Abstract
Vimentin, an intermediate filament protein, is an intracellular protein that is involved in various cellular processes. Several groups have recently reported that vimentin also appears in the extracellular space and shows novel protein activity. We previously reported that denosomin improved motor dysfunction in mice with a contusive spinal cord injury (SCI). At the injured area, astrocytes expressing and secreting vimentin were specifically increased and axonal growth occurred in a vimentin-dependent manner in denosomin-treated mice. However, the axonal growth that was induced by extracellular vimentin was only investigated in vitro in the previous study. Here, we sought to clarify whether increased extracellular vimentin can promote the axonal extension related to motor improvement after SCI in vivo. Extracellular vimentin treatment in SCI mice significantly ameliorated motor dysfunction. In vimentin-treated mice, 5-HT-positive axons increased significantly at the rostral and central areas of the lesion and the total axonal densities increased in the central and caudal parts of the lesioned area. This finding suggests that increased axonal density may contribute to motor improvement in vimentin-treated mice. Thus, our in vivo data indicate that extracellular vimentin may be a novel neurotrophic factor that enhances axonal growth activity and motor function recovery after SCI.
Similar content being viewed by others
Introduction
Reconstruction of neuronal networks after trauma in the adult central nervous system (CNS) is still difficult to achieve in both clinical and basic science studies. Spinal cord injury (SCI) causes irreversible damage to the spinal cord and leads to serious motor, sensory and/or autonomic nervous system dysfunction due to disruption of the descending and ascending fiber tracts. Although it has been thought for decades that adult CNS axons do not have the plasticity needed to repair lesions through regeneration, numerous studies have suggested that CNS axons have the ability to regenerate and that the regrowth of injured and/or spared axons may lead to locomotor dysfunction recovery1,2,3. In contrast, several types of inhibitory factors that are derived from glial cells have been shown to potently obstruct axonal restoration4,5. Several studies have confirmed that glial scarring, which is an inhibitory obstacle, is formed by reactive astrocytes, which generate chondroitin sulfate proteoglycans (CSPGs); these molecules are known to inhibit axonal growth6,7,8,9,10. Thus, overcoming CSPG inhibition is necessary to achieve functional recovery after SCI.
In a previous study, we developed a novel compound, denosomin, that exhibited axonal growth activity in cultured neurons11. Denosomin treatment in SCI mice improved motor function and both extended and enhanced axonal growth (the serotonin (5-HT)-positive tract)12. Interestingly, denosomin treatment increased astrocyte proliferation at the injury site and also promoted vimentin release from those astrocytes12. Elongated axons following denosomin treatment were well correlated with extracellular vimentin12. Prior to that study, the functional role of secreted vimentin was unknown. However, the extracellular addition of recombinant vimentin to cultured cortical neurons resulted in axonal growth that was even sustained on CSPG-coated dishes12.
Vimentin is an intermediate filament protein and is generally recognized as an intracellular protein that is involved in cellular processes, such as cell adhesion and cell migration13,14,15,16. Although two reports have suggested vimentin secretion from unstimulated astrocytes17,18, there have been no reports of the potential role of extracellular vimentin in specific signaling to cells. Our previous study demonstrated that extracellular vimentin directly binds and activates insulin-like growth factor 1 receptor (IGF1R) in neurons19. The new discovery that extracellular vimentin induces axonal growth in cultured cortical neurons suggests that extracellular vimentin may act as a novel nerve growth factor. Therefore, in this study, we sought to determine whether increased extracellular vimentin could promote axonal growth and locomotor recovery in SCI mice.
Results
Extracellular vimentin treatment improved motor dysfunction in contusive SCI mice
To investigate the effect of extracellular vimentin on motor improvement in SCI mice, recombinant vimentin or vehicle solution (ACSF; artificial cerebrospinal fluid) was continuously administered into the lateral ventricle using a micro-osmotic pump for 21 days following SCI.
