Abstract
Trilobites were one of the most successful groups of marine arthropods during the Palaeozoic era, yet their soft-part anatomy is only known from a few exceptionally-preserved specimens found in a handful of localities from the Cambrian to the Devonian. This is because, even if the sclerotized appendages were not destroyed during early taphonomic stages, they are often overprinted by the three-dimensional, mineralised exoskeleton. Inferences about the ventral anatomy and behavioural activities of trilobites can also be derived from the ichnological record, which suggests that most Cruziana and Rusophycus trace fossils were possibly produced by the actions of trilobites. Three specimens of the asaphid trilobite Megistaspis (Ekeraspis) hammondi, have been discovered in the Lower Ordovician Fezouata Konservat-Lagerstätte of southern Morocco, preserving appendages and digestive tract. The digestive structures include a crop with digestive caeca, while the appendages display exopodal setae and slight heteropody (cephalic endopods larger and more spinose than thoracic and pygidial ones). The combination of these digestive structures and the heteropody has never been described together among trilobites, and the latter could assist in the understanding of the production of certain comb-like traces of the Cruziana rugosa group, which are extraordinarily abundant on the shallow marine shelves around Gondwana.
Similar content being viewed by others
Introduction
The Moroccan Fezouata Biota is the most important Ordovician Burgess Shale-type Lagerstätte known to date, having produced a very diverse marine assemblage, which includes not only articulated mineralized taxa, but also soft-bodied or lightly sclerotized organisms (e.g, annelids, priapulids, lobopodians)1,2. Preservation usually involves pyritization and subsequent weathering to iron oxides, thus the reddish colours of the fossils. The assemblage has been dated as late Tremadocian (~478 Ma), from relatively shallow waters, at or just above the storm-wave base3. Among the fossil arthropods, preserved appendages are frequent in the following groups: anomalocaridids, marrellomorphs, xyphosurans, aglaspidids, etc.2,4. Additionally, fossilized digestive tracts have been described in machaeridian annelids5.
Trilobites are common constituents of the Fezouata Biota, found as complete carcasses as well as putative articulated exuviae. However, soft-body preservation is extremely rare, with only antennae and some distal appendage remains recognized in Bavarilla3 and the nileid Symphysurus, where a single specimen of the latter exceptionally showed “preserved antenna, walking legs and midgut glands”1. Here we present the first occurrence of the preserved gut with associated digestive structures, plus a complete set of endopods and exopods in specimens of the asaphid trilobite Megistaspis (Ekeraspis) hammondi. They come from site “F-4”6, which is located about 7 km north of the Bou Izargane village and 19 km north of the city of Zagora, central Anti-Atlas of Morocco (Supplementary Fig. 1). At this locality, large trenches have been excavated in search of fossils, for scientific and commercial purposes, which were deposited during the late Tremadocian Araneograptus murrayi graptolite Biozone. The studied trilobites were collected by the Ben Moula family (Taychout village, near Alnif-Tinghir, Morocco), later bought by a professional fossil dealer and offered to the Museo Geominero of the Spanish Geological Survey, in Madrid (catalogue numbers MGM-6755X to MGM-6757X). Restoration of fossil material from Morocco is common, as it is often intended for amateur collectors. However, extreme care has been taken by the authors in authenticating the material studied here. Only specimens collected by reputable sources have been considered, as is the case of the Ben Moula family, who have provided the best Fezouata material1,2,3,5. These specimens have later been re-checked for consistency in lithology, colouration and slab thickness to ensure their authenticity and integrity. The areas that have been retouched in any form in the specimen of Fig. 1a have a lighter shade and outlined in red, and this restoration has not affected either the digestive structures or the diagnostic features. The details of the type of restoration in the specimen are indicated in Supplementary Fig. 2a, with the trilobite having ~10% of extensively reconstructed parts, ~12% of slightly reconstructed parts, ~18% of polished surfaces of the internal mould and ~5% of pieces transferred from the external mould (shown in Supplementary Fig. 2b).
