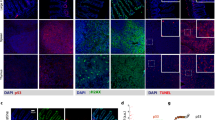
Abstract
The AND gate functions such that when all inputs are activated the downstream gene will be transcribed and it is off otherwise. To accomplish optimal and targeted gene therapy in solid tumor patients, we have constructed an AND gate genetic circuit and investigated whether it could be activated by low-dose radiation in vitro and in vivo. The enhancement green fluorescent protein (EGFP) expression in different tumor cells transfected with control vector plxsn-EGFP confirmed that 2 Gy of radiation and 1% O2 for 3 h could activate our AND gate. Besides, the obvious different levels of EGFP expression between 2 and 6 Gy of radiation demonstrated that the AND gate could be regulated by radiation doses. Additionally, through EGFP expression and the codistribution of p53 and HIF-1α in xenografts, we illustrated the targeted activation property of the AND gate and real-time monitoring to hypoxic districts in vivo. Moreover, significant growth inhibition and cell cycle arrest in vitro and apoptosis-inducing effects in vitro and in vivo proved that the AND gate induced ideal antitumor effects. In conclusion, the radiation dose-regulated AND gate genetic circuit could not only effectively monitor the therapeutic process in real-time but also induce ideal antitumor efficacy, and can be further exploited for personal therapy in clinical tumor patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun JM . Cancer statistics. CA-Cancer J Clin 2009; 59: 225–249.
Hofman V, Bonnetaud C, Ilie MI, Vielh P, Vignaud JM, Fléjou JF et al. Preoperative circulating tumor cell detection using the isolation by size of epithelial tumor cell method for patients with lung cancer is a new prognostic biomarker. Clin Cancer Res 2011; 17: 827–835.
Barnett GC, West CML, Dunning AM, Elliott RM, Coles CE, Pharoah PDP et al. Normal tissue reactions to radiotherapy: towards tailoring treatment dose by genotype. Nat Rev Cancer 2009; 9: 134–142.
Henríquez-Hernández LA, Bordón E, Pinar B, Lloret M, Rodríguez-Gallego C, Lara PC . Prediction of normal tissue toxicity as part of the individualized treatment with radiotherapy in oncology patients. Surg Oncol 2012 (in press).
Abdollahi A, Folkman J . Evading tumor evasion: current concepts and perspectives of anti-angiogenic cancer therapy. Drug Resist Updat 2010; 13: 16–28.
Velez JM, Miriyala S, Nithipongvanitch R, Noel T, Plabplueng CD, Oberley T et al. p53 regulates oxidative stress-mediated retrograde signaling: a novel mechanism for chemotherapy-induced cardiac injury. PLoS ONE 2011; 6: e18005.
Citrin D, Cotrim AP, Hyodo F, Baum BJ, Krishna MC, Mitchell JB . Radioprotectors and mitigators of radiation-induced normal tissue injury. Oncologist 2010; 15: 360–371.
Lo HW, Day CP, Hung MC . Cancer-specific gene therapy. Adv Genet 2005; 54: 235–255.
Pennacchietti S, Michieli P, Galluzzo M, Mazzone M, Giordano S, Comoglio PM . Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 2003; 3: 347–361.
Stewart GD, Nanda J, Katz E, Bowman KJ, Christie JG, Brown DJG et al. DNA strand breaks and hypoxia response inhibition mediate the radiosensitisation effect of nitric oxide donors on prostate cancer under varying oxygen conditions. Biochem Pharmacol 2011; 81: 203–210.
Pawlik TM, Keyomarsi K . Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol 2004; 59: 928–942.
Martin JB . Tumor hypoxia in cancer therapy. Methods Enzymol 2007; 435: 295–321.
Hockel M, Vaupel P . Biological consequences of tumor hypoxia. Semin Oncol 2001; 28: 36–41.
Greco O, Joiner MC, Doleh A, Powell AD, Hillman GG, Scott SD . Hypoxia- and radiation-activated Cre//loxP ‘molecular switch’ vectors for gene therapy of cancer. Gene Ther 2005; 13: 206–215.
Wang W, Chen Z, Li R, Li D, Duan Y, Cao Z . Enhanced efficacy of radiation-induced gene therapy in mice bearing lung adenocarcinoma xenografts using hypoxia responsive elements. Cancer Sci 2005; 96: 918–924.
