Abstract
Demands for faster and more accurate methods to analyze microbial communities from natural and clinical samples have been increasing in the medical and healthcare industry. Recent advances in next-generation sequencing technologies have facilitated the elucidation of the microbial community composition with higher accuracy and greater throughput than was previously achievable; however, the short sequencing reads often limit the microbial composition analysis at the species level due to the high similarity of 16S rRNA amplicon sequences. To overcome this limitation, we used the nanopore sequencing platform to sequence full-length 16S rRNA amplicon libraries prepared from the mouse gut microbiota. A comparison of the nanopore and short-read sequencing data showed that there were no significant differences in major taxonomic units (89%) except one phylotype and three taxonomic units. Moreover, both sequencing data were highly similar at all taxonomic resolutions except the species level. At the species level, nanopore sequencing allowed identification of more species than short-read sequencing, facilitating the accurate classification of the bacterial community composition. Therefore, this method of full-length 16S rRNA amplicon sequencing will be useful for rapid, accurate and efficient detection of microbial diversity in various biological and clinical samples.
Similar content being viewed by others
Introduction
Microbiotas are complex microbial communities containing hundreds of species-level phylotypes and are found everywhere, from humans (e.g., the microbiota within the gut) to environments. Interestingly, these communities and their genetic blueprint, referred as the microbiome1, has been implicated in a variety of human diseases2, including inflammatory bowel diseases3, type 2 diabetes4, and brain abnormalities, such as autism spectrum disorder5. Thus, the microbiome has attracted much attention in the medical and healthcare industries, and elucidation of the microbiota composition in the human body is critical for further advancements in our understanding of related diseases and physiological states. In this regard, because species in the same taxonomic units from genus up to phylum play a variety of roles, some may be crucial, others may not be correlated with the phenotype6, it is critical to obtain the higher taxonomic resolution to species level for better understanding of the functional effects of microbiota on health and further identifying key players in a specific phenotype7.
Until recently, second-generation sequencing has been widely used to assess the composition of the microbial community with higher accuracy and greater throughput than previous methods, enabling the completion of high-profile microbiome projects, such as the Human Microbiome Project8. Despite the high-throughput and high sequencing accuracy, second-generation sequencing can produce only a partial (~100–500 bp) sequence of the 16S rRNA gene. Within this technical limitation, researchers have to select the most effective target regions to identify taxa from full-length 16S rRNA gene sequences containing nine hypervariable regions (V1–V9) as phylogenetically informative markers. Additionally, the genetic distance of individual species is related to the similarities among subregions and full-length sequences9. Thus, long reads sequencing of the 16s rRNA gene is a promising approach to provide high-resolution analysis of microbial communities at the species level.
Recently, researchers have developed a new nanopore DNA sequencer10 (MinION) that has significant advantages, such as long-read output, low cost, portability, and rapid real-time analysis, as compared with other DNA sequencing technologies. Despite the relatively lower accuracy of this method (~80%) compared with other sequencing technologies, nanopore sequencing has been applied to sequencing of eukaryotic11, bacterial12,13,14, and viral genomes15,16. Furthermore, nanopore sequencing has been successfully adapted for cDNA and amplicon sequencing17,18,19,20,21. However, the plausibility of using this platform to analyze the gut microbiota composition at the species level has not been fully elucidated in comparison with that of short-read sequencing technologies. In this study, we investigated whether the nanopore sequencing is suitable for analyzing the composition of the mouse gut microbiota at the species level and compared the results with those obtained by the short-read sequencing that has so far been widely employed in the field.
Results
Short-read sequencing of 16S rRNA V3-V4 region amplicon libraries
To compare the ability of the short-read and nanopore sequencing platforms to analyze the microbial community, we first prepared short-read sequencing libraries (Illumina platform) from biologically duplicated metagenomic DNAs isolated from the gut microbiome of 50-week-old mice (Fig. 1A). To this end, the V3–V4 hypervariable region (approximately 469 bp) of the 16S rRNA gene, which has been used for taxonomic classification of the microbial community in human microbiome studies, was amplified using a two-step PCR method (see the Materials and Methods for experimental details)22. Illumina 250-bp paired-end sequencing of the amplicon targeting the V3–V4 region of the 16S rRNA gene generated 249,593 and 183,029 sequencing reads. Each paired-end read was joined to produce 127,969 and 111,058 reads for samples A and B, respectively, using the QIIME pipeline23. Joined reads having less than 75% of their original length were removed in this step. We then performed quality trimming of joined paired sequencing reads (≥Q20), which generated 105,590 and 92,119 sequencing reads. These criteria resulted in a mean read length of about 447 bp, and approximately 83% of sequencing reads were retained for further microbial community analysis (Table 1).
(A) Schematic workflow to examine the composition of the mouse gut microbiota using the nanopore (MinION) and the short-read (Illumina MiSeq) sequencing. (B) The distribution of PHRED quality scores of short-read sequencing data and pass 2D reads of nanopore sequencing data. (C) Density plot for length distribution comparison of short-read sequencing data and pass 2D reads of nanopore sequencing data. Each sample is colored separately.