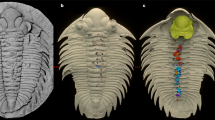
(a – d) MGM-6755X, with digestive structures. (a) Overall view of specimen, with restored areas outlined in red and in lighter shade (for detailed indication of type of restoration see Supplementary Fig. 2); (b) detail of cephalic area and first thoracic segments with crop, intestine and digestive caeca; (c) latex cast of posterior cephalic and anterior thoracic caeca; (d) detail of the tip of the pygidial axis with remains of intestine and possible ?caeca. (e–h) MGM-6756X, part (e,f) and counterpart (g,h), of a specimen with preserved endopods. (e) Ventral right-hand side. (f) Detail of the cephalic and first thoracic endopods. (g) Counterpart of pygidium and five thoracic segments. (h) Detail of the last five thoracic endopods in positive relief. (b–d) have been whitened with MgO. Scale bar, 30 mm for (a,e,g); 20 mm for (b); 10 mm for (c,d,f,h).
Megistaspis (Ekeraspis) hammondi7 (=M. (E.) cf. filacovi8,9) is a medium-large asaphid trilobite, with eight thoracic segments and a pygidium with a long caudal spine (Fig. 1a). It differs from the remaining species of the subgenus in the more anterior position of the eyes.
The original description of the species needs revision, because the diagnostic characters are barely recognizable on the holotype7, and the purported differences of the subspecies M. (E.) hammondi forteyi7 could be due to preparation by the fossil dealers (Supplementary Fig. 3a,b). Thus, the latter could be synonimized with the forma typica of M. (E.) hammondi.
Results
Digestive system
The first specimen studied was the internal mould of a complete exoskeleton (Fig. 1a), partially reconstructed (Supplementary Fig. 2), which shows the internal anatomy in the glabellar–anterior thoracic axis and the posterior end of the pygydial axis. The digestive system can be recognized under the carapace of the glabellar area (Fig. 1b). The axial tube consists of an 8 mm-wide (tr.) crop (one-third of glabellar width), which extends from the anterior (exsagittal) edge of the eyes, tapering to 4 mm at the posterior end of the cephalon. This is followed by a 3 mm-wide intestine, preserved up to the third thoracic segment (Fig. 1b) and reappearing for 22 mm just before the axial end of the pygidium (Fig. 1d). Anterior to the crop, there seem to be two small, bilaterally-symmetrical branching caeca, which occupy the whole anterior region below the glabella. Behind the eyes, we can recognize at least 4 pairs of laterally-oriented, lobed caeca, whose tips are aligned longitudinally (posterior ones lengthen transversely as crop tapers). The bilateral position, together with the consistent size and similar shape of these structures suggests that these are true body features rather than irregular fractures of the exoskeleton revealing underlying cavities. The digestive caeca under the thoracic axis are also preserved in the first three segments (Fig. 1c), and are similar in size and shape to the posteriormost cephalic pair. The posterior end of the intestine seems to preserve three (?) pairs of small, simple caeca below the posterior part of the pygidial axis. However, due to the quality of preservation it cannot be ascertained whether caeca were present along the whole length of the digestive tract.
The crop and intestine present a positive dorsal relief, while the caeca have a negative ventral relief. The former have the same texture and grain size as the matrix, indicating active sediment ingestion or rapid sediment infill. The digestive caeca were presumably permineralized (possibly pyritized) in the early diagenetic stages, preserving their shape and relief. This has been described in other trilobites and soft-bodied arthropods and is attributed to these digestive structures being chemically enriched in life and prone to preferential mineralization10,11. These structures were later altered and eventually left the void observable now.
Appendages
The second specimen of M. (E.) hammondi preserved a complete set of endopods, but no exopods (Fig. 1e–h). The specimen is incomplete, only preserving the right-hand side of the animal, from the base of the antennae to the last pygidial appendage. Its counterpart (Fig. 1g) is composed of the posterior 5 thoracic segments plus the pygidium. The endopods are preserved as external moulds of the dorsal side and some are also casts in positive relief (Fig. 1f,h). The endopods show some degree of heteropody: the cephalic ones are larger and heavier than the thoracic and pygidial ones and they are armed by dorsal and ventral spines on the proximal podomeres (Fig. 2). Podomere boundaries are not recognizable, but the spines probably correspond to podomeres 2–4, while the distal podomeres (5–6) appear smooth. The spines are stout and triangular; in the best-preserved cephalic appendage (2nd), a maximum of 11 spines can be counted dorsally and 7 ventrally (Figs 1f and 2). These spines are also recognizable in the cast of the third cephalic appendage. No spines are recognisable in the preserved thoracic and pygidial appendages. A total of 10 pygidial appendages can be recognized, which is more than the pygidial axial rings and pleural ribs (up to 7 and 5–6, respectively8). This is not a rare phenomenon in trilobites where appendages are known, like Triarthrus12. This decoupling between observed pygidial segments and number of pygidial appendages could be especially significant in those trilobites with effaced pygidial features like asaphoids and illaenoids.