The motor function in the right and left hindlimbs was scored separately using the Basso Mouse Scale (BMS) (Fig. 1a), Body Support Scale (BSS) (Fig. 1b) and Toyama Mouse Score (TMS) (Fig. 1c). The BMS, BSS and TMS scores of vimentin-treated mice gradually and significantly increased compared with control SCI mice over the 21-day course of administration. The F and P values of interaction effects between treatment and days after SCI were calculated as follows: F(2, 680) = 3.18; P < 0.0001 in BMS, F(2, 680) = 5.59; P < 0.0001 in BSS and F(2, 680) = 7.25; P < 0.0001 in TMS. Sham-operated mice received the maximum scores on all tests. We repeated these experiments twice and confirmed that a similar degree of motor dysfunction was reproducibly observed in our contusion model. Vimentin administration did not cause a loss in body weight (Fig. 1d) or induce abnormal behaviors in the mice (data not shown). At 21 days post-injury, the movements of the mice during walking were captured (Fig. 2). As shown in Fig. 2a, the ACSF-treated control mice dragged their legs during movement. As shown in the 0.9 s of sequentially captured images, the ACSF-treated control mice showed an abnormal walking gait. In contrast, the mice in the vimentin-treated group showed high ankle mobility and trunk stability during walking (Fig. 2b).
Vimentin enhances the hind limb function in SCI mice.
The BMS score (a), BSS score (b), TMS (c) and body weight (d) were measured. SCI mice received vimentin (red circles, 8 mice, 16 hind limbs, n = 16) or vehicle solution (grey circles, 10 mice, 20 hindlimbs, n = 20). Sham-operated mice were administered vehicle solution (open circles, 9 mice, 18 hindlimbs, n = 18). #P < 0.0001, drug × day interaction analyzed using a repeated-measures two-way ANOVA; *P < 0.05 compared with the vehicle-treated SCI mice on the same day using the Bonferroni post hoc test.
Vimentin treatment increased NF-H-positive axonal densities at the SCI site in SCI mice
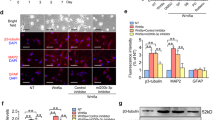
The injured spinal cords of vimentin-treated SCI mice were examined by immunohistochemistry. First, positive staining for NF-H (an axonal marker) was assessed at the lesion center (Fig. 3a). The NF-H-positive area was significantly higher in the center of the lesion in the vimentin-treated group than in the vehicle-treated and non-injured control groups (Fig. 3b). NF-H-positive staining was also measured in regions that were rostral and caudal to the lesion center (Fig. 3b). After vimentin treatment, the NF-H-positive axonal staining was significantly increased in the caudal area but not in the rostral area (Fig. 3c). Therefore, these results suggest that extracellular vimentin promotes axonal elongation at the lesion center and the area caudal to the lesion center.
Vimentin enhances the axonal density in SCI mice.
Sagittal sections of the spinal cord of vehicle-treated or vimentin-treated SCI mice were immunostained for NF-H (a,b). The relative areas of immunostaining were measured in three locations of the spinal cord: at the center of the lesion and the sites that were 2 mm rostral and 2 mm caudal to the injury center (c). The white dashed squares are magnified in the right panels in (a). The yellow dashed lines in (a,b) indicate the outlines of the spinal cords. The blue dashed area in (a) corresponds to the glial scar region. *P < 0.05 vs. vehicle, unpaired t-test (two-tailed), one-way ANOVA Bonferroni post hoc test. Veh, vehicle; Vim, vimentin. In (c), Veh, n = 5; Vim, n = 5. The white scale bar indicates 500 μm and the yellow bar indicates 100 μm.
