Abstract
Nidelric pugio gen. et sp. nov. from the Cambrian Series 2 Heilinpu Formation, Chengjiang Lagerstätte, Yunnan Province, China, is an ovoid, sac-like metazoan that bears single-element spines on its surface. N. pugio shows no trace of a gut, coelom, anterior differentiation, appendages, or internal organs that would suggest a bilateral body plan. Instead, the sac-like morphology invites comparison with the radially symmetrical chancelloriids. However, the single-element spines of N. pugio are atypical of the complex multi-element spine rosettes borne by most chancelloriids and N. pugio may signal the ancestral chancelloriid state, in which the spines had not yet fused. Alternatively, N. pugio may represent a group of radial metazoans that are discrete from chancelloriids. Whatever its precise phylogenetic position, N. pugio expands the known disparity of Cambrian scleritome-bearing animals and provides a new model for reconstructing scleritomes from isolated microfossils.
Similar content being viewed by others
Introduction
Abundant, non-biomineralised and biomineralised (phosphatic, siliceous and carbonate) spines and sclerites are a characteristic component of small carbonaceous and small shelly fossil assemblages from Cambrian sedimentary rocks worldwide1,2,3,4. These elements represent the disarticulated scleritomes of diverse metazoans including chancelloriids5,6,7, palaeoscolecid worms8,9, lobopodians10, putative stem molluscs11,12, stem brachiopods13,14, euarthropods15 and various unidentified groups. As these spines and sclerites occur isolated, the overall morphology and affinities of many of these metazoans is problematic and their phylogenetic affinities may remain unclear even where such structures are recovered articulated, as in the halwaxiids from the Burgess Shale and Sirius Passet Lagerstätten16,17.
Amongst Cambrian spine-bearing metazoans, the chancelloriids represent an enigmatic group that have been interpreted as coeloscleritophorans18, poriferans19, ascidians20 and echinoderms21. Known from disarticulated sclerites in Cambrian sedimentary deposits worldwide7, exceptionally preserved chancelloriids have sac-like bodies that are covered with spines that insert into a surface integument5. Walcott22 was the first to suggest a sponge affinity for chancelloriids, but similarities between the spines of chancelloriids and those of coeloscleritophoran animals have argued against this assignment18, instead suggesting a relationship with halkieriids, siphogonuchitids or sachitids.
Here we describe a new spine-bearing metazoan from the Cambrian Series 2 Heilinpu Formation, Chengjiang Lagerstätte, Yunnan Province, China. Nidelric pugio gen. et sp. nov. closely resembles the sac-like morphology of chancelloriids, but differs in the single-element morphology of its spines. We examine the morphological significance of this species and assess its possible bearing on the early evolution of chancelloriids and on the origins of multi-element spines in the scleritomes of early metazoans.
Results
Geological Setting
The single specimen of Nidelric pugio gen. et sp. nov. was collected in 2007 from the Heilinpu Formation of the Mafang section near Haikou, west of Dianchi Lake, Yunnan Province, China23. Over 30 species with exceptional preservation have been documented from the Mafang Section, including some that are unique to this site, such as the chain-forming arthropod Synophalos xynos24 and the pterobranch Galeaplumosus abilus25. Mafang has additionally yielded some of the best-preserved Chengjiang material, including the lobopodian Luolishania longicruris and the arthropod Cindarella eucalla23. The Chengjiang Lagerstätte has yielded thousands of specimens over 30 years, but the rarity of N. pugio is confirmed by the absence of any further records of either articulated scleritomes or disarticulated spines from Mafang or other Chengjiang localities.
Systematic Palaeontology
Phylum uncertain, Order Chancelloriida Walcott, 1920?
Genus Nidelric gen. nov.
Type species
Nidelric pugio gen. et sp. nov., Cambrian Series 2 Heilinpu Formation, Mafang section, Haikou, Yunnan Province, China.