Interpretation of specimen in Fig. 1f (MGM-6756X). View shows ventral structures in right-hand side: antenna (ant), three cephalic endopods (ce1–3) and first thoracic endopods (te1–2). Appendages are recognised by iron-stained external moulds, but some still preserve the casts (ce1, ce3, te1, te2). The second and third cephalic endopods are heavily armoured with dorsal and ventral spines (yellow arrowheads). Other recognisable features are facial suture (fs), the sediment infill (white arrows) under the right librigena (rlb) and the cranidium-thoracic boundary (ctb). The dashed line corresponds to the sagittal plane of the specimen, while ‘gr’ indicates fragments of graptolite stipes that were lying on the seafloor under the trilobite. Scale bar, 10 mm.
The third specimen displaying non-mineralized structures (Fig. 3) is an almost-complete, articulated carapace. Only the anterior portion of the cephalon and the posterior region of the pygidium are missing. The specimen is slightly flattened, and the dorsal cuticle has been lost in some regions, exposing the underlying soft-parts. The first and second left pleurae preserved their respective endopods with their tips folded underneath. Though most significantly, on the right side of the carapace, the cranidium, the thoracic pleurae and the pygidial pleural fields have split to reveal the postero-laterally oriented fine lamellae fringing the exopods (Fig. 3b). The lamellae of the posterior-most pygidial exopods, if complete in the specimen in Fig. 3a, were quite short, terminating before the border of the pygidium (Fig. 3c).
Specimen MGM-6757X (in dorsal aspect) preserving exopodal setae. (a) Overall view of specimen. Yellow arrows point to setae; black arrows indicate first and second thoracic endopods; green arrow points to left eye; blue arrowheads indicate the facial suture; white arrowheads indicate the thoracic-pygidial boundary. (b) Detail of the setae fringing the right cephalic exopods, exposed through a break in the cranidium. (c) Detail of the setae from right thoracic and pygidial exopods. The slab’s underside contains a well-preserved specimen of the graptolite “Tetragraptus” bulmani Thomas (=T. longus Lindhom), a very common species in the Araneograptus murrayi Biozone of the Fezouata Shale. Scale bar, 20 mm for (a); 10 mm for (b,c).
Discussion
The digestive system of M. (E.) hammondi differs from the types described to date in trilobites10,11. It has lateral metameric-paired digestive caeca (recently11 referred to as “type 1”) of similar shape and relative size, but these extend to the posterior end of the midgut (pygidial axis), but it also has a well-defined crop (twice as wide as the intestine), characteristic of “type 2”, which lacks digestive caeca. This is the first instance of digestive caeca described among typical asaphids, where simple guts (“type 2”) have been recognized in the genera Isotelus and Birmanites, where the cephalic portion of the alimentary canal is not preserved13. However, these and other asaphid genera, where the presence of auxiliary muscular impressions in the glabella suggest the existence of a crop11, differ from Megistaspis in having a forked hypostome, related with predatory/scavenging habits14, although such an interpretation has not been undisputed15. Based on the gut preserved with the same texture as the matrix surrounding the fossil, the non-forked hypostome of Megistaspis8,16 and the slight heteropody in the cephalic appendages we can infer a detritus feeding habit for M. (E.) hammondi. This feeding habit is very common among other benthic trilobites14. A gut tract preserved under the pygidial axis has been also described in the asaphid Isotelus17, but there was no evidence of digestive caeca.
