Abstract
Serum anti-mitochondrial antibody type 2 (AMA-M2) is considered as a pivotal biomarker for the diagnosis of primary biliary cholangitis (PBC). However, serological tests have many limitations, including inconvenience, invasiveness, and infection risks. Thus, a less invasive approach to detect AMA-M2 titer is desirable. We examined salivary AMA-M2 of potential PBC patients and found that AMA-M2 could be detected only in saliva of serum AMA-M2-positive PBC patients, but not in saliva of serum AMA-M2-negative PBC patients, oral lichen planus patients (OLP) patients, or healthy controls. Furthermore, the concentration of salivary AMA-M2 was positively correlated with the amount of serum AMA-M2 in patients. The salivary inflammatory cytokines were increased in the PBC, consistent with the results of serum test. These findings indicated that saliva might be a less invasive and cost-effective medium to accurately test for AMA-M2 levels and this is a promising development for the diagnosis and monitoring of PBC.
Similar content being viewed by others
Introduction
Saliva is an exocrine secretion from the salivary glands composed of 99% water. It also contains gingival crevicular fluid, serum, and other cellular components, such as proteins, enzymes, antibodies, and cytokines1, 2. It was shown that proteins and other substances enter saliva from the blood through passive diffusion or active transport, indicating that many substances found in the blood might also be present in saliva3. For example, salivary glucose level was reported to be associated with serum glucose level in healthy individuals4. Likewise, a positive correlation of endothelin concentrations was found between saliva and plasma in patients with congestive heart failure5. Taken together, saliva could be an effective and much less invasive medium for effectively diagnosing human diseases and monitoring a patient’s health.
Primary biliary cholangitis (PBC) is a chronic autoimmune liver disease associated with progressive destruction of small intrahepatic bile ducts6. Similar to other autoimmune diseases, like systemic lupus erythematosus7 and rheumatoid arthritis8, females are diagnosed more often than males with a frequency of about ten to one6. The diagnosis of PBC is typically based on abnormal serum biochemical parameters, such as the presence of anti-mitochondrial antibody (AMA), increased alkaline phosphatase (ALP), and a positive reaction for gamma-glutamyl transferase (GGT)9. Serum AMA, especially the AMA-M2 subtype, is regarded as one of the most specific and acceptable diagnostic indicators for PBC10, 11. Since the presence of AMA was first recognized by Walker et al. in serum samples of PBC patients in 196512, the high titer of AMA has become a serological diagnostic hallmark of PBC with approximately 95% sensitivity6, 10, 13. However, the clinical features of PBC are non-specific, which make the early detection and diagnosis is still rather difficult. Anyone with findings of chronic cholestasis or raised concentrations of ALP should be considered a suspected PBC case. When diagnosed early and properly treated, PBC patients generally respond well to medical therapy, on the contrary, others with delayed diagnosis and treatment usually have to undergo liver transplantation14. Blood test may be tough to perform especially in suspected patients living in remote villages with poor medical condition or rather away from the hospital. Recently, immense interest has been converted to develop a more convenient, less-invasive diagnostic approach as an alternative to blood test. Salivary test is, therefore, a preferable option once its clinical value has been identified. Currently, the clinical value of this approach has been increasingly highlighted in the literature, suggesting that saliva could be an improved diagnostic medium15,16,17,18.
Herein, we investigated whether AMA-M2 could be detected in the saliva of PBC patients, and whether salivary AMA-M2 could provide a novel and practical biomarker for precise diagnosis of PBC.
Results
AMA-M2 was detectable in saliva of PBC patients
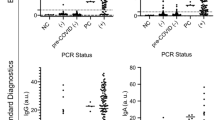
In this study, we measured the levels of salivary and serum AMA-M2 in all participants. Positive results (i.e. that the value of serum AMA-M2 exceed 40 RU/ml)9 for serum AMA-M2 were detected in 33 out of 49 PBC patients (469.20 ± 71.31 RU/ml), while all 60 HC subjects showed negative (3.11 ± 0.43 RU/ml) (Fig. 1a). Importantly, salivary AMA-M2 could be detected only in the serum AMA-M2-positive patients, whereas significant levels could not be detected in any serum AMA-M2-negative patients or HC subjects. Additionally, none of the patients diagnosed with the immune-related mouth disease OLP displayed detectable levels of salivary AMA-M2 (Fig. 1b, Supplementary Figure 1a).