Nanopore sequencing of full-length 16S rRNA amplicon libraries
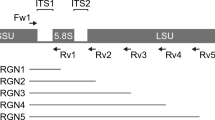
At the same time, broad amplification of the full-length 16s rRNA genes from metagenomic DNA samples was achieved using the 16S rRNA gene-specific primers adapted from S-D-bact-0008-c-S20 and S-D-bact-1391-a-A-17 (Fig. 1A)24. Using these amplicons, nanopore sequencing libraries were constructed with internal control DNA (see the Materials and Methods for details of nanopore sequencing). The nanopore sequencing generated 101,269 and 33,174 sequencing reads from 923 and 822 pores for samples A and B, respectively (Table 2). The raw data contained the one-dimensional (1D) template, 1D complement, and 2D reads consensus sequence with enhanced accuracy. To obtain high-quality reads, the pass 2D reads were sorted specifically from raw data using the Metrichore 2D base calling program. Integrated information from the template and complement reads could be used for 2D base-calling, which results in a higher mean quality score (Supplemental Fig. S1) and better accuracy than total reads11,25. Although sample B data (11,078 reads) showed fewer sequencing reads than sample A (36,166 reads), the pass 2D reads were 36% and 33% of total reads with mean quality scores of 9.69 and 9.77, respectively (Fig. 1B and Table 2). The read length had a narrow length distribution, and the mean read length was approximately 1,393 bp, which was nearly the full-length of the 16s rRNA gene (about 1,550 bp; Fig. 1C and Table 2). The unexpected long reads seemed to be the products of concatemers formed at the hairpin adapter ligation step, based on the presence of multiple 16S rRNA gene-specific primer binding sites in their sequences.
The accuracy of the nanopore sequencing was computed based on the coverage (≥80%) of the sequencing reads of the internal control DNA (DNA CS) against the reference sequence using the LAST aligner (version 658)26. The LAST alignment algorithm, using adaptive seeds for alignment, has been used to align nanopore sequencing reads to references27. Sequencing accuracy was defined as the number of matching nucleotides divided by the total number of matches, mismatches, insertions, and deletions26. Consequently, nanopore sequencing exhibited an average accuracy of 79.6%, with 9.0% mismatches, 6.4% insertions, and 5.0% deletions (Supplemental Fig. S2). This accuracy was similar to that reported in previous studies (70–80%)26,28.
Sequencing data analysis
The short-read sequencing data sets were then analyzed using the operational taxonomic unit (OTU) approach. To this end, the QIIME pipeline was used to cluster the 16S rRNA gene sequences based on their similarity; this approach has been widely used for microbial community analysis8,23. Within these data, 104,899 and 91,571 sequencing reads were clustered into 1,055 and 978 OTUs from the data for samples A and B, respectively, at the 0.03 dissimilarity threshold (Table 3). OTU taxonomy was then determined using the Ribosomal Database Project classifier retrained toward the Greengenes database of 13_8 version29. The richness of gut microbiota was estimated by the rarefaction curve, which showed similar patterns between biological duplicates (Fig. 2A and Supplemental Fig. S3).
(A) Rarefaction curves of mouse fecal samples based on short-read sequencing (Illumina). Total OTUs were generated by 3% distances. Total sample richness estimates were calculated by the observed OTUs. (B) Percentage of taxonomic units assigned as reads at the order, family, genus, and species levels. (C) Venn diagram showing the shared and specific phylotypes between Illumina A and B data. The Spearman rank correlation test (R2 = 0.8345, p < 0.0001) showed the significance of relationships between duplicates. Asterisks indicate the significance of the pairing (****p < 0.0001) (D) Two-way Venn diagram depicting the number of shared and specific phylotypes between nanopore A and B data. Percentages show the proportion of aligned reads corresponding to each phylotype per total reads. The results of Spearman rank correlation test (R2 = 0.9350, p < 0.0001) showed the significance of relationships between duplicates. Asterisks indicate the significance of the pairing (****p < 0.0001). (E,F) Venn diagram showing the shared and specific taxonomic units at the (E) phylum and (F) class levels between nanopore and Illumina sequencing data. The Spearman rank correlation test showed the significance of the relationship. Asterisks indicate the significance of the pairing (** 0.01 < p < 0.001, ****p < 0.0001).
On the other hand, the microbiota composition was determined based on the nanopore sequencing data obtained with the phylotyping approach (taxonomy-supervised analysis), which allocates sequences directly into taxonomic bins based on their similarity. Since computational clustering based on sequence similarity is not required for this approach30, it is more tolerant to unnatural variants often observed in OTU-based approach31. To assign the taxonomic units, 5,432 and 2,140 of the pass 2D reads were aligned to the GreenGene reference (13_8 version) with a mean length of 1,312 bp and 1,308 bp using LAST, respectively (Table 3). With this method, 15% (5,432 of 36,166) and 19% (2,140 of 11,174) of the pass 2D reads were aligned to the taxonomic reference, and a large portion of nonaligning reads remained unidentified. However, assignments of taxonomic units were significantly similar between duplicates (Fig. 2B).