The cephalic appendages observed in Megistaspis are larger than the thoracic and pygidial ones, and are armed by dorsal and ventral spines in the proximal podomeres. The slight heteropody of the Moroccan Megistaspis may be related with a particular burrowing style, deduced for certain Ordovician trilobites from the trace fossil record. For example, Cruziana rugosa d’Orbigny displays combed dig-marks with up to twelve sharp-crested scratches, forming sets separated by transverse shelves (Fig. 4a). This has been interpreted as discontinuous ploughing of a trace-maker burrowing in head-down (procline) position, with multiple claws or spines on the tip of one or more of its endopodal podomeres18. In both surficial and endostratal variants of C. rugosa, the well-developed sets of scratches would questionably be the result of a strongly serrated tip of the pretarsus18, but more likely be made by the heavier-built, proximal podomeres of the cephalic appendages, especially when these where spine-bearing as in Megistaspis. The sets of parallel scratches make little sense as the “fingerprints” of individual distal claws. The production of this trace has been attributed19 to trilobites with anterior appendages greater in size that the posterior ones, and equipped with multiple or serrated claws. However, other authors20 have interpreted C. rugosa as the digging action of a single pair of broad, multidigited anterior appendages, producing dig marks that remain separate, and for the most part undisturbed by successive excavation strokes. However, in the absence of any record of a trilobite equipped with these structures (see ref. 21 for extensive discussion), this ichnospecies of Cruziana is indeed consistent with the spines observed in the podomeres of the cephalic appendages of M. (E.) hammondi (Figs 1f and 2), which could have produced sets of parallel striae or “combs” (Fig. 4a). The stout spines of this trilobite species are more likely associated with a digging activity than the sharp, fine spines of trilobites such as Olenoides, which probably had a grasping and ripping function for predation or scavenging22. Although those fine spines are present in some deep-water Ordovician olenid trilobites such as Triarthrus12,23, in the asaphid trilobite studied here, the direct evidence does not suggest this as the principal feeding habit. Furthermore, Cruziana rugosa is regarded as a combined feeding and locomotion trace (‘pascichnion’) most likely produced by the food searching strategy of asaphoid trilobites18,24. This attribution is based in the large size reached by the traces (up to 26 cm wide21), only compatible with the size of some of these trilobites distributed around mid- to high-latitude Gondwanan regions, and also due to the local co-occurrence in the same levels of C. rugosa and the trilobite Ogyginus25. However, among asaphoid trilobites there are two types of hypostomes: those with forked hypostomes, possibly related with predatory or scavenging habits on worms and other small invertebrates14, and those with normal hypostomes (such as Ekeraspis, Asaphellus and Ogyginus), which were probably deposit feeders with microphagous habits (“mudtrophobacterivory” feeding21), being the most likely candidates as trace-makers of C. rugosa.
(a) Trail: Cruziana rugosa d’Orbigny, from early Dapingian of the Zenta Range near Laguna Verde, Argentina (range of the ichnospecies: Floian to Sandbian), MGM-6760X. (b) Resting burrow, Rusophycus morgati Baldwin, Armorican Sandstone (Floian) from the Armorican Massif, western France, interpreted as made by the trilobite Ogyginus14, IGR-114954. (c,d) Resting burrows, Rusophycus carleyi (James), upper Fezouata Shale (early Floian beds), from SW Ouzina, southern Tafilalt (eastern Anti-Atlas, Morocco), locally associated with the trilobite Asaphellus23. (c) MGM-6759X. (d) MGM-6758X, detail of traces attributed to endopodal spines along all the walking legs and impression of coxae to the right. hyp, hypostome impression; (a,c,d) have been whitened with MgO. Scale bar, 10 mm for (a,c,d); 20 mm for (b).
Some ovate resting traces of Rusophycus, display the impression of most of the ventral morphology of their supposed asaphoid producer. The fine, backward-oriented, oblique fine crests of Rusophycus morgati and R. carleyi (Fig. 4b–d), have been attributed to endopodal spines on possibly all the endopods26. These Lower Ordovician trilobite traces recorded in the high-latitude peri-Gondwanan shelves, had previously been assigned to predatory forms14, but have since been reinterpreted as moulting burrows18,27. The trace-makers lack forked hypostomes and have been assigned to Ogyginus14 and Asaphellus26. The ventral soft-body morphology described herein may have produced similar resting traces, except for Megistaspis’ long caudal spine, which could have been directed slightly upward in life.
Conclusions
Some early Palaeozoic trilobites10,11 and numerous non-mineralized arthropods28 had digestive structures that were prone to early permineralization, possibly due to being enzymatically rich areas11,25. Sometimes it was the digestive tract that was preserved, while others it was the glands connected to it, mainly the mid-gut glands. These mid-gut glands are paired, metameric structures located on the sides of the intestine, and in modern arthropods occur in groups with predatory habits, such as chelicerates and remipedes25, but its combination with a crop (for storage of food), non-forked hypostome and stout appendage spines suggests a mixed feeding habit for Megistaspis. This original combination of crop and digestive caeca might be regarded as a new, “type 3” trilobite digestive system11. These Moroccan fossils constitute the first recognition of heteropody in trilobite walking appendages, where Megistaspis would primarily use its spiny cephalic appendages in digging for food, with a comb-like movement already inferred for the producers of the Cruziana rugosa group of traces, an ichnogroup widely distributed throughout the Ordovician of Gondwana.