AMA-M2 was detectable in saliva of PBC patients. Levels of anti-mitochondrial antibody subtype M2 (AMA-M2) were measured in collected serum (a) and saliva (b) of healthy controls (HCs, n = 60), primary biliary cholangitis (PBC) (−) patients (serum AMA-M2-negative PBC patients, n = 16), and PBC (+) patients (serum AMA-M2-positive PBC patients, n = 33). Data presented are the means ± SEM. *P < 0.05; **P < 0.01; ***P < 0.001; ns, not significant.
The level of salivary AMA-M2 was positively associated with the level of serum AMA-M2 in PBC patients
We next tested whether there was a correlation between the detectable levels of salivary versus serum AMA-M2. Indeed, salivary AMA-M2 was shown to be significantly positively associated (r = 0.63, P < 0.001) with levels of serum AMA-M2 in PBC patients (Fig. 2a). Furthermore, in order to predict a threshold value of salivary AMA-M2 that could be used for the diagnosis of PBC, ROC was performed. The area under the curve (AUC) was 0.88 (95% CL = 0.65–0.93), and the best diagnostic threshold value was 0.61 RU/ml with a sensitivity of 81.82% and specificity of 80.00% (Fig. 2b).
Correlation analysis performed between salivary AMA-M2 and serum AMA-M2 and ROC analysis of salivary AMA-M2 in PBC patients. (a) Correlation between salivary AMA-M2 and serum AMA-M2 levels in patients with PBC (serum AMA-M2-positive PBC patients, n = 33). (b) Receiver operating characteristics (ROC) curve of salivary AMA-M2 for determining PBC.
Furthermore, we collected 10 serum and saliva samples from return visit PBC patients to validate our results. Serum AMA-M2 tests were positive in all the samples, while the levels of salivary AMA-M2 were higher than the cut-off point in 90% of the return visit PBC patients. (Supplementary Figure 2a,b)
Moreover, we analyzed changes in serum and salivary AMA-M2 levels in two collections from the same PBC patients. The variation of salivary AMA-M2 in most of the PBC patients was consistent with that of serum AMA-M2. (Supplementary Figure 3).
Salivary AMA-M2 does not correlate with other serum hepatic function indices related to cholestasis
Elevated ALP is associated with cholestasis and biliary tract injury. Cholestasis and biliary tract injury present in PBC patients, and these conditions worsen as the disease progresses9, 19. Furthermore, elevated GGT is a valuable indicator of bile duct obstruction20,21,22, which often manifests in PBC patients. We tested whether ALP or GGT levels correlated with serum or salivary AMA-M2. However, neither serum ALP levels (Fig. 3a,b) nor serum GGT levels (Fig. 3c,d) were found to correlate with serum or salivary AMA-M2 levels in PBC patients. Taken together, salivary AMA-M2 could be a promising proxy for accurately diagnosing PBC.
Serum and salivary AMA-M2 levels did not correlate with other hepatic function indicators. (a) Correlation between serum AMA-M2 and serum ALP levels in patients with PBC (serum AMA-M2-positive PBC patients, n = 33). (b) Correlation between salivary AMA-M2 and serum ALP levels in patients with PBC (serum AMA-M2-positive PBC patients, n = 33). (c) Correlation between serum AMA-M2 and serum GGT levels in patients with PBC (serum AMA-M2-positive PBC patients, n = 33). (d) Correlation between salivary AMA-M2 and serum GGT levels in patients with PBC (serum AMA-M2-positive PBC patients, n = 33).
The elevated levels of inflammatory cytokines in saliva reflect a disorder of the oral immune system
Next, we tested the relationship between levels of inflammatory cytokines in the serum versus saliva in PBC patients. Since IL-17A has been reported to play a key role in the inflammatory reaction of PBC patients23,24,25, we examined the levels of inflammatory cytokines associated with IL-17 signaling and immune activation in the serum of all participants. We found that the serum levels of IL-6, IL-17A, IL-23, IFN-γ, and TNF-α in PBC patients were significantly higher compared to HC subjects. Furthermore, the serum immunosuppressive cytokine IL-10 was significantly downregulated in PBC patients (Fig. 4a).
