Abstract
Bacteriophages are promising therapeutic agents that can be applied to different stages of the commercial food chain. In this sense, bacteriophages can be orally administered to farm animals to protect them against intestinal pathogens. However, the low pH of the stomach, the activities of bile and intestinal tract enzymes limit the efficacy of the phages. This study demonstrates the utility of an alginate/CaCO3 encapsulation method suitable for bacteriophages with different morphologies and to yield encapsulation efficacies of ~100%. For the first time, a cocktail of three alginate/CaCO3-encapsulated bacteriophages was administered as oral therapy to commercial broilers infected with Salmonella under farm-like conditions. Encapsulation protects the bacteriophages against their destruction by the gastric juice. Phage release from capsules incubated in simulated intestinal fluid was also demonstrated, whereas encapsulation ensured sufficient intestinal retention of the phages. Moreover, the small size of the capsules (125–150 μm) enables their use in oral therapy and other applications in phage therapy. This study evidenced that a cocktail of the three alginate/CaCO3-encapsulated bacteriophages had a greater and more durable efficacy than a cocktail of the corresponding non-encapsulated phages in as therapy in broilers against Salmonella, one of the most common foodborne pathogen.
Similar content being viewed by others
Introduction
In farm animals and in aquaculture, antibiotics have been used not only in the treatment of infections but also as growth promoters. However, this intensive use of antibiotics has given rise to the emergence of resistant strains of commensal and pathogenic microorganisms that are able to spread to humans, either directly through the food chain or indirectly via the environmental pollution caused by farm effluents1. The European Food Safety Authority (EFSA) has played an essential role in monitoring and detecting the risks posed by the emergence of multi-drug resistant bacteria within the food industry2. As an alternative to the use of antibiotics, bacteriophages are promising therapeutic agents due to their ubiquity, high specificity, replication capacity inside their host, and their ease of isolation and production. The use of bacteriophage-based products in different stages of the food chain3,4 as well as in humans5,6 and animals4,7 via different routes of administration has been investigated. However, a challenge of oral phage therapy is that the phages must overcome numerous adverse conditions, including the low pH of the stomach, the activity of bile, and the activities of enzymes in the digestive tract8,9,10 and also their short residence time in the digestive tract11. In addition, when used in food protection bacteriophages may interact with food components and be exposed to unfavourable conditions or their stability during food processing and storage may be compromised. These problems can be successfully addressed by encapsulation, which would be able to protect bacteriophages under similar potentially deleterious conditions. Among the many diverse biomaterials used for phage encapsulation are cellulose, liposomes, alginate, whey proteins, and gelatin, which have been applied using different techniques12,13. Alginate is one of the most common biomaterials used in microencapsulation, including that of probiotics14,15 and, as recently shown, bacteriophages. The latter was achieved using alginate either alone or in combination with other components, in which wet capsules ranging in size from 310 μm to almost 1 mm were obtained8,10,16,17,18. Alginate, a linear copolymer containing blocks of (1, 4)-linked β-D-mannuronate and α-L-guluronate residues, is of low toxicity and immunogenicity and thus regarded as a biocompatible material19. When used as a hydrogel in biomedical applications19, alginate is combined with ionic cross-linking agents, such as divalent cations. In oral bacteriophage therapy, it protects the phage by shrinking at low pH values, dissolving at high pH values8, and participating in mucoadhesive interactions with gastrointestinal mucus20. These results were obtained from in vitro studies that examined the interactions of alginate-encapsulated bacteriophages with simulated gastric and intestinal fluids. By contrast, the use of alginate-encapsulated bacteriophages in oral phage therapy has been tested in only a few in vivo experiments involving Salmonella in pigs21,22. To our knowledge, the present study is the first in which wet bacteriophage-containing alginate/CaCO3 microcapsules <150 μm in size were obtained and then used as oral therapy to protect broiler chickens against Salmonella infection, under conditions mimicking those of poultry production farms. In particular, a cocktail composed by three virulent bacteriophages (UAB_Phi20, UAB_Phi78, and UAB_Phi87) was prepared according to previous promising results of phage-therapy studies11,23,24.
Results
Characterization of alginate/CaCO3-encapsulated phages
The three bacteriophages were encapsulated separately into alginate/CaCO3 and their properties then examined. The mean size of alginate-encapsulated UAB_Phi20, UAB_Phi78, and UAB_Phi87 as determined by laser diffraction was 124 ± 9 μm, 141 ± 16 μm, and 149 ± 6 μm, respectively. The alginate/CaCO3-encapsulated bacteriophages were then characterized by labelling the phage and the alginate with SYBR gold and DAPI, respectively. The 3D spatially superimposed SYBR-gold (green) and DAPI- (blue) fluorescence intensities are shown in Fig. 1. The images show that all of the phages were properly encapsulated in the capsules. This was confirmed by the encapsulation yields of 98.0% ± 1.5 for UAB_Phi20, 99.0% ± 0.1 for UAB_Phi78, and 99.0% ± 0.7 for UAB_Phi87 (Table 1).
3D confocal microscopy images of SYBR-gold-labelled UAB_Phi20 (a), UAB_Phi78 (b), and UAB_Phi87 (c) (green) encapsulated in DAPI-labelled alginate capsules (blue). 3D images of the capsules are shown on the left (1), and the corresponding cross-sectional images in the middle (2) and on the right (3). Scale bars, 30 μm.