Etymology
From Latin Nidus (‘bird's nest’; fancied resemblance to) and adelric, derived from the Old English personal name ‘Aedelic’ (‘adel’, noble and ‘ric’, a ruler), a source for the name Aldridge. In memory of friend and colleague Professor Richard Aldridge, palaeontologist and keen ornithologist. Gender Masculine.
Diagnosis
Ovoid body that bears external, elongate, outward projecting spines: spines have a triangular profile, are gently curved with a non-flaring base and bear triangular scales that are absent from the spine base.
Remarks
Chancelloriida Walcott, 1920 are bag-shaped sessile metazoans with approximate radial symmetry and bear external sclerites with multiple hollow rays7. Exceptionally preserved scleritomes are known for the chancelloriid genera Chancelloria Walcott, Allonnia Doré and Reid and Archiasterella Sdzuy, each genus being distinguished by the arrangement of the spines within the complex, multi-element spine-rosettes. Although the external spines of Nidelric are single elements that are not fused into rosettes, the overall anatomy of its body suggests a radial symmetry and a possible biological affinity with chancelloriids.
The single-element spines of Nidelric are also very similar to the blade-like spines of the unnamed, small (ca 2 cm long) metazoan referred to as a chancelloriid from the slightly younger Cambrian Guanshan biota26, signaling that metazoans with this morphology may have been more widespread in early Cambrian Chinese faunas. Because there is limited morphological information from the spines to allow a precise comparison with characters of other chancelloriid and coeloscleritophoran spines, the assignment of Nidelric to the Chancelloriida remains tentative (see Discussion).
Nidelric pugio sp. nov.
Etymology
Pugio, a dagger; alluding to the shape of the spines.
Material and measurement
Holotype specimen YKLP11081a, b (part and counterpart), housed in the Yunnan Key Laboratory for Palaeobiology, Yunnan University, Kunming, China. The specimen is 9.45 cm long and 5.3 cm at its widest point.
Locality and stratigraphy
Cambrian Series 2 Heilinpu Formation, Mafang section, Haikou, Yunnan Province, China.
Diagnosis
Species of Nidelric bearing single-element spines that have a triangular profile and increase in size from the narrow to the broader end of the body: spines point outwards from the body, the long-axes typically oriented either normal to the margin of the body, or being deflected outwards in the direction of the narrow end of the body.
Description
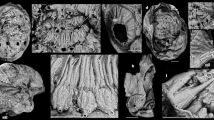
The body has a maximum width of 5.3 cm and tapers to just over 1 cm at its narrowest point (Fig. 1a). It is ovoid and is preserved as a mosaic of red-grey patches interspersed with areas of lighter-coloured sediment that may represent degraded epithelial integument and internal soft tissues. In one place on the body, towards the broader end, there are a series of small, oval, probably interlocking structures, about 150 μm in maximum diameter, that probably represent the integument (Fig. 1e, g). No traces of internal organs are discerned. Single element spines are present at low-density over the whole surface of the animal, with the greatest concentration along the margins of the body and at the broader end of the animal (Figs 1a–d, 2, 3a, b). The spines are elongate-triangular in profile and gradually tapering; they have variable preservation, mostly highlighted by iron oxide. These spines are typically 2.0 to 4.6 mm long, about 2.0 mm broad at their base, sharply pointed distally (Fig. 1b–d, f) and are preserved overlapping each other at the body margins (Fig. 1b–d), suggesting that they might have been concentrated into rows. At the broader end of the body, small and larger spines occur adjacent to each other, but generally the spines become smaller towards the narrower end of the body; many of the spines also point outwards in the direction of the narrow end of the body (Fig. 1a). The bases of some spines appear to be associated with a circular connection in the integument of the animal (Figs 1d, 2), but there is no evidence that the spines were embedded beneath the surface of the integument and the exposed nature of the spines is clearly evident at the body margins (Fig. 1a–c). The margins and distal portions of the spines are covered with a sculpture of small triangular scales on at least one (upper or lower) surface (e.g. Fig. 1b–d, f), there being about 10–11 such scales across the maximum width of the spines. The medial proximal area of each spine is flat, relatively smooth and apparently lacks scales.