Inflammatory cytokine levels in serum and saliva of PBC patients. The levels of IL-6, IL-17A, IFN-γ, TNF-α, IL-23, IL-4, and IL-10 in serum (a) and saliva (b) of HCs (healthy controls, n = 60) and PBC patients (serum AMA-M2-positive PBC patients, n = 33) were determined using Luminex Bead Assay. Data presented are the means ± SEM. (c) Correlation between salivary IL-6 and serum IL-6 levels in patients with PBC (serum AMA-M2-positive PBC patients, n = 33). (d) Correlation between salivary IFN-γ and serum IFN-γ levels in patients with PBC (serum AMA-M2-positive PBC patients, n = 33). *P < 0.05; **P < 0.01; ***P < 0.001; ns, not significant.
In saliva samples, IL-6 and IFN-γ levels were significantly increased in the PBC group. Meanwhile IL-10, IL-17A, and TNF-α levels were elevated in PBC patients, but these results were not statistically significant (Fig. 4b). IL-4 and IL-23 could not be detected in saliva samples. Furthermore, we found that levels of salivary IL-6, IFN-γ, IL-10, IL-17A, and TNF-α in OLP patients were significantly upregulated, likely because of the oral immune disorder (Supplementary Figure 1b).
To investigate whether saliva inflammatory factors could reflect immune status, we explored the relationship between IL-6 and IFN-γ in saliva and serum. Intriguingly, we discovered that serum IL-6 and IFN-γ levels in PBC patients were positively associated with those in saliva (Fig. 4c,d), but no obvious association was observed for IL-10, IL-17A, or TNF-α in saliva versus serum.
Discussion
Early and precise diagnosis of PBC is still a great challenge. Therefore, many studies have explored ways to optimize diagnostic methods. Tan and his colleagues proposed that serum microRNA had considerable clinical value in PBC diagnosis with high diagnostic accuracy. However, its high cost limits its clinical application. Salivary AMA-M2 tests may be a more cost-effective and convenient diagnostic method for PBC26. Saliva is a particularly tantalizing diagnostic medium, it is easy to collect, store and transport, and these steps do not require highly trained personnel. Patients can even collect samples and complete detection themselves at home, which increases the chances of identifying diseases at early stages and may cost less than blood test by saving the charge of blood collection and processing. Furthermore, saliva collection does not carry with it the risks of infection like serum collection does.
Saliva is already of great clinical value for monitoring a patient’s general health and disease screening for medical problems27,28,29,30,31. Numerous studies have demonstrated that saliva could be used to diagnose human diseases. For example, biomarkers found in saliva might be used to screen for breast cancer with high specificity and sensitivity32. Cabral et al. proposed that salivary cortisol concentration was an excellent indicator of stress of newborns33. Likewise, Mascarenhas et al. suggested that salivary glucose levels could be a potential biomarker for type 2 diabetes mellitus34. It has been speculated that an early factor in the development of PBC is the loss of tolerance to the mitochondrial autoantigen35, 36 and the presence of AMA-M2 in serum is a hallmark of PBC.
For these reasons, our study focused on whether saliva might be valuable in diagnosing PBC. In this study, we found that the established biomarker for PBC, AMA-M2, could be detected in saliva. It was only detected at significant concentrations in the saliva of PBC-positive patients, but not HC subjects or OLP patients. Furthermore, salivary and serum AMA-M2 levels were positively correlated, and ROC analysis of salivary AMA-M2 demonstrated its high sensitivity and specificity for making a PBC diagnosis. A salivary AMA-M2 test would be a beneficial tool to screen or monitor groups at high-risk of PBC in the future.
It is reported that AMA-M2 levels are unaffected by treatment37. Meanwhile, we examined whether salivary AMA-M2 is related to ALP or GGT, either of which is elevated during the pathogenesis of cholestasis in PBC patients. Surprisingly, salivary AMA-M2 did not correlate with these vital indices of cholestasis. Cumulatively, our results suggested that detection of salivary AMA-M2 is a useful proxy for estimating serum AMA-M2 levels. Even though salivary AMA-M2 is not predictive of factors associated with cholestasis in PBC patients, saliva may still be an excellent alternative to serum for general diagnosis of this disease. Besides, in future, we will try to enlarge our sample size and prolong follow-up time to further clarify whether there exists potential relations between salivary AMA-M2 titer and clinical features.