Greco O, Marples B, Dachs GU, Williams KJ, Patterson AV, Scott SD . Novel chimeric gene promoters responsive to hypoxia and ionizing radiation. Gene Ther 2002; 9: 1403–1411.
Mayr NA, Huang Z, Sohn JW, Lo SS, Teh BS, Lu JJ et al. Emerging application of stereotactic body radiation therapy for gynecologic malignancies. Expert Rev Anticancer 2011; 11: 1071–1077.
Vujaskovic Z, Willett CG, Tepper JE, Kinsella TJ, Gunderson LL . Normal-tissue tolerance to IOERT, EBRT, or both: animal and clinical studies. In: Gunderson LL, Willett CG, Calvo FA, Harrison LB (eds). Current Clinical Oncology, 2nd edn. Humana Press: Totowa, 2011, pp 119–138.
Zelefsky MJ, Levin EJ, Hunt M, Yamada Y, Shippy AM, Jackson A et al. Incidence of late rectal and urinary toxicities after three-dimensional conformal radiotherapy and intensity-modulated radiotherapy for localized prostate cancer. Int J Radiat Oncol 2008; 70: 1124–1129.
Maingon P, Marchesi V, Crehange G . Intensity modulated radiation therapy. Br J Cancer 2010; 97: 759–768.
Yu ST, Li C, Lu MH, Liang GP, Li N, Tang XD et al. Noninvasive and real-time monitoring of the therapeutic response of tumors in vivo with an optimized hTERT promoter. Cancer 2012; 118: 1884–1893.
Asano S . Gene therapy as translational research. Mol Urol 2000; 4: 41.
Chang Q, Jurisica I, Do T, Hedley DW . Hypoxia predicts aggressive growth and spontaneous metastasis formation from orthotopically grown primary xenografts of human pancreatic cancer. Cancer Res 2011; 71: 3110–3120.
Ammoun S, Zhou L, Barczyk M, Hilton D, Hafizi S, Hanemann C et al. Gas6/axl-family receptors in schwannoma pathological proliferation, adhesion and survival. Neuro Oncol 2011; 13: 1–14.
Crosby ME, Kulshreshtha R, Ivan M, Glazer PM . MicroRNA regulation of DNA repair gene expression in hypoxic stress. Cancer Res 2009; 69: 1221–1229.
Avalosse B, Dupont F, Burny A . Gene therapy for cancer. Curr Opin Oncol 1995; 7: 94–100.
Baum C, Dullmann J, Li Z, Fehse B, Meyer J, Williams DA et al. Side effects of retroviral gene transfer into hematopoietic stem cells. Blood 2003; 101: 2099–2113.
Fukazawa T, Matsuoka J, Yamatsuji T, Maeda Y, Durbin ML, Nanomato et al. Adenovirus mediated cancer gene therapy and virotherapy. Int J Mol Med 2010; 25: 3–10.
Wang W, Li R, Chen Z, Li D, Duan Y, Cao Z . Cisplatin-controlled p53 gene therapy for human non-small cell lung cancer xenografts in athymic nude mice via the CArG elements. Cancer Sci 2005; 96: 706–712.
Pines A, Bivi N, Romanello M, Damante G, Kelley MR, Adamson ED et al. Cross-regulation between Egr-1 and APE/Ref-1 during early response to oxidative stress in the human osteoblastic HOBIT cell line: evidence for an autoregulatory loop. Free Radical Res 2005; 39: 269–281.
Acknowledgements
We thank the College of High Altitude Military Medicine of Third Military Medical University for support. This work was supported by the Chongqing Science Fund (CSCT2008BB5024) and the National Natural Science Fund of China (NSFC30670625, 30500144).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ding, M., Zhang, E., He, R. et al. The radiation dose-regulated AND gate genetic circuit, a novel targeted and real-time monitoring strategy for cancer gene therapy. Cancer Gene Ther 19, 382–392 (2012). https://doi.org/10.1038/cgt.2012.11
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2012.11
Keywords
This article is cited by
-
SynBioLGDB: a resource for experimentally validated logic gates in synthetic biology
Scientific Reports (2015)
-
Creation of a gated antibody as a conditionally functional synthetic protein
Nature Communications (2014)