Vimentin treatment increased the 5-HT-positive axonal densities in the raphespinal tracts in the spinal cord tissues of SCI mice
Next, spinal cord sections were immunostained for 5-HT to visualize raphespinal tracts in the spinal cord because these tracts are known to predominantly regulate locomotor function19,20,21. Positive 5-HT staining was detected in the lesion center and in regions 2 mm rostral and caudal to the lesion center (Fig. 4a). Many more 5-HT-positive axons were detected along the scar rim and in the rostral area in the vimentin-treated mice than in the vehicle-treated group. The quantitative evaluation of 5-HT-positive fiber-like staining is shown in Fig. 4b. Compared with the vehicle-treated group, the vimentin-treated animals had a significantly larger 5-HT-positive area at sites that were 2 mm rostral and caudal to the lesion center. At the lesion center, the 5-HT staining increased non-significantly after vimentin treatment. Compared with the 5-HT staining in the non-injured control group, the 5-HT-positive areas in the vimentin-treated group were larger at the lesion center and rostral to the lesion center (Fig. 4b). 5-HT-positive axons rarely crossed over the glial scar in either group; however, at the rostral site, the vimentin-treated mice showed slightly more 5-HT-positive axons that extended to the caudal region (Fig. 4b). The scar area was defined as the inside space that was surrounded by GFAP-positive astrocytes. The scar sizes of the analyzed areas did not differ between groups (Fig. 4c). In addition, the area of GFAP-positive reactive astrocytes was not different between groups (Fig. 4d). These results indicate that extracellular vimentin enhances the elongation of 5-HT-positive fiber tracts.
Vimentin enhances the 5-HT-positive axonal density in SCI mice.
Sagittal sections of the spinal cord of control, vehicle-treated or vimentin-treated SCI mice were immunostained for 5-HT (a). The relative areas of immunostaining were measured in three locations of the spinal cord: at the center of the lesion and at sites that were 2 mm rostral and 2 mm caudal to the lesion center (b). The blue dashed lines in (a) indicate the outlines of the glial scar. The yellow dashed lines in (a) indicate the outlines of the spinal cord. The size of scar area was measured (c). The area of GFAP-positive reactive astrocytes was measured (d). *P < 0.05 vs. vehicle, unpaired t-test (two-tailed), one-way ANOVA Bonferroni post hoc test. Veh, vehicle; Vim, vimentin; In (b), Veh, n = 5; Vim, n = 5. In (c,d), Vehicle, n = 10; Vimentin, n = 6. The scale bar indicates 500 μm.
Discussion
We previously demonstrated that denosomin increased the number of vimentin-expressing astrocytes inside the glial scars of SCI mice and that 5-HT-positive axonal growth occurred in a vimentin-dependent manner12. Here, we showed that extracellular vimentin promoted axonal growth and also ameliorated motor dysfunction in SCI mice. This is the first report of the effects of extracellular vimentin in an in vivo SCI model.
Spinal motor neurons terminate in the skeletal muscle in hindlimbs and are regulated by multiple descending tracts, propriospinal neurons and interneurons20,21. The serotonergic raphespinal tract is one of the main descending tracts that regulate voluntary movement22,23,24. Extracellular vimentin administration in the SCI mice enhanced the growth of 5-HT-positive axons in the areas that were rostral and caudal to the lesion (Fig. 4). Furthermore, at the lesion center, the number of 5-HT-positive axons increased following vimentin treatment. NF-H-positive axons were notably increased both at the lesion center and at the caudal area following vimentin treatment (Fig. 3). Because NF-H-positive staining should appear in a majority of axons, the increase in axons at the center and caudal areas may reflect the elongation from a variety of sources, including interneurons, propriospinal neurons, and/or descending tracts, such as the raphespinal, corticospinal or rubrospinal tract. The regeneration of axons descending from the brain and propriospinal relay connections are important for functional recovery after SCI25,26. In the mammalian spinal cord, the locomotor circuit is functionally organized by interneurons27. Because projections to motor neurons by the serotonergic raphespinal tracts are established by either direct or interneuron-mediated indirect pathways, it is possible that some of the observed elongated 5-HT-positive axons may innervate motor neurons via interneurons in the spinal cord of extracellular vimentin-treated mice. We supposed that the extracellular administration of vimentin in the cerebral ventricle might affect at multiple levels of the spinal cord and brain areas. IGF1R was shown to be a receptor of extracellular vimentin19 and it is expressed widely throughout the brain and spinal cord28. Therefore, vimentin most likely stimulates axonal growth via IGF1R through a variety of mechanisms.
