercuric ions using photon upconverting nanoparticles. Biosens. Bioelectron. 25, 2431–2435 (2010).
Song, H. D. et al. Picomolar selective detection of mercuric ion (Hg2+) using a functionalized single plasmonic gold nanoparticle. Nanotechnology 21, 145501 (2010).
Hinkle, P. M., Kinsella, P. A. & Osterhoudt, K. C. Cadmium uptake and toxicity via voltage-sensitive calcium channels. J. Biol. Chem. 262, 16333–16337 (1987).
Rashed, M. N. Cadmium and lead levels in fish (Tilapia nilotica) tissues as biological indicator for lake water pollution. Environ. Monit. Assess. 68, 75–89 (2001).
Nakanishi, H. et al. Photoconductance and inverse photoconductance in films of functionalized metal nanoparticles. Nature 460, 371–375 (2009).
DeVries, G. A. et al. Divalent metal nanoparticles. Science 315, 358–361 (2007).
Jackson, A. M., Myerson, J. W. & Stellacci, F. Spontaneous assembly of subnanometre-ordered domains in the ligand shell of monolayer-protected nanoparticles. Nature Mater. 3, 330–336 (2004).
Witt, D., Klajn, R., Barski, P. & Grzybowski, B. A. Applications properties and synthesis of omega-functionalized n-alkanethiols and disulfides—the building blocks of self-assembled monolayers. Curr. Org. Chem. 8, 1763–1797 (2004).
Kowalczyk, B., Apodaca, M. M., Nakanishi, H., Smoukov, S. K. & Grzybowski, B. A. Lift-Off and micropatterning of mono- and multilayer nanoparticle films. Small 5, 1970–1973 (2009).
Klajn, R., Bishop, K. J. M. & Grzybowski, B. A. Light-controlled self-assembly of reversible and irreversible nanoparticle suprastructures. Proc. Natl Acad. Sci. USA 104, 10305–10309 (2007).
Kowalczyk, B., Lagzi, I. & Grzybowski, B. A. Nanoarmoured droplets of different shapes formed by interfacial self-assembly and crosslinking of metal nanoparticles. Nanoscale 2, 2366–2369 (2010).
Han, S. B. et al. Chromatography in a single metal-organic framework (MOF) crystal. J. Am. Chem. Soc. 132, 16358–16361 (2010).
Sullivan, K. A. & Mason, R. P. The concentration and distribution of mercury in Lake Michigan. Sci. Total Environ. 213, 213–228 (1998).
Jackson, B., Taylor, V., Baker, R. A. & Miller, E. Low-level mercury speciation in fresh waters by isotope dilution GC-ICP-MS. Environ. Sci. Technol. 43, 2463–2469 (2009).
Pople, J. & Beveridge, D. Approximate Molecular Orbital Theory (McGraw-Hill, 1970).
Stewart, J. J. P. Stewart Computational Chemistry (Colorado Springs, 2007).
Nitzan, A. Chemical Dynamics in Condensed Phases : Relaxation, Transfer and Reactions in Condensed Molecular Systems Ch. 16 (Oxford, 2006).
Burlakov, V. M. et al. Discrete hopping model of exciton transport in disordered media. Phys. Rev. B 72, 075206 (2005).
Holmlin, R. E. et al. Electron transport through thin organic films in metal-insulator-metal junctions based on self-assembled monolayers. J. Am. Chem. Soc. 123, 5075–5085 (2001).
Landauer, R. The electrical resistance of binary metallic mixtures. J. Appl. Phys. 23, 779–784 (1952).
Kirkpatr, S. Classical transport in disordered media—scaling and effective-medium theories. Phys. Rev. Lett. 27, 1722–1725 (1971).
Sips, R. On the structure of a catalyst surface. J. Chem. Phys. 16, 490–495 (1948).
