Abstract
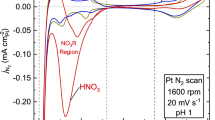
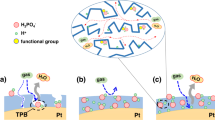
The specific adsorption of ionomer sulfonate groups on Pt-based catalysts in membrane electrode assemblies (MEAs) has severely restricted Pt catalytic activity, Pt utilization, proton conductivity and mass transport. Here we report a blocking strategy using cyclohexanol to mitigate the detrimental impacts of the Nafion ionomer. Cyclohexanol with a chair or boat conformation blocked the adsorption path of the ionomer onto the Pt surface via coordination with the ionomer, which released the Pt activity sites and dramatically improved the mass transport path. This MEA with cyclohexanol exhibits striking performance improvement in the kinetic and mass transport regions, along with strong stability. The proposed strategy provides a direction to tune the Pt/ionomer interface and improve the catalytic activity of Pt in MEA.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
References
Xie, M. et al. Pt-Co@Pt octahedral nanocrystals: enhancing their activity and durability toward oxygen reduction with an intermetallic core and an ultrathin shell. J. Am. Chem. Soc. 143, 8509–8518 (2021).
Hu, Y. et al. Coplanar Pt/C nanomeshes with ultrastable oxygen reduction performance in fuel cells. Angew. Chem. Int. Ed. 60, 6533–6538 (2021).
Li, M. et al. Lavender-like Ga-doped Pt3Co nanowires for highly stable and active electrocatalysis. ACS Catal. 10, 3018–3026 (2020).
Cheng, Q. et al. High-loaded sub-6-nm Pt1Co1 intermetallic compounds with highly efficient performance expression in PEMFCs. Energy Environ. Sci. 15, 278–286 (2022).
Huang, L. et al. Boosting oxygen reduction via integrated construction and synergistic catalysis of porous platinum alloy and defective graphitic carbon. Angew. Chem. Int. Ed. 60, 25530–25537 (2021).
Ding, H. et al. Epitaxial growth of ultrathin highly crystalline Pt-Ni nanostructure on a metal carbide template for efficient oxygen reduction reaction. Adv. Mater. 34, 2109188 (2022).
Chong, L. et al. Ultralow-loading platinum-cobalt fuel cell catalysts derived from imidazolate frameworks. Science 362, 1276–1281 (2018).
Kodama, K., Nagai, T., Kuwaki, A., Jinnouchi, R. & Morimoto, Y. Challenges in applying highly active Pt-based nanostructured catalysts for oxygen reduction reactions to fuel cell vehicles. Nat. Nanotechnol. 16, 140–147 (2021).
Sun, Y. et al. Advancements in cathode catalyst and cathode layer design for proton exchange membrane fuel cells. Nat. Commun. 12, 5984 (2021).
Xiao, F. et al. Atomically dispersed Pt and Fe sites and Pt-Fe nanoparticles for durable proton exchange membrane fuel cells. Nat. Catal. 5, 503–512 (2022).
Fan, J. et al. Bridging the gap between highly active oxygen reduction reaction catalysts and effective catalyst layers for proton exchange membrane fuel cells. Nat. Energy 6, 475–486 (2021).
Ahn, C. Y. et al. Differences in the electrochemical performance of Pt-based catalysts used for polymer electrolyte membrane fuel cells in liquid half- and full-cells. Chem. Rev. 121, 15075–15140 (2021).
Doo, G. et al. Tuning the ionomer distribution in the fuel cell catalyst layer with scaling the ionomer aggregate size in dispersion. ACS Appl. Mater. Interfaces 10, 17835–17841 (2018).
Lazaridis, T., Stühmeier, B. M., Gasteiger, H. A. & El-Sayed, H. A. Capabilities and limitations of rotating disk electrodes versus membrane electrode assemblies in the investigation of electrocatalysts. Nat. Catal. 5, 363–373 (2022).
Kongkanand, A. & Mathias, M. F. The priority and challenge of high-power performance of low platinum proton-exchange membrane fuel cells. J. Phys. Chem. Lett. 7, 1127–1137 (2016).
Modestino, M. A. et al. Self-assembly and transport limitations in confined Nafion films. Macromolecules 46, 867–873 (2013).
Page, K. A. et al. Confinement-driven increase in ionomer thin-film modulus. Nano Lett. 14, 2299–2304 (2014).
Tymoczko, J. et al. Oxygen reduction at a Cu-modified Pt(111) model electrocatalyst in contact with Nafion polymer. ACS Catal. 4, 3772–3778 (2014).
Kodama, K. et al. Effect of the side-chain structure of perfluoro-sulfonic acid ionomers on the oxygen reduction reaction on the surface of Pt. ACS Catal. 8, 694–700 (2017).
Ott, S. et al. Ionomer distribution control in porous carbon-supported catalyst layers for high-power and low Pt-loaded proton exchange membrane fuel cells. Nat. Mater. 19, 77–85 (2020).
Yarlagadda, V. et al. Boosting fuel cell performance with accessible carbon mesopores. ACS Energy Lett. 3, 618–621 (2018).
Jinnouchi, R. et al. The role of oxygen-permeable ionomer for polymer electrolyte fuel cells. Nat. Commun. 12, 4956 (2021).
Katzenberg, A. et al. Highly permeable perfluorinated sulfonic acid ionomers for improved electrochemical devices: insights into structure-property relationships. J. Am. Chem. Soc. 142, 3742–3752 (2020).