The stability of the alginate/CaCO3-encapsulated phages was determined by storing freshly encapsulated phages at 4 °C for 6 months and then again measuring the percentage of encapsulated phages. As shown in Table 1, for cold-stored UAB_Phi20 and UAB_Phi78 there was only a slight decrease in the encapsulation percentage compared to the values of freshly encapsulated phages, whereas for cold-stored encapsulated UAB_Phi87 phage there was no loss of encapsulated phage. Similarly, no decrease in the encapsulation percentage was observed after maintaining the alginate/CaCO3-encapsulated phages two weeks at room temperature (data not shown).
Stability of the encapsulated bacteriophages in simulated gastric fluid
The acid stability of the alginate/CaCO3-encapsulated phages was tested by incubating them in simulated gastric fluid (SGF) (pH 2.8) for 60 min. These phages proved to be much more stable under acidic conditions than their non-encapsulated counterparts (Fig. 2) (p < 0.05). Thus, titre losses of phage UAB_Phi78 and phage UAB_Phi87 were 3.1 and 2.4 log10 after 30 min and 2.9 and 3.5 log10 after 60 min of incubation, respectively. Remarkably, there was no reduction in the titre of UAB_Phi20 after 60-min incubation with SGF.
Encapsulated phage release by simulated intestinal fluid
The kinetics of phage release from alginate/CaCO3 capsules incubated for up to 40 min with simulated intestinal fluid (SIF) was also assessed. After 20 min of incubation in SIF, the percentage of UAB_Phi20, UAB_Phi78, and UAB_Phi87 release was 79.0% ± 9.1, 60.7% ± 2.4, and 95.6% ± 7.6, respectively (Fig. 3). After 40 min, release was nearly complete for all phages (97.7% ± 18.6, 88.4% ± 7.6, and 100.0% ± 20.8, respectively; Fig. 3).
In vivo retention of bacteriophage in the chicken caecum
Previously to do the studies of retention and phage therapy in chicken, we observed that alginate/CaCO3 capsules without phages were innocuous in a mouse model (data not shown).
The caecal residence time of a cocktail of the three non-encapsulated and alginate/CaCO3-encapsulated phages differed significantly at all three time points tested. Thus, after 2 h the encapsulated phages were retained in 95.2% of the chickens and the non-encapsulated phages in 57.1% (p < 0.05; Fig. 4). After 48 and 72 h the differences were still significant (p < 0.001): 95.2% vs. 38.1%, and 71.4% vs. 9.5%, respectively (Fig. 4).
Bacteriophage therapy against Salmonella infection in broiler chickens
Cocktails prepared from non-encapsulated and alginate/CaCO3-encapsulated phages differed in their long-term efficacies in protecting commercial broiler chickens against Salmonella infection, as determined based on the caecal Salmonella concentration (Table 2). Thus, within the first 6 days post-infection, reductions in the Salmonella concentration in the two treated groups were nearly the same and both were significant compared to the control untreated chickens (p < 0.001). However, on day 1 post-infection, a greater reduction of Salmonella counts was obtained with the non-encapsulated than the encapsulated phages (p < 0.05). The reduction in the Salmonella concentration in chickens administered the cocktail of non-encapsulated phages was significant until day 8, the first day after treatment cessation (1.5 log10 reduction; p < 0.05). By contrast, the protection achieved with the cocktail of alginate/CaCO3-encapsulated phages was significant vs. the control from de beginning until day 15 post-infection (days 8 and 10: p < 0.001; day 15: p < 0.05), with reductions in Salmonella counts on days 8, 10, and 15, of 3.6, 3.4, and 1.7 log10, respectively. A further comparison of the two phage treatments showed that the reduction achieved with the cocktail of alginate/CaCO3-encapsulated phages was significantly greater than that obtained with the non-encapsulated phages from day 8 until the end of the experiment (days 8 and 10: p > 0.001; day 15: p > 0.05; Table 2). It must be mentioned that we did not find any bacteria isolated from phage-treated groups to be resistant against the three phages of the cocktail administered (manuscript in preparation).
Bacteriophage concentrations remained stable throughout the experiment and were slightly higher in the non-encapsulated (4.2–4.4 log10 pfu/g of caecum) than in the alginate-encapsulated (3.8–3.0 log10 pfu/g of caecum) group from day 1 to day 10 post-infection (Table 3). However, the concentration in the caecum of the non-encapsulated group was significantly greater (5.1 log10 pfu/g; p < 0.001) than in the alginate-encapsulated (0.2 log10 pfu/g) on day 15. It must be noted that the presence of the three bacteriophages in caecum samples could be demonstrated due to their different plaque morphology (Fig. 5).