From the short-read sequencing data, we observed 54 phylotypes shared by two biological duplicates (>99.99% of sequencing reads of each sample). Only three and nine phylotypes were detected from samples A and B, respectively, with less than 0.01% of sequencing reads (Fig. 2C and Supplemental Table S1). Similarly, 34 phylotypes were identified from both nanopore sequencing data sets (Spearman’s rank correlation, R2 = 0.935, p < 0.0001) with 99.1% and 99.9% of the pass 2D reads, respectively (Fig. 2D and Supplemental Table S2). Fourteen and one phylotypes were assigned by 0.9% (49 of 5,432) and 0.09% (2 of 2,140) of the pass 2D reads, respectively. Although sample-specific phylotypes were observed at a negligible level, and a relatively high error rate (20.4%) was determined from the nanopore sequencing reads, high reproducibility and correlation were achieved between the biological duplicates. Thus, 34 major phylotypes were reproducibly detected using nanopore amplicon sequencing.
Comparison of microbial composition determined by two sequencing platforms
Next, we compared the microbial compositions determined using the two sequencing platforms. Both platforms identified eight bacteria phyla and 13 bacteria classes (Fig. 2E,F). Statistically significant similarity was observed in the relative proportions of members of the major phyla (Spearman’s rank correlation, R2 = 0.850, p = 0.003) and classes (Spearman’s rank correlation, R2 = 0.829, p < 0.0001) between short-read and nanopore sequencing data.
The relative abundances of microbial compositions detected from the two sequencing platforms were then depicted using heat maps at the order, family, genus, and species levels (Fig. 3A). All taxonomic units were classified into three groups based on whether they were detected by the short-read sequencing platform only (group III), the nanopore sequencing platform only (group II), or both platforms (group I). The relative abundances of the most dominant phylotype in nanopore and short-read sequencing data were quite similar. In the short-read sequencing data, taxonomic units (39 units) with high abundance (>0.5%) were observed only in group I. On the other hand, taxonomic units (66 units) with low abundance (<0.5%) were observed in all groups. Although one phylotype (o__Verrucomicrobiales; f__Verrucomicrobiaceae; g__Akkermansia; s__muciniphila) and three taxonomic units (g__Blautia, s__pseudolongum, and s__ovatus) showed different abundance deviations (log2 fold-change: −1.58–1.81), the others (89%) had no significant differences (log2 fold-change: >−1 or <1) in group I (Fig. 3A). All genera-based taxonomic units detected from the nanopore sequencing data were similar to the previous mouse gut microbiota analysis32,33,34,35. For example, Prevotella was reported to be present at relatively low abundance in the mouse gut microbiota32. Furthermore, all bacterial species detected from groups I and II have also been identified in the mouse gut microbiota36,37,38,39,40,41,42,43. For example, the compositions of Lactobacillus reuteri and Bifidobacterium animalis are associated with animal obesity44, and Bacteroides ovatus has been reported to provide XyG catabolism to the host as a common gut symbiont38. Overall, the bacterial compositions were significantly similar between the two platforms at the order (Spearman’s rank correlation, R2 = 0.8851, p < 0.0001), family (Spearman’s rank correlation, R2 = 0.9086, p < 0.0001), genus (Spearman’s rank correlation, R2 = 0.8354, p < 0.0001), and species levels (Spearman’s rank correlation, R2 = 0.5389, p = 0.0124). Thus, comparative analysis of the microbial composition independently profiled by nanopore and short-read sequencing platforms showed that nanopore sequencing was capable of determining the correct microbial composition up to the species level.
(A) Heat map for the mean relative abundances for the two platforms at the order, family, genus, and species levels. Nanopore and Illumina columns are colored with relative abundances ranging from 0% to 100% according to the color key in the lower left corner of the figure. The +/− column is colored with log2 fold-changes ranging from −2 to 2 according to the color key in the lower left corner of the figure. The black border indicates significantly different abundance deviation (log2 fold-change, lower than −1 or higher than 1). The taxonomic units were categorized as I, II, and III according to whether they were detected by Illumina only (I), nanopore only (II), or both platforms (III). The Spearman rank correlation test (R2) showed the significance of relationships between the two platforms. Asterisks indicate the significance of the pairing (*p < 0.05, ****p < 0.0001). (B) Proportion of groups (I–III) at the levels of taxonomic classification from order to species.