Our previous reports have shown that extracellular vimentin induced axonal growth in primary cultured neurons and that this growth even persisted when the neurons were grown on CSPG substrates12. Additionally, we showed that denosomin-treated astrocytes secreted vimentin into the extracellular space and that 5-HT-positive axons were especially increased in vimentin-positive areas. These previously published results suggest that extracellular vimentin could potentially contribute to axonal growth in injured areas. Other groups have reported the spontaneous secretion of vimentin from astrocytes without stimulation17,18. Furthermore, in our previous study, cultured astrocytes released modest levels of vimentin without stimulation. In contrast, denosomin treatment resulted in a four-fold increase in vimentin secretion12. The vimentin level is known to increase after contusive injury in the spinal cord4. These results may suggest that the extracellular vimentin that is secreted from astrocytes is also related to the spontaneous partial improvement of SCI. However, the exogenous administration of extracellular vimentin may be required for significant recovery.
Our preliminary data show that a single direct microinjection of vimentin to the SCI site was effective in improving motor activity in SCI mice. In the current study, the continuous i.c.v. delivery of vimentin was performed using a micro-osmotic pump and the vimentin-treated group showed a significant increase in axonal density at the SCI site. This result suggests that an increase in extracellular vimentin contributes to the enhancement of axonal densities in injured spinal cord tissues.
As a mechanism of vimentin-induced axonal growth, we have already demonstrated that IGF1R is the direct target of extracellular vimentin in cultured neurons19. IGF1R is broadly expressed in several types of cells in the CNS29,30. The IGF1R knockout mouse shows abnormal morphology and failure of some tissues and organs during various developmental stages31, indicating that IGF1R has an important role in cell growth and/or development. In preliminary experiments, an IGF1R inhibitor, IGF1-analog, showed lethality when administered by i.c.v. injection (data not shown). Therefore, to investigate whether the vimentin-induced effect is mediated by IGF1R in vivo, IGF1R conditional knockout mice should be used32.
Because neurotrophic factors are important for activating axonal growth33, the vimentin-mediated release of neurotrophic factors might be involved in the axonal growth that is induced by vimentin injection. However, our previous in vitro data showed that vimentin did not increase the neuronal secretion of IGF1, IGF2 and insulin, which are physiological ligands for IGF1R19. Additionally, our unpublished in vitro data showed that vimentin-treated astrocytes and vimentin-treated microglia do not secrete axonal growth facilitators. This finding suggests that the vimentin-induced axonal growth and motor function improvements that were observed in SCI mice might not be mediated by other neuron-, astrocyte- or microglia-derived neurotrophic factors. Whether extracellular vimentin influences additional cell types in SCI mice requires further investigation.
Many studies have shown a relationship between vimentin and neurons, but most studies have focused on the role of vimentin as an intracellular protein. One report showed that the expression of vimentin in neuroblastoma cells increased the number of axonal neurites34. During development, the corticospinal tract elongates in proximity to vimentin-positive areas35. Additionally, Hsu et al. showed that vimentin-positive astrocytes induced axonal regrowth after spinal cord hemisection and Schwann cell implantation in adult rats36. Another group also suggested that increased vimentin expression is involved in spontaneous recovery after contusive SCI4. Together, these reports indicate that vimentin increases axon growth, although these studies focused on its role as an intracellular molecule rather than an extracellular one. In contrast, vimentin secretion was detected in not only astrocytes17,18 but also activated macrophages37 and vascular endothelial cells38. Vimentin is categorized as a ‘moonlighting’ or ‘gene-sharing’ protein that has been increasingly found in unexpected cellular locations39,40. Some of these proteins are derived from a single gene but adopt different properties and functional roles. Vimentin is now recognized as one of these multi-functional proteins, although the differences between its intracellular and extracellular functions and its secretory pathway remain unclear41. Extracellular vimentin facilitates axonal growth and increases the number of reactive astrocytes in damaged CNS tissue areas. If the vimentin-secreting function is enhanced in astrocytes, vimentin may contribute to axonal elongation after injury. Further elucidation of the detailed mechanisms underlying the function of vimentin will be essential for developing vimentin-based drug and/or gene therapies.