PBC is a complex autoimmune disease that affects numerous organ systems. It is commonly concomitant with other disorders, including oral disease. Dry mouth and oral candidiasis are always found in PBC patients6, in addition to rampant dental caries38. This might imply that there is an imbalance in the oral immune environment in PBC patients. Therefore, we examined inflammatory cytokines in the saliva of patients with PBC. We found that salivary IL-6, IFN-γ, IL-17A, and TNF-α were higher in PBC patients compared to HCs. OLP is a chronic inflammatory disease of the oral mucosa. Several researchers have demonstrated that immune dysregulation and complex cytokine networks play important roles in the origin and development of OLP39,40,41. In this study, we similarly observed that salivary inflammatory cytokines were significantly increased in OLP patients. This may reflect similarities in the aberrant oral immune environment of PBC and OLP patients.
To date, current AMA-M2 kits are mostly designed for serum. Since our ROC analysis revealed that AMA-M2 levels in saliva are much lower than in serum, detection methods with higher sensitivity must be developed to achieve the clinical application of AMA-M2 salivary test. Thus, we further applied a new measurement technique called single molecule counting (SMC) technology42 to detect salivary AMA-M2. This method demonstrated a higher sensitivity in saliva tests. However, SMC technology is still new, and there are many limitations to practical applications, such as high costs and inconvenient operation. Nonetheless, with the rapid advancements in SMC and other technologies, the detection of salivary AMA-M2 will undoubtedly become easier and more accurate in the future.
In conclusion, our study showed that salivary AMA-M2 has important value for PBC diagnosis, and it might serve as a useful biomarker for this disease. We suggest that this approach may provide a new non-invasive method for the early diagnosis of PBC. In addition, inflammatory cytokine levels in saliva could be used to assess immune disorders of the oral cavity in these patients. A multi-disciplinary cooperative approach should be taken to ensure the wide clinical application of this diagnostic technique.
Methods
Ethics statement
Written informed consent was obtained from all the enrolled participants. The study protocol was conducted with the approval of the Ethics Review Committee of the First Affiliated Hospital, Zhejiang University (Permit number: 2016–261), and the use of human blood and saliva samples was in accordance with the Guidelines of the Declaration of Helsinki.
Subjects and Samples
We recruited 49 patients with PBC for this study, which included 42 females and 7 males. All serum and saliva samples from these patients were obtained from the First Affiliated Hospital at Zhejiang University, and stored at −80 °C for later analysis. The diagnosis of PBC was confirmed by clinical experts based upon the diagnostic criteria proposed by the AASLD9. Specifically, in previous clinical examination, immunoserological tests were positive for AMA-M2 in all 49 patients. Additionally, serum ALP levels ranged from 53 to 542. Sixty healthy individuals matched for sex and age were also recruited as controls. Clinical data from the 49 PBC patients and 60 healthy controls (HCs) were collected, including age, gender, and serum levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma-glutamyl transferase (GGT), alkaline phosphatase (ALP), and total bilirubin (TBIL) (Table 1). In order to explore the immunoenvironment of the oral cavity, saliva samples from 42 patients with oral lichen planus (OLP) matched for gender and age were collected and served as positive controls (Supplementary Table 1). All OLP patients were clinically confirmed, but none displayed systemic disease or received medication.
Measurement of Serum Cholestasis Indices
ALP and GGT were measured using a standard clinical automated analyzer (SRL, Tokyo, Japan).
Serum and Salivary AMA-M2 Detection
The levels of serum AMA-M2 in PBC patients and HC subjects were determined by enzyme-linked immunosorbent assay (ELISA) (Euroimmun, Germany) according to the manufacturer’s instructions.