Species detection using full-length 16S rDNA amplicon sequencing
We also observed the different proportions of groups at different taxonomic resolutions (Fig. 3B). According to the taxonomic resolution from order to species, the relative proportions of groups I (both sequencing platforms) and III (short-read sequencing only) were decreased. This observation may reflect in the fact that the insufficient read length or sequencing depth was used to analyze the microbial composition. In contrast, the relative proportion of group II (nanopore sequencing only) was increased. In this regard, we hypothesized that long reads generated by nanopore sequencing spanned multiple variable regions and could better separate the microbial composition than Illumina sequencing due to the nearly full-length 16s rDNA reads. To identify the effects of long reads on the microbial composition analysis at the species level, we performed phylogenetic analysis of combined datasets by priority and identified 16 phylogenetically distinct species distributed in 13 genera (Fig. 4A). As a result, four species (Bacteroides acidifacies, Bacteroides ovatus, Bifidobacterium animalis, and Bifidobacterium psudolongum) were separated from their genera Bacteroides and Bifidobacterium in the phylogenetic tree. Interestingly, the taxonomic separation of Bifidobacterium genus was only observed in the nanopore sequencing data, unlike the Bacteroides genus. This indicated that Bacteroides acidifacies and Bacteroides ovatus could be separated by OTUs defined as a cluster of short reads with 97% similarity, whereas Bifidobacterium animalis could not (Fig. 4A).
(A) Maximum-likelihood phylogenetic tree of 16 species identified in this study. The tree was generated in MEGA655. Reference sequences were obtained from the GreenGene (13_8) database. The clustering of the sequences was tested by a bootstrap approach with 1,000 repeats, and bootstrap values below 70 were clipped. The red box indicates the species separated from their genus. (B) Detected variants between two 16S rRNA gene sequences of the separated species are represented as a vertical line on the 16S rDNA sequences. Variants were defined as nucleotide present in less than 50% of aligned position frequencies. The black and red arrows indicate the binding positions of primer sets for the amplification of V3–V4 regions and nearly full-length regions on 16S rDNA sequences, respectively.
For further investigation, we compared the divergence of each reference 16S rRNA sequence. Each taxonomic reference 16S rRNA sequence corresponding to the species was extracted from the aligned data of nanopore reads, followed by aligning them using the ClustalW multiple alignment algorithm45. All variants of each reference 16S rRNA sequence were determined by detecting the allele frequencies (less than 50%) from the multiple aligned taxonomic references. Nine variants of V3–V4 regions (total 37 variants) were observed between Bacteroides acidifacies and Bacteroides ovatus, whereas four variants (total 39 variants) were detected between Bifidobacterium species (Fig. 4B). The variants of the 16S rRNA gene between Bifidobacterium animalis and Bifidobacterium pseudolongum were enriched, particularly in the V1–V2 regions, compared with other variable regions. As expected, however, other phylogenetically similar species within different genera had more variants than species within the same genus (Supplemental Fig. S4). Taken together, these findings suggested that the V3–V4 region was insufficient for analysis of the microbial community composition at the species level and that full-length 16S rRNA sequencing had the advantage of covering multiple variable regions of 16s rRNA genes.
Discussion
To investigate the relationship between host and microbiota, several methods have been employed for the detection of microbial community composition from natural and clinical samples. In particular, the composition of the gut microbiota has been elucidated using both metagenomic and 16S rRNA amplicon sequencing approaches, the latter of which is most commonly used8. Currently, metagenomic and metatranscriptomic sequencing of microbiota are actively applied with increased sequencing depth to improve our understanding of the microbial community with better resolution46,47. In addition, despite the limited sequencing output level, long reads produced by PacBio sequencing platform have begun to demonstrate their potential to provide accurate analysis of the microbial community composition48.
Here, we examined the potential of a third-generation sequencer, MinION, for identification of the microbiome composition in mouse fecal samples. Although long reads generated from the nanopore sequencer were found to have relatively higher error rates compared with other platforms, sequencing reads of nearly full-length 16S rRNA could provide more accurate taxonomy assignment of the entire microbial community than short sequencing reads obtained from the hypervariable region (V3-V4) amplicon library. Aside from this result, high proportion (81–85%) of unmapped pass 2D reads was observed due to the current error rate of MinION platform and may directly give rise to assigning an unrelated organism or low-throughput. Consequently, the sequencing accuracy may influence the further analysis of the microbiome composition.
When comparing nanopore sequencing results from the two biological duplicates, however, most abundant taxonomic units (89%) did not exhibit significant differences (log2 fold-change, lower than −1 or higher than 1), except one phylotype and three taxonomic units, and the sequencing data were highly similar (R2 = 0.829–0.909) at all taxonomic resolutions, except the species level. At the species level, we observed that nanopore sequencing data had better resolution than the short-read sequencing data. In particular, we obtained Bifidobacterium animalis and Bifidobacterium pseudolongum, which have been reported as key members of the gut microbial community49, at the species level. This result showed that the identification of some strains at the species level could be impossible when using a partial region of the 16S rRNA (e.g., Neisseria meningitidis and Neisseria lactamica). Therefore, full-length 16s rRNA sequencing provided higher taxonomic resolution than second-generation sequencing. Moreover, within the clinical setting, the long reads from the 16S rRNA sequence may be promising. According to our data, if appropriate clinical standard references are constructed, such references could be used as specific microbial markers to profile individual microbiomes without the computational burden of forming OTUs.