Our findings suggest that extracellular vimentin facilitates axonal growth and improves motor dysfunction in SCI mice. These data provide unprecedented scientific evidence about vimentin and suggest that it may be a potential therapeutic target for promoting axonal growth.
Materials and Methods
All experiments were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals of the University of Toyama and the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals. The Committee for Animal Care and Use of the University of Toyama approved the study protocols. All efforts were made to minimize the number of animals used.
Animals and SCI model experiments
Six- to seven-week-old female ddY mice (SLC, Shizuoka, Japan) were used for the SCI experiments. The mice were housed with ad libitum access to food and water and were maintained under constant environmental conditions (22 ± 2 °C, 50 ± 5% humidity and a 12-hr light: 12-hr dark cycle starting at 07:00). The surgical operations for producing contusive SCI were performed as previously described12 with slight modifications. The mice were anesthetized with trichloroacetaldehyde monophosphate (500 mg/kg, i.p.). After laminectomy at the T10 level, contusion injuries were produced by dropping a 6.5 g weight from a height of 3 cm onto the exposed L1 level using a stereotaxic instrument (Narishige, Tokyo, Japan). The incision was closed with sutures.
After SCI surgery, surgical procedures to place the cannula and micro-osmotic pump were performed. The mice were placed in a stereotaxic apparatus to keep the head in a fixed position. The scalp was shaved, followed by a sagittal midline incision to expose the skull. A cannula (Brain Infusion Kit 3, Alzet, Cupertino, CA, USA) was positioned into a lateral ventricle at the following coordinates: bregma −0.22 mm, lateral to the left +1 mm and −2.5 mm depth. The free end of the cannula was connected to a micro-osmotic pump (Alzet model 1004) via a 3.5-cm piece of polyvinylchloride (PVC) tubing (Alzet). The pump was placed into a subcutaneous pocket on the back of the mouse. The cannula was fixed to the skull with Loctite 454 cyanoacrylate. The infusion rate of the micro-osmotic pump was 0.11 μl/hr. The filled pumps were incubated in 0.9% sterile saline at 37 °C for at least 16 hr in a CO2 incubator before implantation. For the vehicle-treated group, the micro-osmotic pump and connected PNC tube were filled with artificial cerebrospinal fluid (ACSF) containing 130 mM NaCl, 24 mM NaHCO3, 3.5 mM KCl, 1.3 mM NaH2PO4, 2 mM CaCl2, 2 mM MgCl2 · 6 H2O and 10 mM glucose at pH 7.4. For the vimentin-treated group, the micro-osmotic pump and tube were filled with 1 μg of recombinant human vimentin (ProSpec, Rehovot, Israel) that was dissolved in ACSF. The delivery rate of the solution from the pump was 0.11 μl/hr. In this way, the concentration of vimentin that was delivered to the CSF was always 61 ng/ml. Previously, our study showed that the effective concentration of extracellular vimentin for axonal growth was 10–200 ng/ml19. During and after surgery, the mice were placed on a heating pad to maintain body temperature.
Behavioral evaluation
For behavioral scoring after surgery, the mice were individually placed in an open field (42 cm × 48 cm × 15 cm) and observed for 5 min once per day for a total of 21 days. Open-field locomotion was evaluated using three scoring methods: the 0–8 scale BMS score (without the tail score), which evaluates ankle movement and walking stability42; the 0–4 scale BSS score, whose criteria we developed to assess the degree of hindlimb muscle strength for support of the body trunk12; and the 0–30 scale TMS, the new criteria that we developed by combining and modifying the BMS and BSS scores43. Animals were allowed to move freely in the plastic box. Movements of the left and right hindlimbs were evaluated independently.