Discussion
Methods allowing the stable and controlled delivery of bacteriophages are of great value in phage therapy. One such method is encapsulation, which in farm animals protects orally administered bacteriophages from the harsh environment of the stomach and facilitates their retention during passage through the intestinal tract to ensure a successful therapeutic effect24,25,26. By maintaining bacteriophage stability, micro- or nano-encapsulation enables not only their oral administration through feed or water but also their administration in other forms, such as inhalation, thereby assuring an adequate dose of the therapeutic phage. Furthermore, encapsulation can overcome other problems related to the application of bacteriophages in food industry processes. The materials used in bacteriophage encapsulation have been examined in several studies12,13 and include alginate, alone or in combination with other materials8,10,16,17,18. However, few studies have described the in vivo use of alginate-encapsulated bacteriophages21,22,27. Thus, our study is the first to test a cocktail of three alginate/CaCO3-encapsulated virulent bacteriophages (UAB_Phi20, UAB_Phi78, and UAB_Phi87)11,23 as oral therapy in Salmonella-infected poultry under farm-like conditions. Phages prepared according to this method were shown to be effective in protecting broilers against infection for up to 15 days.
The encapsulation methodology described herein allows the encapsulation of bacteriophages with different morphologies, without jeopardizing infectivity. The encapsulation efficiency values obtained in this study were ~99%, similar to the percentages reported by other authors8,10,16,17,25. Moreover, the alginate/CaCO3 encapsulated bacteriophages showed excellent stability when stored at 4 °C for 6 months, with minor losses determined only for UAB_Phi20 and UAB_Phi78. Also, the encapsulated phages were stable at room temperature at least for two weeks, period of time that is sufficient for the administration to animals in drinking water.
Another promising feature of the alginate/CaCO3 microcapsules was their size (124–149 μm), which was almost ten times smaller than other types of capsules described in the literature8,10,16,17,25 and facilitated their potential commercial applications. All this is the result of various systematic studies aimed at optimizing the alginate concentration (1.8%) and the posterior curing time of the capsules in the bath of CaCl2 (90 min).
Phages orally administered to broilers must withstand the low pH of the stomach contents of the chickens, which is typically in the range of 2.1–3.628,29. Under acidic conditions, the alginate/CaCO3-encapsulated phages proved to be much more stable than their non-encapsulated counterparts (Fig. 2) (p < 0.05), resulting in titre losses after 60 min of incubation in SGF that were around 5 times lower for phages UAB_Phi78 and UAB_Phi87. There was no loss of encapsulated UAB_Phi20. It is reported that the incorporation of CaCO3 slows the gelation rate of alginate capsules, whereas CO3− ions dissociated from CaCO3 diffuse into the medium and slightly increase the pH as a role of antacid, thus protecting the bacteriophages10,19. Our results are in accordance with those obtained by10, who also reported a good protection effect on the bacteriophage K once encapsulated in a mixture of alginate/CaCO3 capsules of a diameter of ~900 μm. However, in our case, this protection effect was achieved even lowering the size of the capsules down to ~150 μm of size.
Another important feature of alginate/CaCO3-encapsulated bacteriophages is their release in the animal intestine, where the host pathogen is located. In this study, the three phages exhibited slightly different in vitro release kinetics. Thus, whereas almost a complete release of bacteriophage UAB_Phi87 was observed after 20 min of incubation in SIF, UAB_Phi20 and UAB_Phi78 were released more slowly, being this release completed after 40 min (Fig. 3). A potential explanation of the faster release of UAB_Phi87 is that this phage has a larger size than the other two phages, which could provoke the formation of alginate capsules with a lower reticular or cross-linked structure and therefore, a faster release of the encapsulated phages. In addition, a comparison of our release results with those of other authors is difficult, since the size and composition of the microcapsules, the composition of the SIF and the incubation conditions (pH and temperature) differed8,10,16,18.
However, bacteriophage release kinetics will undoubtedly differ in vivo. Currently, whether phages adhere to the intestinal epithelium30 or their presence becomes insignificant in the absence of the bacterial host is unclear11,31. The mucoadhesive properties of alginate32 could prolong the presence and the effect of bacteriophages used in oral phage therapy. Our in vivo results of residence time demonstrate that alginate/CaCO3 encapsulation enables the significant intestinal retention of the bacteriophages even in the absence of host. Thus, bacteriophages were detected in 71.4% of the chickens 72 h after oral administration of a single dose of the cocktail of encapsulated bacteriophages compared to 9.5% of the chickens treated with the non-encapsulated phages (p < 0.001). The percentage obtained with the alginate/CaCO3 capsules was better than obtained in a previous in vivo study of liposome-encapsulated bacteriophages24, perhaps due to the higher encapsulation efficiency achieved with the former.
The cocktail composed of the three alginate/CaCO3-encapsulated bacteriophages demonstrated long-term efficacy when used in commercial broilers chickens infected with Salmonella, mimicking real farm conditions. Colonization by Salmonella was effectively reduced by administering a phage cocktail composed of alginate/CaCO3-encapsulated bacteriophages to the poultry 1 day prior to infection with the bacterium and for an additional 7 days during treatment. Moreover, the protective effect was significantly maintained for 1 week after treatment was stopped (on day 15-post infection). Differently, the non-encapsulated bacteriophages quickly loosed their effect once the treatment was stopped. During the first 6 days post-infection, the encapsulated and non-encapsulated cocktails were of similar efficacies, with maximum reductions in Salmonella counts of 3.1 and 2.8 log10, respectively. Only on the first day of treatment was the non-encapsulate cocktail significantly more effective (reduction of 2.9 vs. 1.3 log10; p < 0.05; Table 2). This may have reflected the additional time required for the encapsulated phages to accumulate to an effective therapeutic concentration following their release. Then, it is likely that the phage release in vivo would be slower than in vitro SIF studies. However, the effect of the non-encapsulated cocktail was abolished 1 day after the cessation of treatment (day 7 post-infection) whereas the encapsulated cocktail maintained its effectiveness until the end of the experiment.