Photographic images of Nidelric pugio.
Images a-c and e and f are YKLP11081a; image d and g are YKLP11081b. The images of ‘a’ and ‘e’ are taken using polarized light. (a), whole specimen, orientated with the narrow end at the bottom of the picture. (b), (c), are details of spine morphology preserved on the ‘left’ side of YKLP11081a (in the top half of image ‘a’, indicated by the arrows). (d), is the ‘counterpart’ of ‘c’ and shows a circular structure at the proximal end of the second spine from the left (arrowed). (f), is a close-up of the spine on the left side of image ‘c’, showing scaly sculpture on the surface of the spine. (e), (g), details of oval-shaped structures, representing possible integument at the broad end of the body: four of these oval structures form a fan shape at the mid-length of image ‘g’ (arrowed). Scale bars are: a, e, 1 cm; b, c, 1 mm; d, 2 mm; f, 740 μm; g, 400 μm.
Camera lucida drawing of Nidelric pugio, YKLP11081b.
The image is a representation of Figure 1d and shows circular structures at the base of some of the spines. Scale bar is 2 mm.
High Resolution X-ray Computed Tomography 3D reconstructions of Nidelric pugio (YKLP11081a).
(a), shows the surface of the specimen towards the broader end of the fossil (left) with areas containing Fe oxide-bearing material highlighted in green, with the highest density material (highest volume of Fe oxide) coloured yellow-green (centre) and a view with the less dense material stripped away to show just the highest density material that makes up the tips of the spines. Note the concentration of spines at this end of the fossil. (b), is a transverse view of the rock slab, with the sediment material stripped away. The grey areas represent parts of the material that contain some Fe-oxide and were identified as being higher density than the surrounding sediment. The highest density material is coloured red, which is where the amount of Fe-oxide is highest and corresponds with the tips of the spines. The view is looking beneath the surface and clearly demonstrates spines pointing down into the rock, interpreted as spines on the ‘lower’ surface of YKLP11081a as preserved (see Fig. 1a–c, for spines that are clearly on the ‘upper’ surface of YKLP11081a as preserved). Scale bars are indicated on the figure.
Remarks
Spines are the most obvious feature of the exposed surface of the specimen and detailed photography coupled with High Resolution X-ray Computed Tomography scanning (HRXCT) has resolved spines projecting outwards from the body both above and below the rock surface (e.g. Fig. 3b), indicating that spines were present over the whole surface of the animal and not just around the outer edge. Although the greatest concentration of spines, as preserved, is along the margins of the body, it is unclear whether this reflects distinct zonation in the original scleritome or post-mortem compression. HRXCT scanning also indicates a greater concentration of spines (and possibly other structures) preserved at the broader end of the animal (Fig. 3a). This might possibly be analogous to the tuft of spines interpreted to surround an apical orifice that is a feature of chancelloriids27 and it would be consistent with an overall sac-like body broadening upwards from a basal attachment: as spines tend to be smaller nearer the narrow end in N. pugio, this may also suggest the attached end of the body20. Nevertheless, this orientation for N. pugio is counter-intuitive to the general orientation of the spines at the margin of the body, which point outwards dominantly in the direction of the narrow end of the ovoid body (Fig 1a). Therefore, the orientation of N. pugio remains problematic.
Discussion
Preservation and taphonomy
The rock has split erratically through a longitudinal plane of the fossil and ‘part’ and ‘counterpart’ expose only an irregular surface of the anatomy. The body is preserved as iron oxides with a variety of habits including relatively poorly ordered framboids, octahedral forms and micro-crystals but not cubes. In the Chengjiang biota the chancellorid Allonniaphrixothrix is preserved compressed, but retains some relief5 and is commonly highlighted in the rock by a reddish colour imparted by iron oxide. A similar style of preservation appears to be afforded by a specimen interpreted as a chancelloriid in the Guanshan biota26.