Antibodies
The primary antibodies for immunohistochemistry included a rabbit anti-neurofilament-H (NF-H) polyclonal antibody (dilution 1:1000; Chemicon, Temecula, CA, USA), a mouse anti-GFAP monoclonal antibody (clone G-A-5, dilution 1:1000; Sigma, St. Louis, MO, USA) and a rabbit anti-5-HT polyclonal antibody (dilution 1:3000; Sigma). The secondary antibodies that were used included Alexa Fluor 594-conjugated goat anti-mouse IgG1 (dilution 1:400) and Alexa Fluor 488-conjugated goat anti-rabbit IgG (dilution 1:400; Invitrogen, Carlsbad, CA, USA).
Immunohistochemistry
After behavioral scoring, the mice were deeply anesthetized and fixed by transcardial perfusion with 4% paraformaldehyde in phosphate-buffered saline. Serial spinal cord slices were obtained as 14-μm sagittal sections using a cryostat (CM3050S, Leica, Heidelberg, Germany). The slices were fixed with 4% paraformaldehyde and stained with antibodies to detect NF-H- or 5-HT-positive axons or GFAP-positive astrocytes. The fluorescence images were captured at a 670 × 890 μm size with a 10 × NA 0.45 dry objective lens (Plan-Apochromat, Carl Zeiss, Oberkochen, Germany) and CCD camera (AxioCam MRm) on an inverted microscope (Axio Observer Z1, Carl Zeiss). The captured images were converted and tiled using the AxioVision Z-stack and Mosaix software system (AxioVision 4.8, Carl Zeiss). The optical densities of the 5-HT- and NF-H-positive axons were determined using ImageJ software (NIH). The center of each lesion was determined based on the pattern of GFAP staining. Fluorescence images were then also captured at 2 mm rostral and 2 mm caudal to the injury center.
Statistical analysis
Statistical comparisons were performed using repeated-measures two-way ANOVA followed by the Bonferroni post hoc test, a one-way ANOVA followed by the Bonferroni post hoc test or an unpaired (two-tailed) t-test. GraphPad Prism 5 (GraphPad) was used for the statistical analyses. P-values less than 0.05 were considered significant. The data are presented as the mean ± SEM.
Additional Information
How to cite this article: Shigyo, M. and Tohda, C. Extracellular vimentin is a novel axonal growth facilitator for functional recovery in spinal cord-injured mice. Sci. Rep. 6, 28293; doi: 10.1038/srep28293 (2016).
References
David, S. & Aguayo, A. J. Axonal elongation into peripheral nervous system “bridges” after central nervous system injury in adult rats. Science 214, 931–933 (1981).
Bradbury, E. J. & McMahon, S. B. Spinal cord repair strategies: why do they work? Nat. Rev. Neurosci. 7, 644–653 (2006).
Carmel, J. B. & Martin, J. H. Motor cortex electrical stimulation augments sprouting of the corticospinal tract and promotes recovery of motor function. Front. Integr. Neurosci. 8, 51 (2014).
Bareyre, F. M. & Schwab, M. E. Inflammation, degeneration and regeneration in the injured spinal cord: insights from DNA microarrays. Trends Neurosci. 26, 555–563 (2003).
Gerin, C. G. et al. Combination strategies for repair, plasticity and regeneration using regulation of gene expression during the chronic phase after spinal cord injury. Synapse 65, 1255–1281 (2011).
Andrews, E. M., Richards, R. J., Yin, F. Q., Viapiano, M. S. & Jakeman, L. B. Alterations in chondroitin sulfate proteoglycan expression occur both at and far from the site of spinal contusion injury. Exp. Neurol. 235, 174–187 (2012).
Iseda, T. et al. Single, high-dose intraspinal injection of chondroitinase reduces glycosaminoglycans in injured spinal cord and promotes corticospinal axonal regrowth after hemisection but not contusion. J. Neurotrauma 25, 334–349 (2008).
Jones, L. L., Margolis, R. U. & Tuszynski, M. H. The chondroitin sulfate proteoglycans neurocan, brevican, phosphacan and versican are differentially regulated following spinal cord injury. Exp. Neurol. 182, 399–411 (2003).
McKeon, R. J., Schreiber, R. C., Rudge, J. S. & Silver, J. Reduction of neurite outgrowth in a model of glial scarring following CNS injury is correlated with the expression of inhibitory molecules on reactive astrocytes. J. Neurosci. 11, 3398–3411 (1991).
