Abstract
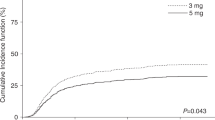
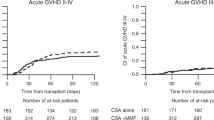
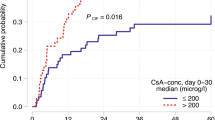
Hematopoietic SCT (HSCT) after non-myeloablative conditioning is associated with reduced TRM, and increased risk of graft rejection. Although preclinical data have shown the importance of post transplant immunosuppression in achieving engraftment, little is known about the role of CSA in the clinical setting of non-myeloablative transplantation. In a retrospective analysis of patients treated with allogeneic HSCT after fludarabine and 2 Gy TBI, 15 of 77 evaluable patients (20%) experienced primary (n=2) or secondary graft rejection at a median of 66 days post transplant. Mean day 1–28 CSA trough levels were inversely associated with day 28 chimerism (median 99, 85 and 70% for mean CSA <300, 300–600 and >600 ng/mL, respectively; P=0.003). A similar association was observed for the cumulative incidence of graft rejection, which occurred in 8% (<300 ng/mL), 26% (300–600 ng/mL) and 50% (>600 ng/mL, P=0.005) of patients. The detrimental effect of high CSA levels on engraftment was confirmed in multivariable models and was found to operate comparably in sibling and unrelated donor transplants. Impairment of donor T-cell function by high serum levels of CSA might account for this finding, which should be verified in a larger patient group to better understand the role of CSA in non-myeloablative transplantation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gratwohl A, Baldomero H, Passweg J, Urbano-Ispizua A . Increasing use of reduced intensity conditioning transplants: report of the 2001 EBMT activity survey. Bone Marrow Transplant 2002; 30: 813–831.
Gratwohl A, Baldomero H, Schwendener A, Rocha V, Apperley J, Frauendorfer K et al. The EBMT activity survey 2007 with focus on allogeneic HSCT for AML and novel cellular therapies. Bone Marrow Transplant 2009; 43: 275–291.
Scott BL, Sandmaier BM, Storer B, Maris MB, Sorror ML, Maloney DG et al. Myeloablative vs nonmyeloablative allogeneic transplantation for patients with myelodysplastic syndrome or acute myelogenous leukemia with multilineage dysplasia: a retrospective analysis. Leukemia 2006; 20: 128–135.
Sandmaier BM, Mackinnon S, Childs RW . Reduced intensity conditioning for allogeneic hematopoietic cell transplantation: current perspectives. Biol Blood Marrow Transplant 2007; 13 (Suppl 1): 87–97.
Niederwieser D, Maris M, Shizuru JA, Petersdorf E, Hegenbart U, Sandmaier BM et al. Low-dose total body irradiation (TBI) and fludarabine followed by hematopoietic cell transplantation (HCT) from HLA-matched or mismatched unrelated donors and postgrafting immunosuppression with cyclosporine and mycophenolate mofetil (MMF) can induce durable complete chimerism and sustained remissions in patients with hematological diseases. Blood 2003; 101: 1620–1629.
de Lima M, Anagnostopoulos A, Munsell M, Shahjahan M, Ueno N, Ippoliti C et al. Nonablative versus reduced-intensity conditioning regimens in the treatment of acute myeloid leukemia and high-risk myelodysplastic syndrome: dose is relevant for long-term disease control after allogeneic hematopoietic stem cell transplantation. Blood 2004; 104: 865–872.
Baron F, Storb R . Allogeneic hematopoietic cell transplantation following nonmyeloablative conditioning as treatment for hematologic malignancies and inherited blood disorders. Mol Ther 2006; 13: 26–41.
Marmont AM, Horowitz MM, Gale RP, Sobocinski K, Ash RC, van Bekkum DW et al. T-cell depletion of HLA-identical transplants in leukemia. Blood 1991; 78: 2120–2130.
Platzbecker U, Ehninger G, Bornhauser M . Allogeneic transplantation of CD34+ selected hematopoietic cells—clinical problems and current challenges. Leuk Lymphoma 2004; 45: 447–453.