Spines at the periphery of the body are preserved as thin, mainly oxidized carbon films coated with Fe oxides after pyrite28,29. Energy dispersive X-ray analyses (EDX) of the Fe oxides shows peaks in Mg, Fe, Al, Si, P and K. The presence of Al, Mg, Si and K probably indicates that the beam penetrated some Fe oxides into clay minerals in the underlying matrix. There is a consistent, but small peak in phosphorus associated with the Fe oxide minerals but not associated with other areas of the fossil or any mineral in the matrix. The lack of calcium indicates that the phosphorus is not occurring in apatite. The phosphorus may be chelated on to the Fe oxides and this must have occurred after oxidation of the pyrites (cf.30). The oxidation of early-formed iron pyrite would produce a low pH environment and small amounts of H2SO4 on and perhaps around the carcass. Thus, original biominerals have been dissolved28 (but see31, who report un-weathered sponge spicules showing a robust carbon film and no pyrite) and it is not possible to determine the original composition of the spines.
On many of the spines the proximal part comprises Fe oxide after pyrite, crystals of which are anhedral, together with ordered framboids. The distal tip of many spines comprises a high concentration of cubic crystals (Fig. 4c). It is possible that in life the tips of the spines comprised more recalcitrant material relative to the proximal parts of the spines and the rest of the animal28. Moreover, in at least one spine, the external surface of the spine is missing, revealing that beneath this surface the ‘core’ of the spine comprises octahedral iron minerals (Fig. 4d) and indicating that in life the spines had some aspect of three-dimensionality. We have no evidence to suggest that the spines were multi-structured in the manner of the cone-in-cone preservation exhibited by some lobopodian spines10, or possessed an internal structure of longitudinal lineations. Variable preservation is characteristic of the multi-element sclerites of A.phrixothrix5, with sclerites unfilled with sediment being compressed and flat. Although most of the spines across the body in N. pugio are generally flattened, a few suggest that the original shape was more three-dimensional (see also Fig. 4a, d). Variability in spine size, noted in N. pugio, is recorded within individual scleritomes of chancelloriids, including Chancelloria6.
Scanning Electron Microscope images of Nidelric pugio (YKLP11081a).
(a), secondary electron image of a spine at the periphery of the body showing surface ornament: the spine has some thickness and can be seen extending to the lamina below (bottom right of image). (b), backscatter (BSE) image of ‘a’; the bright mineral is Fe oxide. (c), BSE image of the tip of a spine showing many euhedral cubic Fe minerals. (d), BSE image showing that the spine has some aspect of three-dimensionality. The main surface of the spine comprises scattered microcrystals (texture on left of image) and small framboids: where some of the surface has been removed a ‘window’ exposes large poorly formed framboids comprising large octahedral microcrystals.
That no internal organs are evident in N. pugio may be due to their rapid decay, so that these tissues were lost prior to the commencement of preservation processes that only captured its body and spines. However, there are records across a wide range of taxa from the Chengjiang Lagerstätte of excellent preservation of internal organs (most recently, for example32,33). Another possibility that might account for the apparent lack of internal organs is if the integument and spines of N. pugio were sufficiently robust so as to obscure any internal organs beneath its external surface, but this is unlikely, given that our HRXCT scans record no such structures in the cross sections through the internal anatomy (Fig. 3b). We conclude, therefore, that N. pugio was a sac-like metazoan without internal organs.