Tang, X., Davies, J. E. & Davies, S. J. Changes in distribution, cell associations and protein expression levels of NG2, neurocan, phosphacan, brevican, versican V2 and tenascin-C during acute to chronic maturation of spinal cord scar tissue. J. Neurosci. Res. 71, 427–444 (2003).
Matsuya, Y. et al. Synthesis of sominone and its derivatives based on an RCM strategy: discovery of a novel anti-Alzheimer’s disease medicine candidate “denosomin”. Org Lett 11, 3970–3973 (2009).
Teshigawara, K. et al. A novel compound, denosomin, ameliorates spinal cord injury via axonal growth associated with astrocyte-secreted vimentin. Br. J. Pharmacol. 168, 903–919 (2013).
Eckes, B. et al. Impaired mechanical stability, migration and contractile capacity in vimentin-deficient fibroblasts. J. Cell Sci. 111(Pt 13), 1897–1907 (1998).
Ivaska, J., Pallari, H. M., Nevo, J. & Eriksson, J. E. Novel functions of vimentin in cell adhesion, migration and signaling. Exp. Cell Res. 313, 2050–2062 (2007).
Tsuruta, D. & Jones, J. C. The vimentin cytoskeleton regulates focal contact size and adhesion of endothelial cells subjected to shear stress. J. Cell Sci. 116, 4977–4984 (2003).
Wang, K., Bekar, L. K., Furber, K. & Walz, W. Vimentin-expressing proximal reactive astrocytes correlate with migration rather than proliferation following focal brain injury. Brain Res. 1024, 193–202 (2004).
Cordero-Llana, O. et al. Clusterin secreted by astrocytes enhances neuronal differentiation from human neural precursor cells. Cell Death Differ. 18, 907–913 (2011).
Greco, T. M., Seeholzer, S. H., Mak, A., Spruce, L. & Ischiropoulos, H. Quantitative mass spectrometry-based proteomics reveals the dynamic range of primary mouse astrocyte protein secretion. J. Proteome Res. 9, 2764–2774 (2010).
Shigyo, M., Kuboyama, T., Sawai, Y., Tada-Umezaki, M. & Tohda, C. Extracellular vimentin interacts with insulin-like growth factor 1 receptor to promote axonal growth. Sci. Rep. 5, 12055 (2015).
Han, Q. et al. Plasticity of motor network and function in the absence of corticospinal projection. Exp. Neurol. 267, 194–208 (2015).
Lemon, R. N. Descending pathways in motor control. Annu. Rev. Neurosci. 31, 195–218 (2008).
Camand, E., Morel, M. P., Faissner, A., Sotelo, C. & Dusart, I. Long-term changes in the molecular composition of the glial scar and progressive increase of serotoninergic fibre sprouting after hemisection of the mouse spinal cord. Eur. J. Neurosci. 20, 1161–1176 (2004).
Jacobs, B. L., Martin-Cora, F. J. & Fornal, C. A. Activity of medullary serotonergic neurons in freely moving animals. Brain Res. Brain Res. Rev. 40, 45–52 (2002).
Ruschel, J. et al. Axonal regeneration. Systemic administration of epothilone B promotes axon regeneration after spinal cord injury. Science 348, 347–352 (2015).
Courtine, G. et al. Recovery of supraspinal control of stepping via indirect propriospinal relay connections after spinal cord injury. Nat. Med. 14, 69–74 (2008).
Stelzner, D. J. & Cullen, J. M. Do propriospinal projections contribute to hindlimb recovery when all long tracts are cut in neonatal or weanling rats? Exp. Neurol. 114, 193–205 (1991).
Dougherty, K. J. & Kiehn, O. Functional organization of V2a-related locomotor circuits in the rodent spinal cord. Ann. N. Y. Acad. Sci. 1198, 85–93 (2010).
Chung, Y. H. et al. Immunohistochemical study on the distribution of insulin-like growth factor I (IGF-I) receptor in the central nervous system of SOD1(G93A) mutant transgenic mice. Brain Res. 994, 253–259. (2003).