To assess the levels of salivary AMA-M2, saliva samples were diluted with 0.1% Bull Serum Albumin-Phosphate Buffer Solution (BSA-PBS) (1:40). Subsequently, two-fold serial dilutions of the top standard from the ELISA kit using 0.1% BSA-PBS were performed to produce a standard curve for a total of eight points. Other experimental procedures on the salivary AMA-M2 test were the same as those performed on serum AMA-M2.
Luminex Bead Assay
The levels of serum and saliva cytokines were determined using a Bio-Plex Suspension Array System (Bio-Rad) with a commercial 7-plex cytokine (IL-4, IL-6, IL-10, IL-17, IL-23, IFN-γ, TNF-α) detection kit (Millipore, Germany) according to the manufacturer’s protocol.
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 6 (GraphPad Software, La Jolla, CA, USA). All values were expressed as the mean ± the standard error of the mean (SEM). Generally, differences between two groups were analyzed using the Mann-Whitney U test, and multigroup comparisons were performed by one-way ANOVA. Pearson’s correlation analysis was performed, and scatter plots were drawn to analyze the relationship between two groups. Receiver operating characteristics (ROC) curves were constructed to determine the optimal cut-off value. P < 0.05 was considered statistically significant. *P < 0.05; **P < 0.01; ***P < 0.001; ns, not significant.
References
de Almeida Pdel, V., Gregio, A. M., Machado, M. A., de Lima, A. A. & Azevedo, L. R. Saliva composition and functions: a comprehensive review. The journal of contemporary dental practice 9, 72–80 (2008).
Humphrey, S. P. & Williamson, R. T. A review of saliva: normal composition, flow, and function. The Journal of prosthetic dentistry 85, 162–169, doi:10.1067/mpr.2001.113778 (2001).
Pfaffe, T., Cooper-White, J., Beyerlein, P., Kostner, K. & Punyadeera, C. Diagnostic potential of saliva: current state and future applications. Clinical chemistry 57, 675–687, doi:10.1373/clinchem.2010.153767 (2011).
Kadashetti, V. et al. Glucose Level Estimation in Diabetes Mellitus By Saliva: A Bloodless Revolution. Romanian journal of internal medicine = Revue roumaine de medecine interne 53, 248–252, doi:10.1515/rjim-2015-0032 (2015).
Denver, R., Tzanidis, A., Martin, P. & Krum, H. Salivary endothelin concentrations in the assessment of chronic heart failure. Lancet (London, England) 355, 468–469, doi:10.1016/s0140-6736(00)82019-x (2000).
Carey, E. J., Ali, A. H. & Lindor, K. D. Primary biliary cirrhosis. Lancet (London, England) 386, 1565–1575, doi:10.1016/s0140-6736(15)00154-3 (2015).
Lisnevskaia, L., Murphy, G. & Isenberg, D. Systemic lupus erythematosus. Lancet (London, England) 384, 1878–1888, doi:10.1016/s0140-6736(14)60128-8 (2014).
Scott, D. L., Wolfe, F. & Huizinga, T. W. Rheumatoid arthritis. Lancet (London, England) 376, 1094–1108, doi:10.1016/s0140-6736(10)60826-4 (2010).
Lindor, K. D. et al. Primary biliary cirrhosis. Hepatology (Baltimore, Md.) 50, 291–308, doi:10.1002/hep.22906 (2009).
Chantran, Y., Ballot, E. & Johanet, C. Autoantibodies in primary biliary cirrhosis: antimitochondrial autoantibodies. Clinics and research in hepatology and gastroenterology 37, 431–433, doi:10.1016/j.clinre.2013.05.002 (2013).
Hu, S., Zhao, F., Wang, Q. & Chen, W. X. The accuracy of the anti-mitochondrial antibody and the M2 subtype test for diagnosis of primary biliary cirrhosis: a meta-analysis. Clinical chemistry and laboratory medicine 52, 1533–1542, doi:10.1515/cclm-2013-0926 (2014).
Walker, J. G., Doniach, D., Roitt, I. M. & Sherlock, S. Serological tests in diagnosis of primary biliary cirrhosis. Lancet (London, England) 1, 827–831, doi:10.1016/S0140-6736(65)91372-3 (1965).
Oertelt, S. et al. A sensitive bead assay for antimitochondrial antibodies: Chipping away at AMA-negative primary biliary cirrhosis. Hepatology (Baltimore, Md.) 45, 659–665, doi:10.1002/hep.21583 (2007).