Affinities
In overall body form N. pugio resembles a sponge. However, a poriferan affinity is unsupported by the presence of triangular profile spines that are inserted into the surface integument, rather than solid, slender spicules. Individually, the spines somewhat resemble the dorsal spines of hallucigeniid lobopodians and their “small shelly” and “small carbonaceous” counterparts, including Mongolitubulus, Rushtonites and Rhombocorniculum10. In detail, though, the spines of N. pugio are more coarsely sculptured and they are clearly not attached to a lobopodian. Instead, the combination of an ovoid, radially symmetrical body shape and ornamented sclerites suggests a chancelloriid affinity. Furthermore, N. pugio is comparable in size to large chancelloriids such as Allonnia6. Even so, N. pugio differs from described chancelloriids in several important respects. Articulated specimens of confirmed chancelloriids exhibit characteristic multi-element rosettes of spines that insert into the epithelial integument of the body5, in contrast to the single-rayed, unfused spines in N. pugio. However, Moore et al.7 discussed the possibility that forms with a single rayed structure could represent the ancestral type for chancelloriids. They cited similarities with the single-rayed spines of Cambrothyra34, which are recovered as isolated “small shelly fossils” from the pre-trilobite interval of the Cambrian. The spines of N. pugio are morphologically different from the rounded conical spines of Cambrothyra, but both types of single-element spine may support the idea of an ancestral unfused state for the earliest chancelloriids7. The spines of Nidelric also differ from the single-rayed spines of the chancelloriid Eremactis Bengtson and Conway Morris which have a circular cross-section and a smooth or longitudinally striated surface: spines of Eremactis occasionally fuse to form a chancelloriid-like composite35, a pattern that has not been observed in the spines of Nidelric.
Based on a detailed analysis of spine morphology chancelloriids have been considered to be coeloscleritophorans18,27, which includes halkieriids, siphogonuchitids and sachitids. The range of morphologies of coeloscleritophoran spines is varied, but key shared structures include: an organic outer layer; an inner layer of longitudinally arranged aragonite fibres; aragonite layers in bundles that form projections on the upper surface; and a lower surface without projections18. Not all of these characters are likely to be preserved in N. pugio. Even so, the spines bear comparable sculpture (see Fig. 1b–d, f) and overall are compatible with a coeloscleritophoran-type construction. Among described coelosclerites, the form-taxon Hippopharangites includes broad-based triangular forms with a spiny ornamentation35. However, the presence of right- and left-handed sclerite morphologies in Hippopharangites suggests that it came from a bilaterally symmetrical organism unlike N. pugio. The absence of a precise match for N. pugio spines among “small shelly fossils” may reflect their original rarity in assemblages, or a poorly mineralized (or unmineralized) construction in life.
Despite similarities in overall body form and basic spine construction between N. pugio and chancelloriids, an important distinction may exist in the polarity of the body axes. In most chancelloriid specimens, the attached (proximal) end is narrower and the spines tend to be directed towards the broader apical end, with the largest spines towards the apex5,6. In contrast, in N. pugio the spines are directed towards the narrower (more pointed) end, where they are smaller (Fig. 1a). This relationship between spine orientation and body outline lends an overall resemblance – at least in flattened profile – between N. pugio and the problematic Burgess Shale animal Orthrozanclus reburrus, a motile bilaterian with similarities to Wiwaxia and Halkieria36. Therefore, the proposed homology between chancelloriid and halkieriid sclerites18 raises the question of whether N. pugio might form a link between motile and sessile coeloscleritophorans. However, the lack of an anterior shell, long spines or obvious bilateral symmetry in N. pugio means that any resemblance to O. reburrus may be entirely superficial and lends no additional support for hypotheses of a coelosclerite homology, or a transition between motile and sessile lifestyles.
Given the difficulties in reconciling the morphologies of N. pugio and confirmed chancelloriids and coeloscleritophorans, an alternative hypothesis might interpret Nidelric as representative of a group of spine-bearing sac-like metazoans that are unrelated to chancelloriids (and which might also include the unnamed metazoan identified as a chancelloriid from the Guanshan biota26). In this scenario, the overall similarity of Nidelric to chancelloriids would simply reflect homoplasy.