Fernandez, A. M. & Torres-Aleman, I. The many faces of insulin-like peptide signalling in the brain. Nat. Rev. Neurosci. 13, 225–239 (2012).
Walter, H. J., Berry, M., Hill, D. J. & Logan, A. Spatial and temporal changes in the insulin-like growth factor (IGF) axis indicate autocrine/paracrine actions of IGF-I within wounds of the rat brain. Endocrinology 138, 3024–3034 (1997).
Liu, J. P., Baker, J., Perkins, A. S., Robertson, E. J. & Efstratiadis, A. Mice carrying null mutations of the genes encoding insulin-like growth factor I (Igf-1) and type 1 IGF receptor (Igf1r). Cell 75, 59–72 (1993).
Gontier, G., George, C., Chaker, Z., Holzenberger, M. & Aid, S. Blocking IGF Signaling in Adult Neurons Alleviates Alzheimer’s Disease Pathology through Amyloid-beta Clearance. J. Neurosci. 35, 11500–11513 (2015).
McCall, J., Weidner, N. & Blesch, A. Neurotrophic factors in combinatorial approaches for spinal cord regeneration. Cell Tissue Res. 349, 27–37 (2012).
Dubey, M. et al. Reexpression of vimentin in differentiated neuroblastoma cells enhances elongation of axonal neurites. J. Neurosci. Res. 78, 245–249 (2004).
Joosten, E. A. & Gribnau, A. A. Astrocytes and guidance of outgrowing corticospinal tract axons in the rat. An immunocytochemical study using anti-vimentin and anti-glial fibrillary acidic protein. Neuroscience 31, 439–452 (1989).
Hsu, J. Y. & Xu, X. M. Early profiles of axonal growth and astroglial response after spinal cord hemisection and implantation of Schwann cell-seeded guidance channels in adult rats. J. Neurosci. Res. 82, 472–483 (2005).
Mor-Vaknin, N., Punturieri, A., Sitwala, K. & Markovitz, D. M. Vimentin is secreted by activated macrophages. Nat. Cell Biol. 5, 59–63 (2003).
Xu, B. et al. The endothelial cell-specific antibody PAL-E identifies a secreted form of vimentin in the blood vasculature. Mol. Cell. Biol. 24, 9198–9206 (2004).
Jeffery, C. J. Multifunctional proteins: examples of gene sharing. Ann. Med. 35, 28–35 (2003).
Piatigorsky, J. Gene sharing in lens and cornea: facts and implications. Prog. Retin. Eye Res. 17, 145–174 (1998).
Butler, G. S. & Overall, C. M. Proteomic identification of multitasking proteins in unexpected locations complicates drug targeting. Nat. Rev. Drug Discov. 8, 935–948 (2009).
Basso, D. M. et al. Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains. J. Neurotrauma 23, 635–659 (2006).
Shigyo, M., Tanabe, N., Kuboyama, T., Choi, S. H. & Tohda, C. New reliable scoring system, Toyama mouse score, to evaluate locomotor function following spinal cord injury in mice. BMC Res. Notes 7, 332 (2014).
Acknowledgements
This work was partially supported by Grants-in-Aid for Scientific Research (C) (23500439, C.T.) from the Ministry of Education, Culture, Sports, Science and Technology of Japan; Natural Medicine and Biotechnology Research of Toyama Prefecture, Japan (C.T.); and a Grant-in-Aid for the Cooperative Research Project from the Institute of Natural Medicine at the University of Toyama in 2014 and 2015 (C.T.).
Author information
Authors and Affiliations
Contributions
M.S. performed all experiments and wrote the manuscript. C.T. wrote the manuscript and conducted the study. Both authors approved the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Shigyo, M., Tohda, C. Extracellular vimentin is a novel axonal growth facilitator for functional recovery in spinal cord-injured mice. Sci Rep 6, 28293 (2016). https://doi.org/10.1038/srep28293
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep28293
This article is cited by
-
An anti vimentin antibody promotes tube formation
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.