This is the first study to report the use of alginate/CaCO3-encapsulated bacteriophages as an in vivo oral therapy against Salmonella infections in poultry. While our results are similar to those achieved with liposome encapsulation24, preparation of the alginate/CaCO3 capsules is simpler and provides much higher encapsulation rates. Although treatment with the alginate/CaCO3 cocktail did not remove Salmonella totally, the concentration of Salmonella used in the experimental poultry infection was very high (~6 log10/g of caecum). Further studies should seek to ascertain the threshold Salmonella concentration in poultry farming and if the encapsulated cocktail could eliminate Salmonella from the chickens completely in those conditions.
Another important aspect in the success of phage therapy is the relationship between the phages and their bacterial hosts. According to the literature on bacteriophage-bacterial dynamics in vitro, the number of phage increases only when the cell density is sufficient, so that the probability of an encounter between the bacteriophages and the bacteria and the subsequent infection of the latter exceeds the probability of phage death33. Therefore, the success of phage therapy is largely determined by the relationship between the concentration of the bacteriophages and that of their bacterial host34. However, the in vivo dynamics are presumably much more complex because multiple external factors (e.g., rapid clearance of the bacteriophages by passive/active host immunity, spatial refuges, and intestinal mucous) influence treatment success35.
In our in vivo study, the dynamics of the non-encapsulated and encapsulated bacteriophages in controlling Salmonella differed which agree with a previous study performed by us with liposome-encapsulated bacteriophages24. Thus, with the daily administration of non-encapsulated bacteriophages, their uptake together with the new phage progeny produced in the intestinal tract led to a marked decrease in the Salmonella concentration (~50%) 1 day post-infection. Thereafter, until almost day 8 post-infection, the Salmonella concentration increased slightly but with significant therapeutic effect. Once the phage uptake was stopped, the equilibrium between phages and bacteria was disrupted and the Salmonella concentration increased significantly with respect to day 1 post-infection (days 8 and 10, p < 0.05; day 15 post-infection, p < 0.001; Table 2). The requirement for continuous administration of bacteriophages along the time to achieve a low population of Samonella has also been suggested by other authors11,36. Several factors as partial emergence of bacteriophage-resistant Salmonella, bacterial phenotypic changes, physical refuges or slow growth rate of bacterial cells could explain this fact35,36. By contrast, when the encapsulated bacteriophage cocktail was administered, the Salmonella concentration decreased gradually during all the experiment, regardless of whether treatment was ongoing or had stopped. Therefore, the encapsulation of bacteriophages abolished the need for a continuous treatment to achieve a low bacterial concentration. In this case, the mucoadhesiveness of the capsules and the release kinetics of the bacteriophages from them must be the most important features for this effect. It is remarkable that when the bacteriophage uptake was stopped the bacteriophage concentration remained nearly constant (Table 3) in both treatments, but the Salmonella concentration in the gut was higher in the non-encapsulated bacteriophage treatment than in encapsulated one. At this respect, it has been proposed that there is a threshold density of bacteria that must be present in order for the bacteriophage concentration to increase33. Further works are needed to identify the in vivo mechanism(s) underlying this fact.
In summary, this study demonstrated the utility of a simple, efficient, and inexpensive encapsulation method that can be used with bacteriophages of different morphologies. The small size of the resulting microcapsules (124–149 μm) enables their use in diverse applications in phage therapy. Moreover, alginate/CaCO3 encapsulation confers excellent protection against the deleterious effects of gastric juice and promotes greater intestinal retention of the bacteriophages. The results presented here, together with those from our previous investigation of liposome-encapsulated bacteriophages24, show that encapsulation is important for a prolonged and successful oral phage therapy in commercial broilers infected with Salmonella.
Methods
Bacterial strains and growth conditions
Phages UAB_Phi20, UAB_Phi78, and UAB_Phi87 were previously obtained by us from chicken and pig farms11,23. All three bacteriophages belong to the Caudovirales order. UAB_Phi20 and UAB_Phi78 bacteriophages are members of the Podoviridae family with icosahedral heads (60 ± 1.5 nm and 66 ± 1.7, respectively) and short tails (13–14 nm) whereas UAB_Phi87 has also an icosahedral head (68 ± 2.7 nm) but a long contractile tail (114 ± 4.3 nm) and derives from the Myoviridae family11. The genomic features of the three bacteriophages have been recently published37. The bacteriophages were propagated in Salmonella Typhimurium LB5000 (SGSC181; University of Calgary). Broiler chickens were colonized with S. Typhimurium ATCC 14028 RifR. All bacterial strains were grown in Luria-Bertani (LB) broth or on LB agar plates for 18 h at 37 °C. S. Typhimurium ATCC 14028 RifR viable counts were obtained after incubating the bacterium, plated on xylose-lysine-deoxycholate (XLD) plates (Laboratorios Conda, Spain) supplemented with rifampin (75 μg/ml), at 37 °C for 18 h.