Selmi, C., Bowlus, C. L., Gershwin, M. E. & Coppel, R. L. Primary biliary cirrhosis. Lancet (London, England) 377, 1600–1609, doi:10.1016/s0140-6736(10)61965-4 (2011).
Jacobs, R. et al. Host biomarkers detected in saliva show promise as markers for the diagnosis of pulmonary tuberculosis disease and monitoring of the response to tuberculosis treatment. Cytokine 81, 50–56, doi:10.1016/j.cyto.2016.02.004 (2016).
Isaza-Guzman, D. M. et al. Association study between salivary levels of interferon (IFN)-gamma, interleukin (IL)-17, IL-21, and IL-22 with chronic periodontitis. Archives of oral biology 60, 91–99, doi:10.1016/j.archoralbio.2014.09.002 (2014).
Ohyama, K. et al. Saliva as a potential tool for diagnosis of dry mouth including Sjogren’s syndrome. Oral diseases, doi: 10.1111/odi.12252 (2014).
Boppana, S. B. et al. Saliva polymerase-chain-reaction assay for cytomegalovirus screening in newborns. The New England journal of medicine 364, 2111–2118, doi:10.1056/NEJMoa1006561 (2011).
Poupon, R. Liver alkaline phosphatase: a missing link between choleresis and biliary inflammation. Hepatology (Baltimore, Md.) 61, 2080–2090, doi:10.1002/hep.27715 (2015).
Whitfield, J. B. Gamma glutamyl transferase. Critical reviews in clinical laboratory sciences 38, 263–355, doi:10.1080/20014091084227 (2001).
Srivastava, A. Progressive familial intrahepatic cholestasis. Journal of clinical and experimental hepatology 4, 25–36, doi:10.1016/j.jceh.2013.10.005 (2014).
Oliveira, H. M., Pereira, C., Santos Silva, E., Pinto-Basto, J. & Pessegueiro Miranda, H. Elevation of gamma-glutamyl transferase in adult: Should we think about progressive familiar intrahepatic cholestasis? Digestive and liver disease: official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver 48, 203–205, doi:10.1016/j.dld.2015.11.002 (2016).
Shi, T. et al. The Distribution and the Fibrotic Role of Elevated Inflammatory Th17 Cells in Patients With Primary Biliary Cirrhosis. Medicine 94, e1888, doi:10.1097/md.0000000000001888 (2015).
Yang, C. Y. et al. IL-12/Th1 and IL-23/Th17 biliary microenvironment in primary biliary cirrhosis: implications for therapy. Hepatology (Baltimore, Md.) 59, 1944–1953, doi:10.1002/hep.26979 (2014).
Qian, C. et al. Increased IL-23 and IL-17 expression by peripheral blood cells of patients with primary biliary cirrhosis. Cytokine 64, 172–180, doi:10.1016/j.cyto.2013.07.005 (2013).
Tan, Y. et al. Serum microRNAs as potential biomarkers of primary biliary cirrhosis. PloS one 9, e111424, doi:10.1371/journal.pone.0111424 (2014).
Wright, A. A. & Katz, I. T. Home testing for HIV. The New England journal of medicine 354, 437–440, doi:10.1056/NEJMp058302 (2006).
Corstjens, P. L., Abrams, W. R. & Malamud, D. Detecting viruses by using salivary diagnostics. Journal of the American Dental Association (1939) 143, 12s–18s, doi:10.14219/jada.archive.2012.0338 (2012).
Wild, B. et al. Temporal relationships between awakening cortisol and psychosocial variables in inpatients with anorexia nervosa - A time series approach. International journal of psychophysiology: official journal of the International Organization of Psychophysiology, doi: 10.1016/j.ijpsycho.2016.03.002 (2016).
Ellefsen, K. N., Concheiro, M., Pirard, S., Gorelick, D. A. & Huestis, M. A. Oral fluid cocaine and benzoylecgonine concentrations following controlled intravenous cocaine administration. Forensic science international 260, 95–101, doi:10.1016/j.forsciint.2016.01.013 (2016).