Palaeoecology
The ovoid shape and absence of internal organs suggests that N. pugio possessed a sac-like morphology in life and was likely sessile. Sessile benthos represents a diverse component of the Chengjiang biota23 that includes sponges, brachiopods and chancelloriids and N. pugio was likely a component of this benthos, possibly being anchored to the seabed – though no anchoring structures are preserved. The mode of feeding in N. pugio is unknown and it displays no evidence of structures that could be used for predation (or locomotion). It may, like sponges and putatively for chancelloriids, have been a filter feeder, though again there are no preserved structures in N. pugio to confirm or refute this lifestyle. The outwards pointing spines of N. pugio were likely designed to deter predation in a sessile organism without the functional means of escape and this is consistent with the surface (and not internal) arrangement of the spines. N. pugio represents an exceptionally rare component of the Chengjiang biota, with only a single specimen recovered from 30 years of collecting. The rarity of this species is also attested by the absence of its disarticulated spines from the Heilinpu Formation.
Conclusions
N. pugio is a radial sac-like metazoan that possesses morphology similar to chancelloriids: however, it possesses single-element unfused spines that are atypical of this group. N. pugio might represent the ancestral state for chancelloriids, from which forms with multi-element fused rosettes of spines emerged. Alternatively, N. pugio may represent a group of spine-bearing sac-like metazoans that are discrete from chancelloriids, the similarity of design simply reflecting convergence. Until conclusive morphological evidence is available from the spines of Nidelric, either of these two interpretations remains possible. Irrespective, N. pugio provides a new model for reconstructing isolated sclerites and reveals a previously unanticipated disparity among Cambrian spine-bearing animals.
Methods
‘Part’ and ‘counterpart’ (YKLP11081a, b) of N. pugio were prepared at the Key Laboratory for Palaeobiology, Yunnan University using fine needles. The 3D morphology of N. pugio was investigated using High Resolution X-ray Computed Tomography (HRXCT) at Leicester University using an XT225 Metris (Nikon) scanner. Reconstructions were rendered using CT Pro software and VG studio Max Version 2.0 was used to visualize the 3D images. As preserved, N. pugio is only a few millimetres thick. However, this and the preservation of some parts by relatively dense accumulations of pyrite, oxidized to Fe oxide, make X-ray analysis useful for assessing its anatomy. The fossil was analyzed by scanning the specimen in an upright position around a rotational axis that ran the length of the fossil, using voltage of 110 kV, a current of 265 μA and a 0.5 mm Cu filter. Individual scans were performed in each case over a period of ~8 hours.
The fossil was imaged and analysed using a Hitachi S-3600N Environmental Scanning Electron Microscope (Department of Geology, University of Leicester). Chemical analysis was undertaken using an Oxford INCA 350 EDX system, providing elemental mapping and point-and-ID spectrum. The system operated with a standard voltage of 15 kV; however carbon was mapped using 5 kV. Macrophotography was performed on N. pugio at Oxford University, using a Nikon Multiphot stand and associated bellows, with 12 cm and 6.5 cm Micro-Nikkor lenses, together with a Canon 5D digital camera. Incident lighting was used, either from a low angle fibre-optic swan neck source, or a directly illuminated fibre-optic ring light attached with a polarizer for contrast enhancement.
References
Brasier, M. D. The succession of small shelly fossils (especially conoidal microfossils) from English Precambrian-Cambrian boundary beds. Geol. Mag. 123, 237–256 (1986).
Steiner, M., Li, G. X., Qian, Y. & Zhu, M. Y. Lower Cambrian shelly fossils of northern Sichuan and southern Shaanxi (China) and their biostratigraphic importance. Geobios 37, 259–275 (2004).
Harvey, T. H. P., Ortega−Hernández, J., Lin, J. −. P., Zhao, Y. & Butterfield, N. J. Burgess Shale−type microfossils from the middle Cambrian Kaili Formation, Guizhou Province, China. Acta Palaeontol. Pol. 57, 423–436 (2012).