With long reads, nanopore sequencing (e.g., MinION) has been shown to have significant advantages, such as small size, low cost, rapid library construction (<3 h), and real-time detection. These advantages suggest the potential for identification of members of a microbiota community in situ. Additionally, nanopore sequencing can eliminate sample storage steps; such steps may cause loss of important species used as biomarkers (e.g., Bacteroidetes) due to long-term freezing of samples50. Similarly, previous studies have supported that the MinION sequencer can be used as a real-time molecular diagnostic device14,16. Current limitations, such as accuracy and throughput, will be resolved by advanced nanopore chemistry. For example, the SQK-MAP-006 sequencing kit (Oxford Nanopore Technologies), which was used in this paper, provides improved results with 2-fold faster sequencing speed, new hairpin-motor adaptors for high 2D yield, and advanced modeling of base-calling algorithm (6-mers) compared with previous chemistries (SQK–MAP005 and SQK–MAP005.1). Also, next generation chemistry (R9, Oxford Nanopore Technologies) contains a new pore protein, such as the CsgG protein51,52, which is predicted to provide more optimal sensing regions for a DNA strand as compared with MspA and α-hemolysin. It will be helpful in improving the sequencing accuracy and throughputs of the nanopore sequencing platform. Further, automatic devices (e.g. VolTRAX™, Oxford Nanopore Technologies) and improved library construction methods (e.g. adaptor-charged transposase mediated library preparation) will be used as the multiplex, fast, and high-throughput methods for preparing the nanopore sequencing library in the near future.
In conclusion, the full-length 16S rRNA amplicon sequencing with a nanopore sequencer allows rapid, accurate and efficient determination of the microbial diversity at the species level as successfully demonstrated for determining microbiome composition. We anticipate that this method will broaden the utility of microbiome composition analysis for biological, clinical, and environmental origins.
Methods
Metagenomic DNA extraction
Microbial metagenomic DNA was extracted with a PowerSoil DNA Isolation Kit (MoBio, Carlsbad, CA, USA). Snap-frozen fecal samples stored at −80 °C were added to PowerBead tubes and treated as described in the manufacturer’s instructions. The tubes containing the pretreated samples were placed into a benchtop homogenizer FastPrep-24 5G (MP Biomedicals, Santa Ana, CA, USA) and disrupted for 40 s, three times, with a 1-min rest period. The machine speed setting was 6 m/s, and a QuickPrep adapter was used. The concentration of the extracted DNA was measured with a Nanodrop 2000 (Thermo Scientific, Waltham, MA, USA).
Nanopore sequencing library construction
16S-specific primers adapted from S-D-bact-0008-c-S20 and S-D-bact-1391-a-A-17 were used for broad-taxonomic range amplification of the bacterial 16S rRNA gene24. For polymerase chain reaction (PCR) amplification using Phusion High-Fidelity polymerase (Thermo Scientific), 3 μg of the 16S-specific primers were added to the 30 ng of metagenomic DNA. The amplification was monitored with SYBR Green gel staining solution (Invitrogen, Grand Island, NY, USA) on a CFX96 Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA) and stopped at the beginning of the saturation point to reduce PCR bias. The PCR conditions were as follows: 98 °C for 30 s; 15 cycles of 98 °C for 10 s, 47 °C for 30 s, and 72 °C for 60 s; followed by 72 °C for 5 min. PCR products were purified using a MinElute Gel Extraction kit (Qiagen, Venlo, Netherlands). The amount of recovered DNA was quantified using a Qubit 3.0 fluorometer (Life Technologies, Carlsbad, CA, USA), and 300 ng of purified amplicon DNA with 5 μL of internal control DNA (DNA CS from the SQK-MAP006 kit) was used as input for generation of MinION-compatible libraries. The amplicons were end repaired using the NEBNext End Repair module (NEB, Ipswich, MA, USA). Subsequently, the end-repaired amplicon was dA-tailed using the NEBNextdA-tailing module (NEB) at 37 °C for 10 min. Then, 30 μL dA-tailed DNA, 50 μL Blunt/TA ligase master mix (NEB), 10 μL of Adapter Mix (Oxford Nanopore Technologies, Oxford, UK), and 2 μL HP adapter (Oxford Nanopore Technologies) were added and incubated at room temperature for 10 min. The adaptor-ligated libraries were purified using MyOne C1-beads (Thermo Scientific), eluted in 25 μL elution buffer (Oxford Nanopore Technologies), and incubating at 37 °C for 10 min.
Nanopore sequencing and base-calling
Each nanopore sequencing library was run on a FLO-MAP103 flow cell after performing platform QC analysis. The FLO-MAP103 flow cell was primed twice with a mixture of Fuel Mix (Oxford Nanopore Technologies). Ten microliters of the amplicon library (8 ng) was diluted in 75 μL of 2× running buffer with 61 μL nuclease-free water and 4 μL Fuel Mix. A 48 h sequencing protocol was initiated using the MinION control software, MinKNOW (version 0.50.2.15). Raw FAST/HDF files were base-called by the Metrichor agent two-dimensional (2D) base-calling workflow. Pass 2D reads were converted for downstream analysis into a FASTA format using poretools53.