Javaid, M. A., Ahmed, A. S., Durand, R. & Tran, S. D. Saliva as a diagnostic tool for oral and systemic diseases. Journal of oral biology and craniofacial research 6, 66–75, doi:10.1016/j.jobcr.2015.08.006 (2016).
Zhang, L. et al. Discovery and preclinical validation of salivary transcriptomic and proteomic biomarkers for the non-invasive detection of breast cancer. PloS one 5, e15573, doi:10.1371/journal.pone.0015573 (2010).
Cabral, D. M., Antonini, S. R., Custodio, R. J., Martinelli, C. E. Jr. & da Silva, C. A. Measurement of salivary cortisol as a marker of stress in newborns in a neonatal intensive care unit. Hormone research in paediatrics 79, 373–378, doi:10.1159/000351942 (2013).
Mascarenhas, P., Fatela, B. & Barahona, I. Effect of diabetes mellitus type 2 on salivary glucose–a systematic review and meta-analysis of observational studies. PloS one 9, e101706, doi:10.1371/journal.pone.0101706 (2014).
Metcalf, J. V. et al. Natural history of early primary biliary cirrhosis. Lancet (London, England) 348, 1399–1402, doi:10.1016/s0140-6736(96)04410-8 (1996).
Hirschfield, G. M. & Gershwin, M. E. The immunobiology and pathophysiology of primary biliary cirrhosis. Annual review of pathology 8, 303–330, doi:10.1146/annurev-pathol-020712-164014 (2013).
Joshi, S. et al. Antimitochondrial antibody profiles: are they valid prognostic indicators in primary biliary cirrhosis? The American journal of gastroenterology 97, 999–1002, doi:10.1111/j.1572-0241.2002.05620.x (2002).
Richards, A., Rooney, J., Prime, S. & Scully, C. Primary biliary cirrhosis. Sole presentation with rampant dental caries. Oral surgery, oral medicine, and oral pathology 77, 16–18 (1994).
Schreurs, O., Karatsaidis, A. & Schenck, K. Phenotypically non-suppressive cells predominate among FoxP3-positive cells in oral lichen planus. Journal of oral pathology & medicine: official publication of the International Association of Oral Pathologists and the American Academy of Oral Pathology, doi: 10.1111/jop.12447 (2016).
Pekiner, F. N., Demirel, G. Y., Borahan, M. O. & Ozbayrak, S. Cytokine profiles in serum of patients with oral lichen planus. Cytokine 60, 701–706, doi:10.1016/j.cyto.2012.08.007 (2012).
Wang, Y., Zhou, J., Fu, S., Wang, C. & Zhou, B. A Study of Association Between Oral Lichen Planus and Immune Balance of Th1/Th2 Cells. Inflammation 38, 1874–1879, doi:10.1007/s10753-015-0167-4 (2015).
Gilbert, M., Livingston, R., Felberg, J. & Bishop, J. J. Multiplex single molecule counting (SMC) technology used to generate IL-4, IL-6 and IL-10 reference limits. Analytical biochemistry 503, 11–20, doi:10.1016/j.ab.2016.03.008 (2016).
Acknowledgements
We thank the contributions of other clinical and technical staffs of The First Affiliated Hospital, College of Medicine, Zhejiang University. This study was supported by the National Natural Science Foundation of China (Nos 81271810, 81571953), Zhejiang Provincial Natural Science Foundation of China (LY16H190002, Y13H140006) and the National Hygiene Bureau and Zhejiang Province Co-construction Project (2015118507, 2016139381).
Author information
Authors and Affiliations
Contributions
H.D. designed the experiments; C.L., H.J., J.C., P.Z. and H.H. performed the experiments; Y.W., J.C. and X.L. analyzed the data; X.H., L.W. and M.L. provided technical and material support. H.D., C.L. wrote the manuscript; all authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lu, C., Hou, X., Li, M. et al. Detection of AMA-M2 in human saliva: Potentials in diagnosis and monitoring of primary biliary cholangitis. Sci Rep 7, 796 (2017). https://doi.org/10.1038/s41598-017-00906-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-00906-1
This article is cited by
-
Soluble immune checkpoints are elevated in patients with primary biliary cholangitis
European Journal of Medical Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.