Anasetti C, Amos D, Beatty PG, Appelbaum FR, Bensinger W, Buckner CD et al. Effect of HLA compatibility on engraftment of bone marrow transplants in patients with leukemia or lymphoma. N Engl J Med 1989; 320: 197–204.
Maris MB, Niederwieser D, Sandmaier BM, Storer B, Stuart M, Maloney D et al. HLA-matched unrelated donor hematopoietic cell transplantation after nonmyeloablative conditioning for patients with hematologic malignancies. Blood 2003; 102: 2021–2030.
Storb R, Yu C, Wagner JL, Deeg HJ, Nash RA, Kiem HP et al. Stable mixed hematopoietic chimerism in DLA-identical littermate dogs given sublethal total body irradiation before and pharmacological immunosuppression after marrow transplantation. Blood 1997; 89: 3048–3054.
Zaucha JM, Yu C, Zellmer E, Takatu A, Junghanss C, Little MT et al. Effects of extending the duration of postgrafting immunosuppression and substituting granulocyte-colony-stimulating factor-mobilized peripheral blood mononuclear cells for marrow in allogeneic engraftment in a nonmyeloablative canine transplantation model. Biol Blood Marrow Transplant 2001; 7: 513–516.
Yu C, Storb R, Mathey B, Deeg HJ, Schuening FG, Graham TC et al. DLA-identical bone marrow grafts after low-dose total body irradiation: effects of high-dose corticosteroids and cyclosporine on engraftment. Blood 1995; 86: 4376–4381.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation 1974; 18: 295–304.
Georges GE, Maris M, Sandmaier BM, Malone DG, Feinstein L, Niederweiser D et al. Related and unrelated nonmyeloablative hematopoietic stem cell transplantation for malignant diseases. Int J Hematol 2002; 76 (Suppl 1): 184–189.
Bearman SI . Reduced-intensity allogeneic stem cell transplantation. Curr Hematol Rep 2003; 2: 277–286.
Sorror ML, Leisenring W, Mielcarek M, Baron F, Diaconescu R, Hogan WJ et al. Intensified postgrafting immunosuppression failed to assure long-term engraftment of dog leukocyte antigen-identical canine marrow grafts after 1 gray total body irradiation. Transplantation 2008; 85: 1023–1029.
Storb R, Yu C, Barnett T, Wagner JL, Deeg HJ, Nash RA et al. Stable mixed hematopoietic chimerism in dog leukocyte antigen-identical littermate dogs given lymph node irradiation before and pharmacologic immunosuppression after marrow transplantation. Blood 1999; 94: 1131–1136.
Jochum C, Beste M, Zellmer E, Graves SS, Storb R . CD154 blockade and donor-specific transfusions in DLA-identical marrow transplantation in dogs conditioned with 1-Gy total body irradiation. Biol Blood Marrow Transplant 2007; 13: 164–171.
Feinstein L, Sandmaier B, Maloney D, McSweeney PA, Maris M, Flowers C et al. Nonmyeloablative hematopoietic cell transplantation. Replacing high-dose cytotoxic therapy by the graft-versus-tumor effect. Ann N Y Acad Sci 2001; 938: 328–337; discussion 337–9.
Maris MB, Sandmaier BM, Storer BE, Maloney DG, Shizuru JA, Agura E et al. Unrelated donor granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cell transplantation after nonmyeloablative conditioning: the effect of postgrafting mycophenolate mofetil dosing. Biol Blood Marrow Transplant 2006; 12: 454–465.
Hardy NM, Hakim F, Steinberg SM, Krumlauf M, Cvitkovic R, Babb R et al. Host T cells affect donor T cell engraftment and graft-versus-host disease after reduced-intensity hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2007; 13: 1022–1030.
Locatelli F, Zecca M, Rondelli R, Bonetti F, Dini G, Prete A et al. Graft versus host disease prophylaxis with low-dose cyclosporine-A reduces the risk of relapse in children with acute leukemia given HLA-identical sibling bone marrow transplantation: results of a randomized trial. Blood 2000; 95: 1572–1579.