Doo, G. et al. Nano-scale control of the ionomer distribution by molecular masking of the Pt surface in PEMFCs. J. Mater. Chem. A 8, 13004–13013 (2020).
Liu, H., Ney, L., Zamel, N. & Li, X. Effect of catalyst ink and formation process on the multiscale structure of catalyst layers in PEM fuel cells. Appl. Sci. 12, 3776–3816 (2022).
Zhang, G. R., Munoz, M. & Etzold, B. J. M. Boosting performance of low temperature fuel cell catalysts by subtle ionic liquid modification. ACS Appl. Mater. Interfaces 7, 3562–3570 (2015).
Zhang, G. R., Munoz, M. & Etzold, B. J. M. Accelerating oxygen-reduction catalysts through preventing poisoning with non-reactive species by using hydrophobic ionic liquids. Angew. Chem. Int. Ed. 55, 2257–2261 (2016).
Wang, T. et al. Enhancing oxygen reduction electrocatalysis by tuning interfacial hydrogen bonds. Nat. Catal. 4, 753–762 (2021).
Wu, X., Chen, N., Klok, H. A., Lee, Y. M. & Hu, X. Branched poly(aryl piperidinium) membranes for anion-exchange membrane fuel cells. Angew. Chem. Int. Ed. 61, e202114892 (2022).
Liu, B. et al. Precise molecular-level modification of Nafion with bismuth oxide clusters for high-performance proton-exchange membranes. Angew. Chem. Int. Ed. 60, 6076–6085 (2021).
Kunimatsu, K., Bae, B., Miyatake, K., Uchida, H. & Watanabe, M. ATR-FTIR study of water in Nafion membrane combined with proton conductivity measurements during hydration/dehydration cycle. J. Phys. Chem. B 115, 4315–4321 (2011).
Kusoglu, A. & Weber, A. Z. New insights into perfluorinated sulfonic-acid ionomers. Chem. Rev. 117, 987–1104 (2017).
Kusoglu, A., Dursch, T. J. & Weber, A. Z. Nanostructure/swelling relationships of bulk and thin-film PFSA ionomers. Adv. Funct. Mater. 26, 4961–4975 (2016).
Kusoglu, A. et al. Impact of substrate and processing on confinement of Nafion thin films. Adv. Funct. Mater. 24, 4763–4774 (2014).
Mikhailenko, S., Guiver, M. & Kaliaguine, S. Measurements of PEM conductivity by impedance spectroscopy. Solid State Ion. 179, 619–624 (2008).
Wei, C. et al. Approaches for measuring the surface areas of metal oxide electrocatalysts for determining their intrinsic electrocatalytic activity. Chem. Soc. Rev. 48, 2518–2534 (2019).
Yamada, H., Kato, H. & Kodama, K. Cell performance and durability of Pt/C cathode catalyst covered by dopamine derived carbon thin layer for polymer electrolyte fuel cells. J. Electrochem. Soc. 167, 084508 (2020).
Zhou, F. et al. Solving Nafion poisoning of ORR catalysts with an accessible layer: designing a nanostructured core-shell Pt/C catalyst via a one-step self-assembly for PEMFC. Int. J. Energy Res. 44, 10155–10167 (2020).
Li, Y., Intikhab, S., Malkani, A., Xu, B. & Snyder, J. Ionic liquid additives for the mitigation of Nafion specific adsorption on platinum. ACS Catal. 10, 7691–7698 (2020).
Garrick, T. R., Moylan, T. E., Yarlagadd, V. & Kongkanand, A. Characterizing electrolyte and platinum interface in PEM fuel cells using CO displacement. J. Electrochem. Soc. 164, F60–F64 (2017).
Acknowledgements
This study was financially supported by the National Key R&D Program of China (2020YFB1506002, S.C.), the National Natural Science Foundation of China (grant nos. 52021004 and 91834301, Z.W.; grant nos. 22178034 and 21978028, S.C.; grant no. 22108020, M.W.) and the Chongqing Talent Program (cstc2022ycjh-bgzxm0096, S.C.).
Author information
Authors and Affiliations
Contributions
S.C., Z.W. and L.G. conceived of the project. S.C. directed the experimental work. S.C. and F.C. analysed the experimental data. F.C. carried out the sample synthesis, characterization, electrochemical measurements and fuel-cell tests. A.W. and M.W. helped with editing of the paper. S.C., L.G. and F.C. prepared the manuscript with feedback from the other authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Hany El-Sayed and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–16, Tables 1 and 2 and references.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, F., Chen, S., Wang, A. et al. Blocking the sulfonate group in Nafion to unlock platinum’s activity in membrane electrode assemblies. Nat Catal 6, 392–401 (2023). https://doi.org/10.1038/s41929-023-00949-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-00949-w
This article is cited by
-
High-temperature annealing improves Pt utilization of proton exchange membrane fuel cell cathode catalysts
Science China Materials (2024)
-
The difference of the ionomer–catalyst interfaces for poly(aryl piperidinium) hydroxide exchange membrane fuel cells and proton exchange membrane fuel cells
Nano Research (2024)
-
Cyclohexanol optimizes Pt/ionomer interface in fuel cell cathodes
Science China Chemistry (2023)
-
Tuning metal-support interaction of Pt-based electrocatalysts for hydrogen energy conversion
Science China Chemistry (2023)