In vitro multiplication of bacteriophages
High-titer (1 × 1011–1 × 1012 pfu/ml in 10 mM MgSO4) lysates were obtained from bacteriophages propagated in S. Typhimurium strain LB5000 (SGSC181; University of Calgary) and subjected to ultracentrifugation at 51,000 × g for 2 h (OptimaTM L-80; Beckman, CA, USA), as previously described24. The phage titre was determined by plating serial dilutions (1:10) onto LB plates using the double agar layer method38.
Bacteriophage encapsulation with alginate/CaCO3
Encapsulation of the phages using alginate/CaCO3 was carried out followed a previously described protocol with slight modifications10. Briefly, 500 mg of CaCO3 (1%) and 900 mg of alginate (1.8%) were added to 50 ml of each bacteriophage suspension in 10 mM MgSO4 at a concentration of 1 × 1011 pfu/ml. This mixture was stirred overnight to allow its proper homogenization and then pumped at a rate of 1.5 ml/min into a bath containing 150 ml of 1.8% CaCl2 using the ViscoMistTM Air AtomiZing spray nozzle (inner diameter: 381 μm; Lechler Inc., St. Charles, IL) under a continuous flow of nitrogen at a pressure of 3 bars. The gelled capsules were left to harden in the bath for 90 min with slow stirring and then centrifuged at 469 × g for 5 min. The resulting pellet was washed thrice with 10 mM MgSO4 and resuspended in 10 mM MgSO4 at a final volume of 50 ml.
Particle size distributions were determined by granulometric assays based on laser diffraction (Mastersizer 2000, Malvern Instruments, UK), which was appropriate because of the micrometric size of the encapsulated phages. Laser diffraction is a well-established technique to determine particle size distributions covered by ISO13320 (2009)39, which measures the angular variation in intensity of light scattered as a laser beam passes through a dispersed particulate sample. The angular scattering intensity data is then analyzed to calculate the size of the particles responsible for creating the scattering pattern, using the Mie theory of light scattering. The mean particle size is reported as a volume equivalent sphere diameter and expressed in terms of the “volume weighted mean”, as the  , where the diameter (d) has d4 dependence and the number of particles is not inherent in the formulae. The D[4, 3] value was calculated using an algorithm from the Mastersizer Software. The undiluted capsules were measured directly after encapsulation and the D[4, 3] value is the media of three different measurements.
, where the diameter (d) has d4 dependence and the number of particles is not inherent in the formulae. The D[4, 3] value was calculated using an algorithm from the Mastersizer Software. The undiluted capsules were measured directly after encapsulation and the D[4, 3] value is the media of three different measurements.
The encapsulation efficiency (EE) of the phages was calculated as EE (%) = 100 − (Cfree/Ctotal) × 100, where Ctotal is the concentration of all bacteriophages and Cfree the concentration of free phages24. To determine Ctotal, the product of the phage encapsulation was titrated directly. Because divalent ions are essential for the stability of the alginate capsules, degradation of the plated capsules may occur due to the sequestration of divalent ions during gelling of the double agar layer. To quantify Cfree, 0.5 ml of the encapsulation product was filtered through a 0.22-μm PES filter syringe to retain the encapsulated phages. The eluted volume was then titered. The stability of the alginate-encapsulated phages at 4 °C was tested for 6 months using the above-described method for EE determination.
Microscopy
The phages were labeled with SYBR gold (Molecular Probes, OR, USA) as previously described24, and encapsulated as described above, but with the addition of 4′,6-diamidino-2′-phenylindole dihydrochloride (DAPI)-labeled alginate to the mixture to a final volume of 10%. Alginate labeling was performed as previously described, but using DAPI rather than fluoresceinamine40. Samples of 30 μl were observed using a Leica TCS SP5 confocal microscope (Leica Microsystems, Germany) but without the resonance scanning mode, as the capsules were large enough to reduce Brownian motion.
Bacteriophage stability in simulated gastric fluid
The stability of the encapsulated phages in SGF (pH 2.8), consisting of 3 mg pepsin (Sigma-Aldrich, MO, USA)/ml in 0.85% NaCl, was tested as previously described24. However, in this study, the solution exhibited a buffering effect till a pH value of 3.8. The bacteriophage titer was determined as described above using aliquots taken at 0, 30, and 60 min of incubation in SGF.
Alginate/CaCO3-encapsulated phage release in simulated intestinal fluid
Phage release from alginate capsules in SIF (pH 8.0), consisting of 1 mg pancreatin (Sigma-Aldrich, MO, USA)/ml, 10 mM bile salts, and 0.85% NaCl41, was determined. Thus, a suspension of alginate-encapsulated phages at a concentration of 1 × 109 pfu/ml was inoculated in SIF and incubated in a water bath at 42 °C with agitation to emulate the conditions of the avian intestine. Ctotal and Cfree were calculated as described above.