Bacigalupo A, Lamparelli T, Gualandi F, Bregante S, Raiola A, Grazia CD et al. Increased risk of leukemia relapse with high dose cyclosporine after allogeneic marrow transplantation for acute leukemia: 10 year follow-up of a randomized study. Blood 2001; 98: 3174–3175.
Elmaagacli AH, Beelen DW, Trenn G, Schmidt O, Nahler M, Schaefer UW . Induction of a graft-versus-leukemia reaction by cyclosporin A withdrawal as immunotherapy for leukemia relapsing after allogeneic bone marrow transplantation. Bone Marrow Transplant 1999; 23: 771–777.
Keil F, Prinz E, Kalhs P, Lechner K, Moser K, Schwarzinger I et al. Treatment of leukemic relapse after allogeneic stem cell transplantation with cytotoreductive chemotherapy and/or immunotherapy or second transplants. Leukemia 2001; 15: 355–361.
Shiratori S, Yasumoto A, Tanaka J, Shigematsu A, Yamamoto S, Nishio M et al. A retrospective analysis of allogeneic hematopoietic stem cell transplantation for adult T cell leukemia/lymphoma (ATL): clinical impact of graft-versus-leukemia/lymphoma effect. Biol Blood Marrow Transplant 2008; 14: 817–823.
Passweg JR, Meyer-Monard S, Gregor M, Favre G, Heim D, Ebnoether M et al. High stem cell dose will not compensate for T cell depletion in allogeneic non-myeloablative stem cell transplantation. Bone Marrow Transplant 2002; 30: 267–271.
Bader P, Kreyenberg H, Hoelle W, Dueckers G, Handgretinger R, Lang P et al. Increasing mixed chimerism is an important prognostic factor for unfavorable outcome in children with acute lymphoblastic leukemia after allogeneic stem-cell transplantation: possible role for pre-emptive immunotherapy? J Clin Oncol 2004; 22: 1696–1705.
Bader P, Kreyenberg H, Hoelle W, Dueckers G, Kremens B, Dilloo D et al. Increasing mixed chimerism defines a high-risk group of childhood acute myelogenous leukemia patients after allogeneic stem cell transplantation where pre-emptive immunotherapy may be effective. Bone Marrow Transplant 2004; 33: 815–821.
Smak Gregoor PJ, van Gelder T, Hesse CJ, van der Mast BJ, van Besouw NM, Weimar W . Mycophenolic acid plasma concentrations in kidney allograft recipients with or without cyclosporin: a cross-sectional study. Nephrol Dial Transplant 1999; 14: 706–708.
Picard N, Premaud A, Rousseau A, Le Meur Y, Marquet P . A comparison of the effect of cyclosporin and sirolimus on the pharmokinetics of mycophenolate in renal transplant patients. Br J Clin Pharmacol 2006; 62: 477–484.
Acknowledgements
The study was supported by a grant of the Swiss National Research Foundation 3200B0-118176.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Gerull, S., Arber, C., Bucher, C. et al. Cyclosporine levels and rate of graft rejection following non-myeloablative conditioning for allogeneic hematopoietic SCT. Bone Marrow Transplant 46, 740–746 (2011). https://doi.org/10.1038/bmt.2010.187
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2010.187
Keywords
This article is cited by
-
Optimized cyclosporine starting dose may reduce risk of acute GvHD after allogeneic hematopoietic cell transplantation: a single-center cohort study
Bone Marrow Transplantation (2022)
-
Cyclosporine levels > 195 μg/L on day 10 post-transplant was associated with significantly reduced acute graft-versus-host disease following allogeneic hematopoietic stem cell transplantation
Annals of Hematology (2019)
-
Pharmacokinetics, Pharmacodynamics and Pharmacogenomics of Immunosuppressants in Allogeneic Haematopoietic Cell Transplantation: Part I
Clinical Pharmacokinetics (2016)