Harvey, T. H. P., Vélez, M. I. & Butterfield, N. J. Exceptionally preserved crustaceans from western Canada reveal a cryptic Cambrian radiation. Proc. Natl Acad. Sci. USA 109, 1589–1594 (2012).
Bengtson, S. & Hou, X. The integument of Cambrian chancelloriids. Acta Palaeontol. Pol. 46, 1–22 (2001).
Janussen, D., Steiner, M. & Zhu, M.-Y. New well-preserved scleritomes of Chancelloridae from the Early Cambrian Yuanshan Formation (Chengjiang, China) and the Middle Cambrian Wheeler Shale (Utah, USA) and paleobiological implications. J. Paleontol. 76, 596–606 (2002).
Moore, J. L., Li, G. & Porter, S. M. Chancellorid sclerites from the lower Cambrian (Meishucunian) of eastern Yunnan and the early history of the group. Palaeontology 57, 833–878 (2014).
Ivantsov, A., Yu & Wrona, R. Articulated palaeoscolecid sclerite arrays from the Lower Cambrian of eastern Siberia. Acta Geol. Pol. 54, 1–22 (2004).
Skovsted, C. B., Brock, G. A. & Topper, T. A. Sclerite fusion in the problematic early Cambrian spine-like fossil Stoibostrombus from South Australia. Bull. Geosci. 86, 651–658 (2011).
Caron, J.-B., Smith, M. R. & Harvey, T. H. P. Beyond the Burgess Shale: Cambrian microfossils track the rise and fall of hallucigeniid lobopodians. Proc. R. Soc. B 280, 20131613 (2013).
Caron, J.-B., Scheltema, A., Schander, C. & Rudkin, D. A soft-bodied mollusc with radula from the Middle Cambrian Burgess Shale. Nature 442, 159–163 (2006).
Vinther, J. The canal system in sclerites of lower Cambrian Sinosachites (Halkieriidae: Sachitida): significance for the molluscan affinities of sachitids. Palaeontology 52, 689–712 (2009).
Skovsted, C. B. et al. The scleritome of Paterimitra: an early Cambrian stem group brachiopod from South Australia. Proc. R. Soc. B 276, 1651–1656 (2009).
Kouchinsky, A., Bengtson, S. & Murdock, D. J. E. A new tannuolinid problematic from the lower Cambrian of the Sukharikha River in northern Siberia. Acta Palaeontol. Pol. 55, 321–331 (2010).
Skovsted, C. B. A carapace of the bradoriid arthropod Mongolitubulus from the Early Cambrian of Greenland. GFF 127, 217–220 (2005).
Conway Morris, S. The Middle Cambrian metazoan Wiwaxia corrugata (Matthew) from the Burgess Shale and Ogygopsis Shale, British Columbia, Canada. Philos. Trans. R. Soc. Lond. B 307, 507–582 (1985).
Conway Morris, S. & Peel, J. Articulated halkieriids from the Lower Cambrian of North Greenland and their role in early protostome evolution. Philos. Trans. R. Soc. Lond. B 347, 305–358 (1995).
Porter, S. M. Skeletal microstructure indicates Chancelloriids and Halkieriids are closely related. Palaeontology 51, 865–879 (2008).
Sperling, E. A., Pisani, D. & Peterson, K. J. in The rise and fall of the Ediacaran biota (Vickers-Rich, P. & Komarower, P.) 355–368 (Geological Society of London Special Publication 286. 2007).
Mehl, D. Organization and microstructure of the chancelloriid skeleton: implications for the biomineralization of the Chancelloriidae. Bull. lnst. océanogr. (Monaco), Numéro spécial 14, 377–385 (1996).
Goryanskij, V. Y. in Problemy paleontologii i biostratigrafiki nizhnego kembriya Sibiri i Dal'nego vostoka (Zhuravleva I. T.) 39–44 (Trudy Instituta Geologii i Geofiziki SO AN SSSR, 49, 1973).