Kumar, K. V. & Porkodi, K. Relation between some two- and three-parameter isotherm models for the sorption of methylene blue onto lemon peel. J. Hazard. Mater. 138, 633–635 (2006).
Hristovski, K., Baumgardner, A. & Westerhoff, P. Selecting metal oxide nanomaterials for arsenic removal in fixed bed columns: From nanopowders to aggregated nanoparticle media. J. Hazard. Mater. 147, 265–274 (2007).
Vijayendran, R. A. & Leckband, D. E. A quantitative assessment of heterogeneity for surface-immobilized proteins. Anal. Chem. 73, 471–480 (2001).
Verma, A. et al. Surface-structure-regulated cell-membrane penetration by monolayer-protected nanoparticles. Nature Mater. 7, 588–595 (2008).
Jackson, A. M., Hu, Y., Silva, P. J. & Stellacci, F. From homoligand- to mixed-ligand-monolayer-protected metal nanoparticles: A scanning tunneling microscopy investigation. J. Am. Chem. Soc. 128, 11135–11149 (2006).
Kuna, J. J. et al. The effect of nanometre-scale structure on interfacial energy. Nature Mater. 8, 837–842 (2009).
Centrone, A. et al. The role of nanostructure in the wetting behavior of mixed-monolayer-protected metal nanoparticles. Proc. Natl Acad. Sci. USA 105, 9886–9891 (2008).
Zabet-Khosousi, A. & Dhirani, A. A. Charge transport in nanoparticle assemblies. Chem. Rev. 108, 4072–4124 (2008).
Acknowledgements
This work was supported by the Non-equilibrium Energy Research Center, which is an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, Office of Basic Energy Sciences under grant number DE-SC0000989. E.S.C. and F.S. acknowledge the support of ENI within the MIT Energy initiative for their work. E.S.C. is supported by a Samsung Scholarship from the Samsung Foundation of Culture. H.J., S.C.G. and F.S. acknowledge support from the Defense Threat Reduction Agency under Grant No. HDTRA1-09-1-0012. T.M.H. is financially supported by the Human Frontier Science Program. We acknowledge L. Meda for her X-ray photoelectron spectroscopy measurements, and R. Borrelli and P. Cesti for helpful discussions. We also thank the USGS for providing fish samples and for helpful discussions.
Author information
Authors and Affiliations
Contributions
E.S.C. synthesized the striped nanoparticles, performed all of their characterization before and after binding to ions, and helped with some of the solid-state work.; J.K. made and characterized all sensors, performed cation capture experiments, measured the conductivities of all sensors and analysed data; B.T. developed quantum-mechanical models and ran quantum-mechanical calculations; T.M.H. developed and validated the binding/percolation models; H.J. ran and analysed MD simulations; S.C.G. analysed and supervised the MD simulations; H.N. performed initial experiments with NP films; M.Y. performed the STM characterization of the particles; A.Z.P. helped with the theory of percolation phenomena; F.S. and B.A.G. conceived the ideas and supervised the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2196 kb)
Rights and permissions
About this article
Cite this article
Cho, E., Kim, J., Tejerina, B. et al. Ultrasensitive detection of toxic cations through changes in the tunnelling current across films of striped nanoparticles. Nature Mater 11, 978–985 (2012). https://doi.org/10.1038/nmat3406
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3406
This article is cited by
-
Urchin-like magnetic microspheres for cancer therapy through synergistic effect of mechanical force, photothermal and photodynamic effects
Journal of Nanobiotechnology (2022)
-
Transistors and logic circuits based on metal nanoparticles and ionic gradients
Nature Electronics (2021)
-
Study the plasmonic property of gold nanorods highly above damage threshold via single-pulse spectral hole-burning experiments
Scientific Reports (2021)
-
Intriguing Plasmonic and Fluorescence Duality in Copper Nanoparticles
Plasmonics (2020)
-
A review of recent applications of porous metals and metal oxide in energy storage, sensing and catalysis
Journal of Materials Science (2019)