Methods
Five of the six examined specimens are housed in the Museo Geominero of Madrid, Spain (prefixed MGM), and one at the Geological Museum of the University of Rennes, France (prefixed IGR). All photographs were taken with a Canon EOS7D digital SLR camera equipped with a Canon MP-E 65 mm 1–5x macro lens. Some of the specimens were whitened with MgO before photographing (Figs 1b–d and 4a,c,d), and digestive structures of specimen MGM-6755X were replicated as latex casts and whitened (Fig. 1c). Illustrations composed and processed, to homogenise light intensity across figures, with Adobe Photoshop CS3. Supplementary Fig. 1 was drafted with CorelDRAW 12 and Adobe Photoshop CS3.
Additional Information
How to cite this article: Gutiérrez-Marco, J. C. et al. Digestive and appendicular soft-parts, with behavioural implications, in a large Ordovician trilobite from the Fezouata Lagerstätte, Morocco. Sci. Rep. 7, 39728; doi: 10.1038/srep39728 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Van Roy, P. et al. Ordovician faunas of Burgess Shale type. Nature 465, 215–218 (2010).
Van Roy, P., Briggs, D. E. G. & Gaines, R. R. The Fezouata fossils of Morocco; an extraordinary record of marine life in the Early Ordovician. J. Geol. Soc. 172, 541–549 (2015).
Martin, E. et al. The Lower Ordovician Fezouata Konservat-Lagerstätte from Morocco: age, environment and evolutionary perspectives. Gondwana Res. 34, 274–283 (2016).
Van Roy, P., Daley, A. C. & Briggs, D. E. G. Anomalocaridid trunk limb homology revealed by a giant Ordovician filter-feeder with paired lateral flaps. Nature 522, 77–80 (2015).
Vinther, J., Van Roy, P. & Briggs, D. E. G. Machaeridians are Palaeozoic armoured annelids. Nature 451, 185–188 (2008).
Lefebvre, B. & Botting, J. P. First report of the mitrate Peltocystis cornuta Thoral (Echinodermata, Stylophora) in the Lower Ordovician of central Anti-Atlas (Morocco). Ann. Paléont. 93, 183–198 (2007).
Corbacho, J. & Vela, J. A. Giant trilobites from Lower Ordovician of Morocco. Batalleria 15, 3–32 (2010).
Vidal, M. Trilobites (Asaphidae et Raphiphoridae) de l’Ordovicien inférieur de l’Anti-Atlas, Maroc. Palaeontographica A 251, 39–77 (1998).
Martin, E. L. O. et al. Biostratigraphic and palaeonvironmental controls on the trilobite associations from the Lower Ordovician Fezouata Shale of the Central Anti-Atlas, Morocco. Palaeogeogr., Palaeoclimat., Palaeoecol 460, 142–154 (2016).
Chatterton, B. D. E., Johanson, Z. & Sutherland, G. Form of the trilobite digestive system: alimentary structures in Pterocephalia . J. Paleont. 68, 294–305 (1994).
Lerosey-Aubril, R. et al. Controls on gut phosphatisation: the trilobites from the Weeks Formation Lagerstätte (Cambrian; Utah). PLoS ONE 7, 1–9 (2012).
Whittington, H. B. & Almond, J. B. Appendages and habits of the Upper Ordovician trilobite Triarthrus eatoni . Phil. Trans. R. Soc. London B 317, 1–46 (1987).
Fatka, O., Lerosey-Aubril, R. & Rak, Š. Fossilised guts in trilobites from the Upper Ordovician Letná Formation (Prague Basin, Czech Republic). Bull. Geosci. 88, 95–104 (2013).
Fortey, R. A. & Owens, R. M. Feeding habits in trilobites. Palaeontology 42, 429–465 (1999).
Hegna, T. A. The function of forks: Isotelus-type hypostomes and trilobite feeding. Lethaia 43, 411–419 (2010).
Jaanusson, V. Untersuchungen über baltoskandische Asaphiden. III. Über die Gattungen Megistaspis n. nom. und Homalopyge n. gen. Bull. Geol. Inst. Uppsala 36, 59–77 (1956).
English, A. M. & Babcock, L. E. Feeding behaviour of two Ordovician trilobites inferred from trace fossils and non-biomineralised anatomy, Ohio and Kentucky, USA. Mem. Ass. Austral. Palaeontol. 34, 537–544 (2007)
Seilacher, A. Trace fossil analysis (Springer-Verlag, 2007).