In vivo assays in broilers
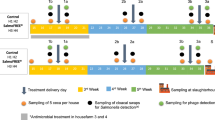
Both the residence time in the chicken intestine and the effectiveness of a cocktail of alginate-encapsulated or non-encapsulated phages against Salmonella was evaluated in vivo in commercial broilers (Gallus gallus, Ross strain 308; Terra-Avant S.A., Girona, Spain), using an experimental model that mimics farm conditions, as described in a previous study24. All in vivo animal experiments were performed in strict compliance with the protocols approved by the Comissió d’Ètica en l’Experimentació Animal i Humana (CEEAH) of the Universitat Autònoma de Barcelona (UAB). The approved study was assigned with the authorization number 1953.
Caecum samples were collected from two euthanized chickens and tested using previously described enrichment protocols24, to confirm that they were free of Salmonella and phages.
A curved oral-dosing needle was used to orally administer the broilers 100 μl of a cocktail containing either the alginate-encapsulated or non-encapsulated phages24. The cocktail consisted of a 1:1:1 mixture of the three phages (UAB_Phi20, UAB_Phi78, and UAB_Phi87) at a concentration of 1011 pfu/ml in MgSO4 buffer without antacid.
Bacteriophage retention in the chicken caecum
The intestinal residence time of the cocktail of either the encapsulated or non-encapsulated phages was determined over 72 h. Two independent experiments were conducted with two groups of 63 1-day-old chickens. The phage cocktail was administered at a dose of 1010 pfu/animal as described previously24. After 2, 48, and 72 h, caecum samples were collected from 21 euthanized animals and processed as previously described24. The total phage number in the homogenized caecum samples was determined as described above. When direct detection of the phages was not possible, an enrichment procedure was carried out using a previously described method24.
Bacteriophage therapy
Bacteriophage therapy against Salmonella was evaluated over 17 days in chickens orally administered the different phage cocktails, as described in a previous work24. Briefly, the animals were orally infected on day 0 with a suspension of S. Typhimurium ATCC14028 RifR (107 cfu/animal). Three sequential experiments with groups of 84 commercial broilers (Gallus gallus, Ross strain 308) each were conducted. Group 1 corresponded to the Salmonella colonization control. Groups 2 and 3 received the non-encapsulated and alginate-encapsulated phages, respectively. In all cases, the oral dose of the three-phage cocktail (1010 pfu/animal) was administered once daily for 9 days (from day −1 to day 7 after Salmonella infection). The control group was orally inoculated with MgSO4 (10 mM). To quantify the Salmonella and the phages, 14 chickens per group were euthanized on days 1, 3, 6, 8, 10, and 15 post-infection. Sample processing and determination of the Salmonella concentration in caecum samples were done as previously described24. The total concentration of bacteriophages was determined as described above. The reduction in the number of bacteria in each treatment was calculated by subtracting the mean caecal concentration (expressed in log 10 units) in groups 2 and 3 from the mean value of the control (group 1).
Statistical analysis
All results were analyzed using IBM SPSS software. For normally distributed samples, an analysis of variance (ANOVA) and Student’s t test were applied; in cases of a non-normal distribution, the Kruskal-Wallis and Mann-Whitney tests were used.
Additional Information
How to cite this article: Colom, J. et al. Microencapsulation with alginate/CaCO3: A strategy for improved phage therapy. Sci. Rep. 7, 41441; doi: 10.1038/srep41441 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Liebana, E. et al. Public health risks of enterobacterial isolates producing extended-spectrum beta-lactamases or AmpC beta-lactamases in food and food-producing animals: an EU perspective of epidemiology, analytical methods, risk factors, and control options. Clin. Infect. Dis. 56, 1030–1037, doi: 10.1093/cid/cis1043 (2013).
European Centre for Disease Prevention and Control (ECDC), European Food Safety Authority (EFSA), European Medicines Agency (EMEA). Joint opinion on antimicrobial resistance (AMR) focused on zoonotic infections. EFSA Journal 7(11), 1372, doi: 10.2903/j.efsa.2009.1372 (2009).
Sillankorva, S. M., Oliveira, H. & Azeredo, J. Bacteriophages and their role in food safety. Int. J. Microbiol. 2012, 863945, doi: 10.1155/2012/863945 (2012).
Endersen, L. et al. Phage therapy in the food industry. Annu. Rev. Food Sci. Technol. 5, 327–349, doi: 10.1146/annurev-food-030713-092415 (2014).
Abedon, S. T., Kuhl, S. J., Blasdel, B. G. & Kutter, E. M. Phage treatment of human infections. Bacteriophage 1, 2, 66–85, doi: 10.4161/bact.1.2.15845 (2011).
Vandenheuvel, D., Lavigne, R. & Brüssow, H. Bacteriophage Therapy: advances in formulation strategies and human clinical trials. Annu. Rev. Virol. 2, 599–618, doi: 10.1146/annurev-virology-100114-054915 (2015).
Ryan, E. M., Gorman, S. P., Donnelly, R. F. & Gilmore, B. F. Recent advances in bacteriophage therapy: how delivery routes, formulation, concentration and timing influence the success of phage therapy. J. Pharm. Pharmacol. 63, 1253–1264, doi: 10.1111/j.2042-7158.2011.01324.x (2011).
Ma, Y. et al. Microencapsulation of bacteriophage Felix O1 into chitosan-alginate microspheres for oral delivery. Appl. Environ. Microbiol. 74, 4799–4805, doi: 10.1128/AEM.00246-08 (2008).