Walcott, C. D. Cambrian geology and paleontology. IV. Middle Cambrian Spongiae. Smithson. Misc. Collect. 67, 261–364 (1920).
Hou, X. G. et al. The Cambrian fossils of Chengjang, China, the flowering of early animal life. (Blackwell Publishing., 2004).
Hou, X. G., Siveter, D. J., Aldridge, R. A. & Siveter, D. J. Collective behavior in an Early Cambrian Arthropod. Science 322, 224–224 (2008).
Hou, X. G. et al. An early Cambrian hemichordate zooid. Curr. Biol. 21, 612–616 (2011).
Hu, S. et al. The Guanshan Biota. (Yunnan Publishing Group Co. Ltd. Yunnan Science and Technology Press., 2004).
Bengtson, S. in Evolving form and function: fossils and development (Briggs D. E. G.) 101–124 (Special Publication of the Peabody Museum of Natural History, Yale University, New Haven, CT, 2005).
Gabbott, S. E., Hou, X. G., Norry, M. J. & Siveter, D. J. Preservation of Early Cambrian animals of the Chengjiang biota. Geology. 32, 901–904 (2004).
Zhu, M. Y., Babcock, L. E. & Steiner, M. Fossilization modes in the Chengjiang Lagerstätte (Cambrian of China); testing the roles of organic preservation and diagenetic alteration in exceptional preservation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 220, 31–46 (2005).
Ingall, E. D., Bustin, R. M. & Van Cappellen, P. Influence of water column anoxia on the burial and preservation of carbon and phosphorous in marine shales. Geochim. Cosmochim. Acta 57, 303–316 (1993).
Forchielli, A., Steiner, M., Hu, S. X. & Keupp, H. Taphonomy of Cambrian (Stage 3/4) sponges from Yunnan (South China). Bull. Geosci. 87, 133–142 (2012).
Ma, X. Y., Hou, X. G., Edgecombe, G. D. & Strausfeld, N. J. Complex brain and optic lobes in an early Cambrian arthropod. Nature 490, 258–261 (2012).
Ma, X. Y., Cong, P. Y., Hou, X. G., Edgecombe, G. D. & Strausfeld, N. J. An exceptionally preserved arthropod cardiovascular system from the early Cambrian. Nat. Commun. 5, 3560 (2014).
Moore, J. L., Porter, S. M., Steiner, M. & Li, G. Cambrothyra ampulliformis, an unusual Coeloscleritophoran from the Lower Cambrian of Shaanxi Province, China. J. Paleontol. 84, 1040–1060 (2010).
Bengtson, S., Conway Morris, S., Cooper, B. J., Jell, P. A. & Runnegar, B. N. Early Cambrian fossils from South Australia. Mem. Assoc. Australas. Palaeontol. 9, 1–364 (1990).
Conway Morris, S. & Caron, J.-B. Halwaxiids and the early evolution of the Lophotrochozoa. Science 315, 1255–1258 (2007).
Acknowledgements
We are very grateful to the National Natural Science Foundation of China (Grant number U1302232) and the Royal Society (Grant number IE131457) for funding this work.
Author information
Authors and Affiliations
Contributions
H.X. arranged fieldwork and discovered the specimen. D.J.S. and D.J.S. prepared figures 1 and 2, D.H. prepared figure 3 and S.G. prepared figure 4. H.X., D.H. and S.G. undertook analyses. M.W. and H.X. wrote the main manuscript with input from D.J.S., D.J.S., S.G., D.H. and T.H.P.H. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Hou, X., Williams, M., Siveter, D. et al. A chancelloriid-like metazoan from the early Cambrian Chengjiang Lagerstätte, China. Sci Rep 4, 7340 (2014). https://doi.org/10.1038/srep07340
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep07340
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.