Illumina sequencing
A 16S rRNA sequencing library was constructed according to the 16S metagenomics sequencing library preparation protocol (Illumina, San Diego, CA, USA) targeting the V3 and V4 hypervariable regions of the 16S rRNA gene, as were also used by the Human Microbiome Project8. KAPA HiFi HotStart ReadyMix (KAPA Biosystems, Wilmington, MA, USA) and Agencourt AMPure XP system (Beckman Coulter Genomics, Brea, CA, USA) were used for PCR and purification of the PCR product, respectively. The initial PCR was performed with 12 ng template DNA using region-specific primers shown to have compatibility with Illumina index and sequencing adapters (forward primer: 5′-TCGTCGGCAGCGTCAGATGTGTATAAGAGACAGTCGTCGGCAGCGTCAGATGTGTATAAGAGACAGCCTACGGGNGGCWGCAG-3′; reverse primer: 5′-GTCTCGTGGGCTCGGAGATGTGTATAAGAGACAGGTCTCGTGGGCTCGGAGATGTGTATAAGAGACAGGACTACHVGGGTATCTAATCC-3′). After magnetic bead-based purification of PCR products, the second PCR was performed using primers from a Nextera XT Index Kit (Illumina) with a limited cycle. Subsequently, purified PCR products were visualized using gel electrophoresis and quantified with a Qubit dsDNA HS Assay Kit (Thermo Scientific) on a Qubit 3.0 fluorometer. The pooled samples were run on an Agilent 2200 TapeStation (Agilent Technologies, Santa Clara, CA, USA) for quality analysis prior to sequencing. The sample pool (4 nM) was denatured with 0.2 N NaOH, diluted further to 4 pM, and combined with 20% (v/v) denatured 4 pM PhiX, prepared following Illumina guidelines. Samples were sequenced on the MiSeq sequencing platform (Illumina) using a 2 × 250 cycle V3 kit, following standard Illumina sequencing protocols.
Nanopore sequencing data analysis
For sequencing analysis, 2D reads were aligned against the GreenGenes 13_8 reference sequences using the LAST aligner v.658 with the following parameters: -q 1 -a 1 -b 1(match score of 1, gap opening penalty of 1, and gap extension penalty of 1). Alignment information was converted to maf files with maf-convert, which was packaged in LAST, to build .axt files. Samtools version 0.1.1954 was used to produce .bam files and alignment information from .axt files. For each read, the highest scoring alignment was retained and assigned with the taxonomic id of corresponding GreenGene reference sequences. The low abundance data of single mapped reads were discarded when considering assigned taxonomic units to reduce spurious taxonomic units. To assess the nanopore sequencing accuracy, total 2D reads were aligned against the phage lambda sequences (NC_001416.1), which were used as the reference for DNA CS with LAST (version 658) using the following parameters: -q 1 -a 1 -b 1. Samtools (version 0.1.19) and count-errors.py, obtained from the poretools repository53, were used to call variants in the region spanned by at least 80% of the strands. The mismatch positions in the sequencing reads aligned to the reference sequence were counted to measure the insertions, deletions, and substitutions. The sequencing accuracy was calculated as the number of matching nucleotides divided by the sum of matches, mismatches, insertions, and deletions aligned against the reference sequence.
Illumina sequencing data analysis
The QIIME pipeline (version 1.9.1) was used to process and filter multiplexed sequence reads. OTUs were clustered against GreenGenes 13_8 reference sequences, and reads failing to hit the reference were subsequently clustered de novo at the 97% similarity level using the UCLUST greedy algorithm. Chimeric sequences were identified by the UCHIME algorithm included in the free version of USEARCH61 and removed. OTU sequences were aligned using PYNAST. OTU taxonomy was determined using the Ribosomal Database Project classifier retrained toward the GreenGenes database. To avoid biases generated by differences in sequencing depth and removal of plastid sequences, the OTU table was rarified to an even depth of 90,000 sequences per sample in comparisons of all sample types.
Animal Experiments
Fifty-week-old male C57BL/6J mice were housed in a room maintained at a condition with 12 h light/dark cycle at 22 ± 2 °C, and allowed free access to unlimited food and water in a specific pathogen-free facility of the Korea Research Institute of Bioscience and Biotechnology (KRIBB, Daejeon, Korea). All mice were humanely euthanized by CO2 asphyxiation and then, the feces samples were directly obtained from the each mouse colon following necropsy. All animal experiments were approved by the Institutional Animal Use and Care Committee of KRIBB and were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication, 8th Edition, 2011).
Additional Information
How to cite this article: Shin, J. et al. Analysis of the mouse gut microbiome using full-length 16S rRNA amplicon sequencing. Sci. Rep. 6, 29681; doi: 10.1038/srep29681 (2016).
References
Ursell, L. K., Metcalf, J. L., Parfrey, L. W. & Knight, R. Defining the human microbiome. Nutr Rev 70 Suppl 1, S38–S44 (2012).
Kuczynski, J. et al. Experimental and analytical tools for studying the human microbiome. Nat Rev Genet 13, 47–58 (2011).