Wang, Q. & Sabour, P. M. Encapsulation and controlled release of bacteriophages for food animal production. In Bacteriophages In The Control Of Food- And Waterbone Pathogens. (eds Parviz, M., Sabour, P. M. & Mansel, W., Griffiths, M. W. ) (ASM Press, Washington, DC, 2010).
Ma, Y. et al. Enhanced alginate microspheres as means of oral delivery of bacteriophage for reducing Staphylococcus aureus intestinal carriage. Food Hydrocoll. 26, 434–440, doi: 10.1016/j.foodhyd.2010.11.017 (2012).
Bardina, C., Spricigo, D. A., Cortés, P. & Llagostera, M. Significance of the bacteriophage treatment schedule in reducing Salmonella colonization of poultry. Appl. Environ. Microbiol. 78, 6600–6607, doi: 10.1128/AEM.01257-12 (2012).
Choinska-Pulit, A., Mituła, P., Sliwka, P., Łaba, W. & Skaradzinska, A. Bacteriophage encapsulation: trends and potential applications. Trends Food Sci. Tech. 45, 212e221, doi: 10.1016/j.tifs.2015.07.001 (2015).
Hussain, M. A. et al. Use of encapsulated bacteriophages to enhance farm to fork food safety. Crit. Rev. Food Sci. Nutr. 11, 0, doi: 10.1080/10408398.2015.1069729 (2015).
Kailasapathy, K. Microencapsulation of probiotc bacteria: technology and potenctial applications. Curr. Issues Intest. Microbiol. 3, 39–48 (2002).
D’Orazio, G. et al. Microencapsulation of new probiotic formulations for gastrointestinal delivery: in vitro study of assess viability and biological properties. Appl. Microbiol. Biotechnol. 99, 9779–9789, doi: 10.1007/s00253-015-6853-1 (2015).
Tang, Z. et al. Whey protein improves survival and release characteristics of bacteriophage FelixO1 encapsulated in alginate microspheres. Food Res. Int. 52, 460–466, doi: 10.1016/j.foodres.2012.12.037 (2013).
Tang, Z., Huang, X., Sabour, P. M., Chambers, J. R. & Wang, Q. Preparation and characterization of dry powder bacteriophage K for intestinal delivery through oral administration. LWT-Food Sci. Technol. 60, 263–270, doi: 10.1016/j.lwt.2014.08.012 (2015).
Samtlebe, M. et al. Carrier systems for bacteriophages to supplement food systems: Encapsulation and controlled release to modulate the human gut microbiota. LWT - Food Sci. Technol. 68, 334–340, doi: 10.1016/j.lwt.2015.12.039 (2016).
Lee K. Y. & Mooney D. J. Alginate: properties and biomedical applications. Prog. Polym. Sci. 37(1), 106–126, doi: 10.1016/j.progpolymsci.2011.06.003 (2012).
Sosnik, A. Alginate particles as platform for drug delivery by the oral route: state-of-the-art. ISRN Pharm. 2014, 926157, doi: 10.1155/2014/926157 (2014).
Wall, S. K., Zhang, J., Rostagno, M. H. & Ebner, P. D. Phage therapy to reduce preprocessing Salmonella infections in market-weight swine. Appl. Environ. Microbiol. 76, 48–53, doi: 10.1128/AEM.00785-09 (2010).
Saez, A. C., Zhang, J., Rostagno, M. H. & Ebner, P. D. Direct feeding of microencapsulated bacteriophages to reduce Salmonella colonization in pigs. Foodborne Pathog. Dis. 8, 1269–1274, doi: 10.1089/fpd.2011.0905 (2011).
Spricigo, D. A., Bardina, C., Cortés, P. & Llagostera, M. Use of a bacteriophage cocktail to control Salmonella in food and the food industry. Int. J. Food Microbiol. 165, 169–174, doi: 10.1016/j.ijfoodmicro.2013.05.009 (2013).
Colom, J. et al. L Liposome-encapsulated bacteriophages for enhanced oral phage therapy against Salmonella spp. Appl. Environ. Microbiol. 81, 4841–4849, doi: 10.1128/AEM.00812-15 (2015).
Dini, C., Islan, G. A., de Urraza, P. J. & Castro, G. R. Novel biopolymer matrices for microencapsulation of phages: enhanced protection against acidity and protease activity. Macromol Biosci 12, 1200–1208, doi: 10.1002/mabi.201200109 (2012).
Dini, C., Islan, G. A. & Castro, G. R. Characterization and stability analysis of biopolymeric matrices designed for phage-controlled release. Appl. Biochem. Biotechnol. 174, 2031–2047, doi: 10.1007/s12010-014-1152-3 (2014).
Stanford, K. et al. Oral delivery systems for encapsulated bacteriophages targeted at Escherichia coli O157:H7 in feedlot cattle. J. Food Prot. 7, 1304–1312 (2010).
Jiménez-Moreno, E., González-Alvarado, J. M., Gónzalez-Sánchez, D., Lázaro, R. & Mateos, G. G. Effects of type and particle size of dietary fiber on growth performance and digestive traits of broilers from 1 to 21 days of age. Poult. Sci. 89, 2197–2212, doi: 10.3382/ps.2010-00771 (2010).