Seilacher, A. Cruziana stratigraphy of “non fossiliferous” Palaeozoic sandstones. In Crimes, T. P. & Harper, J. C. (eds). Trace fossils, Geol. J. Spec. Iss. 3, 447–476 (1970).
Baldwin, C. T. The stratigraphy and facies associations of trace fossils in some Cambrian and Ordovician rocks of north western Spain. In Crimes, T. P. & Harper, J. C. (eds). Trace fossils 2, Geol. J. Spec. Iss. 9, 9–40 (1977).
Whittington, H. B. Exoskeleton, moult stage, appendage morphology, and habits of the Middle Cambrian trilobite Olenoides serratus . Palaeontology 23, 171–204 (1980).
Neto de Carvalho, C. Roller coaster behavior in the Cruziana rugosa group from Penha Garcia (Portugal): Implications for the feeding program of trilobites. Ichnos 13, 255–265 (2006).
Boyer, D. L. & Mitchell, C. E. Aligned trace fossils from the Utica Shale: implications for mode of life and feeding in the trilobite Triarthrus beckii . Lethaia., doi: 10.1111/let.12177 (2016).
Bergström, J. Lower Paleozoic trace fossils from eastern Newfoundland. Canadian Journal of Earth Sciences 13, 1613–1633 (1976).
Mángano, M. G. & Waisfeld, B. Looking for the usual suspects: Trilobites as Cruziana-Rhusophycus producers in lower Paleozoic sandstones of northwest Argentina. Abstracts First International Congress on Ichnology (Ichnia 2004), Trelew (Argentina), 50–51 (2004).
Gibb, S., Chatterton, B. D. E. & Gingras, M. K. Rusophycus carleyi (James, 1885), trace fossils from the Lower Ordovician of southern Morocco, and the trilobites that made them. Ichnos 17, 271–283 (2010).
Seilacher, A., Gibb, S. & Hughes, N. Trilobite trace fossils made for moulting? Palaeontological Society of India 60, 27–32 (2015).
Butterfield, N. J. Leanchoilia guts and the interpretation of three-dimentional structures in Burgess Shale-type fossils. Paleobiology 28, 155–171 (2002).
Vannier, J. & Chen, J.-Y. Digestive system and feeding mode in Cambrian naraoiid arthropods. Lethaia 35, 107–120 (2002).
Vannier, J. et al. Sophisticated digestive systems in early arthropods. Nat. Commun. 5, 3641 (2014).
Acknowledgements
We thank our colleagues Greg Edgecombe and John Paterson for their suggestions to early versions of the manuscript and to Nigel Hughes, an anonymous editor and an anonymous referee for their careful and constructive revision. We are grateful to Carlos Alonso (UCM, Madrid) for photographing the specimens. This work has been supported by the Spanish Ministry of Economy and Competitiveness, project number CGL2012-39471/BTE. D.C.G.-B. would like to acknowledge the support of Australian Research Council Future Fellowship FT130101329. This is a scientific contribution to the projects IGCP 653 (IUGS-UNESCO) and SIMI 5/6 RALI (ANR, French Research Agency).
Author information
Authors and Affiliations
Contributions
J.C.G.-M. designed the research; J.C.G.-M. and D.C.G.-B. performed the research and wrote the paper with help from I.R. and A.A.S.; J.C.G.-M. and A.A.S. carried out field-work in Morocco in order to confirm the location of the fossil site with new specimens of the same taxon (albeit without soft parts) and define its age based on its graptolite assemblage. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Gutiérrez-Marco, J., García-Bellido, D., Rábano, I. et al. Digestive and appendicular soft-parts, with behavioural implications, in a large Ordovician trilobite from the Fezouata Lagerstätte, Morocco. Sci Rep 7, 39728 (2017). https://doi.org/10.1038/srep39728
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep39728
This article is cited by
-
Ordovician-Cambrian Palaeontological Heritage of Zagora Province: A Bibliometric Analysis from 1984 to 2020 (Anti-Atlas, Morocco)
Geoheritage (2022)
-
An Inventory of Geoheritage Sites in the Draa Valley (Morocco): a Contribution to Promotion of Geotourism and Sustainable Development
Geoheritage (2019)
-
Digitization of Fossils from the Fezouata Biota (Lower Ordovician, Morocco): Evaluating Computed Tomography and Photogrammetry in Collection Enhancement
Geoheritage (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.