Perez-Lopez, A., Behnsen, J., Nuccio, S. P. & Raffatellu, M. Mucosal immunity to pathogenic intestinal bacteria. Nat Rev Immunol 16, 135–148, 10.1038/nri.2015.17 (2016).
Qin, J. et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60 (2012).
Hsiao, E. Y. et al. Microbiota Modulate Behavioral and Physiological Abnormalities Associated with Neurodevelopmental Disorders. Cell 155, 1451–1463 (2013).
Zhang, C. et al. Interactions between gut microbiota, host genetics and diet relevant to development of metabolic syndromes in mice. ISME J 4, 232–241 (2010).
Consortium, H. M. P. In Nature 486 207–214 (2012).
Consortium, H. M. P. Structure, function and diversity of the healthy human microbiome. Nature 486, 207–214 (2012).
Schloss, P. D. The effects of alignment quality, distance calculation method, sequence filtering, and region on the analysis of 16S rRNA gene-based studies. PLoS Comput Biol 6, e1000844 (2010).
Branton, D. et al. The potential and challenges of nanopore sequencing. Nat Biotechnol 26, 1146–1153 (2008).
Goodwin, S. et al. Oxford Nanopore sequencing, hybrid error correction, and de novo assembly of a eukaryotic genome. Genome Res 25, 1750–1756 (2015).
Loman, N. J., Quick, J. & Simpson, J. T. A complete bacterial genome assembled de novo using only nanopore sequencing data. Nat Methods 12, 733–735 (2015).
Karlsson, E., Lärkeryd, A., Sjödin, A., Forsman, M. & Stenberg, P. Scaffolding of a bacterial genome using MinION nanopore sequencing. Sci Rep 5, 11996 (2015).
Quick, J. et al. Rapid draft sequencing and real-time nanopore sequencing in a hospital outbreak of Salmonella. Genome Biol 16, 114 (2015).
Wang, J., Moore, N. E., Deng, Y.-M., Eccles, D. A. & Hall, R. J. MinION nanopore sequencing of an influenza genome. Front Microbiol 6, 766 (2015).
Greninger, A. L. et al. Rapid metagenomic identification of viral pathogens in clinical samples by real-time nanopore sequencing analysis. Genome Med 7, 99 (2015).
Kilianski, A. et al. Bacterial and viral identification and differentiation by amplicon sequencing on the MinION nanopore sequencer. GigaScience 4, 12 (2015).
Hargreaves, A. D. & Mulley, J. F. Assessing the utility of the Oxford Nanopore MinION for snake venom gland cDNA sequencing. PeerJ 3, e1441 (2015).
Bolisetty, M. T., Rajadinakaran, G. & Graveley, B. R. Determining exon connectivity in complex mRNAs by nanopore sequencing. Genome Biol 16, 1–12 (2015).
Ashton, P. M. et al. MinION nanopore sequencing identifies the position and structure of a bacterial antibiotic resistance island. Nat Biotechnol 33, 296–300 (2014).
Benítez-Páez, A., Portune, K. J. & Sanz, Y. Species-level resolution of 16S rRNA gene amplicons sequenced through the MinIONTM portable nanopore sequencer. GigaScience 1–9 (2016).
Fadrosh, D. W. et al. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2, 6, 10.1186/2049-2618-2-6 (2014).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7, 335–336, 10.1038/nmeth.f.303 (2010).
Klindworth, A. et al. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res 41, e1 (2013).
Jain, M. et al. Improved data analysis for the MinION nanopore sequencer. Nat Methods 12, 351–356 (2015).
Szalay, T. & Golovchenko, J. A. De novo sequencing and variant calling with nanopores using PoreSeq. Nat Biotechnol 33, 1087–1091 (2015).
Frith, M. C., Hamada, M. & Horton, P. Parameters for accurate genome alignment. BMC Bioinf 11, 80 (2010).
Mikheyev, A. S. & Tin, M. M. Y. A first look at the Oxford Nanopore MinION sequencer. Mol Ecol Resour 14, 1097–1102 (2014).
DeSantis, T. Z. et al. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72, 5069–5072 (2006).
Schloss, P. D. & Westcott, S. L. Assessing and Improving Methods Used in Operational Taxonomic Unit-Based Approaches for 16S rRNA Gene Sequence Analysis. Appl Environ Microbiol 77, 3219–3226 (2011).
Sul, W. J. et al. Bacterial community comparisons by taxonomy-supervised analysis independent of sequence alignment and clustering. Proc Natl Acad Sci USA 108, 14637–14642 (2011).
Hildebrand, F. et al. Inflammation-associated enterotypes, host genotype, cage and inter-individual effects drive gut microbiota variation in common laboratory mice. Genome Biol 14, R4 (2013).
Langille, M. G. et al. Microbial shifts in the aging mouse gut. Microbiome 2, 50 (2014).
Ravussin, Y. et al. Responses of gut microbiota to diet composition and weight loss in lean and obese mice. Obesity 20, 738–747 (2012).