Sacranie, A. et al. The effect of insoluble fiber and intermittent feeding on gizzard development, gut motility and performance of broiler chickens. Poult. Sci. 91, 693–700, doi: 10.3382/ps.2011-01790 (2012).
Barr, J. J. et al. Bacteriophage adhering to mucus provide a non-host-derived immunity. Proc. Natl. Acad. Sci. USA 110, 10771–1776, doi: 10.1073/pnas.1305923110 (2013).
Atterbury, R. J. et al. Bacteriophage therapy to reduce Salmonella colonization of broiler chickens. Appl. Environ. Microbiol. 73, 4543–4549, doi: 10.1128/AEM.00049-07 (2007).
George, M. & Abraham, T. E. Polyionic hydrocolloids for the intestinal delivery of protein drugs: Alginate and chitosan -a review. J. Control Release 114, 1–14, doi: 10.1016/j.jconrel.2006.04.017 (2006).
Payne, R. J. & Jansen, V. A. Understanding bacteriophage therapy as a density-dependent kinetic process. J. Theor. Biol. 208, 37–48, doi: 10.1006/jtbi.2000.2198 (2001).
Payne, R. J. & Jansen, V. A. Pharmacokinetic principles of bacteriophage therapy. Clin. Pharmacokinet. 42, 315–325, doi: 10.2165/00003088-200342040-00002 (2003).
Bull, J. J. & Gill, J. J. The habits of highly effective phages: population dynamics as a framework for identifying therapeutic phages. Front. Microbiol. 5, 618, doi: 10.3389/fmicb.2014.00618 (2014).
Hurley, A., Maurer, J. J. & Lee M. D. Using bacteriophages to modulate Salmonella colonization of the chicken’s gastrointestinal tract: Lessons learned from in silico and in vivo modelling. Avian Dis. 52, 599–607, doi: 10.1637/8288-031808-reg.1 (2008).
Bardina, C. et al. Genetics and genomics of three new bacteriophages useful in the biocontrol of Salmonella . Front. Microbiol. 7, 545, doi: 10.3389/fmicb.2016.00545 (2016).
Green, M. R. & Sambrook, J. In Molecular Cloning: A Laboratory Manual 4rd ed. (Cold Spring Harbor Laboratory Press, 2012).
International Organization for Standardization. Particle size analysis — Laser diffraction methods. ISO 13320:2009 http://www.iso.org/iso/catalogue_detail.htm?csnumber=44929 (2009).
Hadjialirezaei, S. Coating Of Alginate Capsules. PhD Thesis. Norwegian University of Science and Technology (2013).
Musikasang, H., Tani, A., H-Kittikun, A. & Maneerat, S. Probiotic potential of lactic acid bacteria isolated from chicken gastrointestinal digestive tract. World J. Microbiol. Biotechnol. 25, 1337–1345, doi: 10.1007/s11274-009-0020-8 (2009).
Acknowledgements
This work was supported by grants from La Caixa and the Associació Catalana d’Universitats Públiques (2010ACUP00300), AGAUR-ACCIÓ-Generalitat de Catalunya (2010VALOR00114), AGAUR-Generalitat de Catalunya (2014SGR572) and of the Spanish MINECO (BIO2016–77011-R). ICN2 acknowledges the support of the Spanish MINECO through the Severo Ochoa Centres of Excellence Programme, under Grant SEV-2013-0295.We are grateful to the Servei de Granges i Camps Experimentals and the Servei de Microscòpia of the Universitat Autònoma de Barcelona (UAB) for their support. J.C. and J.O. received predoctoral fellowships from the UAB. We thank S. Escribano for her excellent technical assistance.
Author information
Authors and Affiliations
Contributions
J.C. and M.C.-S. carried out the encapsulation of the bacteriophages. Moreover, J.C. and J.O. performed the in vivo experiments. J.A.-S. contributed in the encapsulation experiments. P.C., M.C.-S., D.M. and M.L. participated in the design and coordination of the study and in drafting the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Pilar Cortés and Montserrat Llagostera are the inventors of the patent application EP2750519.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Colom, J., Cano-Sarabia, M., Otero, J. et al. Microencapsulation with alginate/CaCO3: A strategy for improved phage therapy. Sci Rep 7, 41441 (2017). https://doi.org/10.1038/srep41441
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep41441
This article is cited by
-
Isolation and encapsulation of bacteriophage with chitosan nanoparticles for biocontrol of multidrug-resistant methicillin-resistant Staphylococcus aureus isolated from broiler poultry farms
Scientific Reports (2024)
-
Characterization of two novel Salmonella phages having biocontrol potential against Salmonella spp. in gastrointestinal conditions
Scientific Reports (2024)
-
PLGA microparticle formulations for tunable delivery of a nano-engineered filamentous bacteriophage-based vaccine: in vitro and in silico-supported approach
Journal of Nanostructure in Chemistry (2024)
-
Perspectives on phage therapy for health management in aquaculture
Aquaculture International (2024)
-
Effect of phage vB_EcoM_FJ1 on the reduction of ETEC O9:H9 infection in a neonatal pig cell line
Veterinary Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.