Seekatz, A. M. et al. Recovery of the gut microbiome following fecal microbiota transplantation. mBio 5, e00893–00814 (2014).
Dao, M. C. et al. Akkermansia muciniphilaand improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut 65, 426–436 (2016).
Flint, H. J., Scott, K. P., Duncan, S. H., Louis, P. & Forano, E. Microbial degradation of complex carbohydrates in the gut. Gut Microbes 3, 289–306 (2012).
Larsbrink, J. et al. A discrete genetic locus confers xyloglucan metabolism in select human gut Bacteroidetes. Nature 506, 498–502 (2014).
Oh, P. L. et al. Diversification of the gut symbiont Lactobacillus reuteri as a result of host-driven evolution. ISME J 4, 377–387 (2010).
Scher, J. U. et al. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. eLife 2, e01202 (2013).
Schwiertz, A., Le Blay, G. & Blaut, M. Quantification of Different Eubacteriumspp. in Human Fecal Samples with Species-Specific 16S rRNA-Targeted Oligonucleotide Probes. Appl Environ Microbiol 66, 375–382 (2000).
Turnbaugh, P. J. et al. A core gut microbiome in obese and lean twins. Nature 457, 480–484 (2009).
Vasquez, N., Suau, A., Magne, F., Pochart, P. & Pélissier, M.-A. Differential Effects of Bifidobacterium pseudolongum Strain Patronus and Metronidazole in the Rat Gut. Appl Environ Microbiol 75, 381–386 (2009).
Million, M. et al. Obesity-associated gut microbiota is enriched in Lactobacillus reuteri and depleted in Bifidobacterium animalis and Methanobrevibacter smithii. Int J Obes 36, 817–825 (2012).
Larkin, M. A. et al. Clustal W and Clustal X version 2.0. Bioinformatics 23, 2947–2948 (2007).
Franzosa, E. A. et al. Relating the metatranscriptome and metagenome of the human gut. Proc Natl Acad Sci USA 111, E2329–E2338, 10.1073/pnas.1319284111 (2014).
Cao, H. X. et al. Metatranscriptome analysis reveals host-microbiome interactions in traps of carnivorous Genlisea species. Front Microbiol 6, 526, 10.3389/fmicb.2015.00526 (2015).
Fichot, E. B. & Norman, R. S. Microbial phylogenetic profiling with the Pacific Biosciences sequencing platform. Microbiome 1, 10, 10.1186/2049-2618-1-10 (2013).
Turroni, F. et al. Bifidobacterium bifidum as an example of a specialized human gut commensal. Front Microbiol 5, 437 (2014).
Bahl, M. I., Bergström, A. & Licht, T. R. Freezing fecal samples prior to DNA extraction affects the Firmicutes to Bacteroidetes ratio determined by downstream quantitative PCR analysis. FEMS Microbiol Lett 329, 193–197 (2012).
Goyal, P. et al. Structural and mechanistic insights into the bacterial amyloid secretion channel CsgG. Nature 516, 250–253 (2014).
Deamer, D., Akeson, M. & Branton, D. Three decades of nanopore sequencing. Nat Biotechnol 34, 518–524 (2016).
Loman, N. J. & Quinlan, A. R. Poretools: a toolkit for analyzing nanopore sequence data. Bioinformatics 30, 3399–3401 (2014).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Tamura, K., Stecher, G., Peterson, D., Filipski, A. & Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol 30, 2725–2729 (2013).
Acknowledgements
This work was supported by the Intelligent Synthetic Biology Center of Global Frontier Project (2011-0031957 to B.-K.C. and 2011-0031963 to S.Y.L.) through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT, and Future Planning, and the KRIBB Research Initiative Program. Funding for the open access charge was provided by the Intelligent Synthetic Biology Center.
Author information
Authors and Affiliations
Contributions
B.-K.C. designed and supervised the project; J.S., S.L. and M.-J.G. performed experiments; J.S., S.L. and B.-K.C. analyzed the data; J.S., S.L., S.Y.L., S.C.K., C.-H.L. and B.-K.C. wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Shin, J., Lee, S., Go, MJ. et al. Analysis of the mouse gut microbiome using full-length 16S rRNA amplicon sequencing. Sci Rep 6, 29681 (2016). https://doi.org/10.1038/srep29681
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep29681
This article is cited by
-
A robust microbiome signature for autism spectrum disorder across different studies using machine learning
Scientific Reports (2024)
-
Determining the most accurate 16S rRNA hypervariable region for taxonomic identification from respiratory samples
Scientific Reports (2023)
-
MinION, a portable long-read sequencer, enables rapid vaginal microbiota analysis in a clinical setting
BMC Medical Genomics (2022)
-
Investigation of memory-enhancing effects of Streptococcus thermophilus EG007 in mice and elucidating molecular and metagenomic characteristics using nanopore sequencing
Scientific Reports (2022)
-
Comprehensive 16S rRNA and metagenomic data from the gut microbiome of aging and rejuvenation mouse models
Scientific Data (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.