Abstract
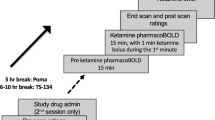
The National Institute of Mental Health (NIMH) ‘fast-fail’ approach seeks to improve too-often-misleading early-phase drug development methods by incorporating biomarker-based proof-of-mechanism (POM) testing in phase 2a. This first comprehensive application of the fast-fail approach evaluated the potential of κ-opioid receptor (KOR) antagonism for treating anhedonia with a POM study determining whether robust target engagement favorably impacts the brain circuitry hypothesized to mediate clinical effects. Here we report the results from a multicenter, 8-week, double-blind, placebo-controlled, randomized trial in patients with anhedonia and a mood or anxiety disorder (selective KOR antagonist (JNJ-67953964, 10 mg; n = 45) and placebo (n = 44)). JNJ-67953964 significantly increased functional magnetic resonance imaging (fMRI) ventral striatum activation during reward anticipation (primary outcome) as compared to placebo (baseline-adjusted mean: JNJ-67953964, 0.72 (s.d. = 0.67); placebo, 0.33 (s.d. = 0.68); F(1,86) = 5.58, P < 0.01; effect size = 0.58 (95% confidence interval, 0.13–0.99)). JNJ-67953964, generally well tolerated, was not associated with any serious adverse events. This study supports proceeding with assessment of the clinical impact of target engagement and serves as a model for implementing the ‘fast-fail’ approach.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Study data have been posted to the NIH/NIMH data archive and are accessible by emailing NDAHelp@mail.nih.gov.
References
Denys, D. & de Geus, F. Predictors of pharmacotherapy response in anxiety disorders. Curr. Psychiatry Rep. 7, 252–257 (2005).
Rush, A. J. et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am. J. Psychiatry 163, 1905–1917 (2006).
Insel, T. R. & Wang, P. S. The STAR*D trial: revealing the need for better treatments. Psychiatr. Serv. 60, 1466–1467 (2009).
National Advisory Mental Health Workgroup. From Discovery to Cure: Accelerating the Development of New and Personalized Interventions for Mental Illness (NIMH, 2010).
Paul, S. M. et al. How to improve R&D productivity: the pharmaceutical industry’s grand challenge. Nat. Rev. Drug Discov. 9, 203–214 (2010).
Kesselheim, A. S., Hwang, T. J. & Franklin, J. M. Two decades of new drug development for central nervous system disorders. Nat. Rev. Drug Discov. 14, 815–816 (2015).
Brady, L. S. & Insel, T. R. Translating discoveries into medicine: psychiatric drug development in 2011. Neuropsychopharmacology 37, 281–283 (2012).
Krystal, A. D. et al. The first implementation of the NIMH FAST-FAIL approach to psychiatric drug development. Nat. Rev. Drug Discov. 18, 82–84 (2018).
Wiedemann, K. Biomarkers in development of psychotropic drugs. Dialogues Clin. Neurosci. 13, 225–234 (2011).
Borsook, D., Becerra, L. & Fava, M. Use of functional imaging across clinical phases in CNS drug development. Transl. Psychiatry 16, e282 (2013).
Insel, T. et al. Research domain criteria (RDoC): toward a new classification framework for research on mental disorders. Am. J. Psychiatry 167, 748–751 (2010).
Whitton, A. E., Treadway, M. T. & Pizzagalli, D. A. Reward processing dysfunction in major depression, bipolar disorder and schizophrenia. Curr. Opin. Psychiatry 28, 7–12 (2015).
Carlezon, W. A. Jr & Krystal, A. D. κ-Opioid antagonists for psychiatric disorders: from bench to clinical trials. Depress. Anxiety 33, 895–906 (2016).
Carlezon, W. A. Jr et al. Depressive-like effects of the κ-opioid receptor agonist salvinorin A on behavior and neurochemistry in rats. J. Pharmacol. Exp. Ther. 316, 440–447 (2006).
Bruijnzeel, A. W. κ-Opioid receptor signaling and brain reward function. Brain Res. Rev. 62, 127–146 (2009).
Chartoff, E. et al. Blockade of κ-opioid receptors attenuates the development of depressive-like behaviors induced by cocaine withdrawal in rats. Neuropharmacology 62, 167–176 (2012).
Ebner, S. R., Roitman, M. F., Potter, D. N., Rachlin, A. B. & Chartoff, E. H. Depressive-like effects of the κ-opioid receptor agonist salvinorin A are associated with decreased phasic dopamine release in the nucleus accumbens. Psychopharmacology 210, 241–252 (2010).
Maisonneuve, I. M., Archer, S. & Glick, S. D. U50,488, a κ-opioid receptor agonist, attenuates cocaine-induced increases in extracellular dopamine in the nucleus accumbens of rats. Neurosci. Lett. 181, 57–60 (1994).
Muschamp, J. W. et al. Activation of CREB in the nucleus accumbens shell produces anhedonia and resistance to extinction of fear in rats. J. Neurosci. 31, 3095–3103 (2011).
Tomasiewicz, H. C., Todtenkopf, M. S., Chartoff, E. H., Cohen, B. M. & Carlezon, W. A. Jr. The κ-opioid agonist U69,593 blocks cocaine-induced enhancement of brain stimulation reward. Biol. Psychiatry 64, 982–988 (2008).
Wee, S. & Koob, G. F. The role of the dynorphin–kappa opioid system in the reinforcing effects of drugs of abuse. Psychopharmacology 210, 121–135 (2010).
Rorick-Kehn, L. M. et al. LY2456302 is a novel, potent, orally-bioavailable small molecule κ-selective antagonist with activity in animal models predictive of efficacy in mood and addictive disorders. Neuropharmacology 77C, 131–144 (2013).
Lowe, S. L. et al. Safety, tolerability, and pharmacokinetic evaluation of single- and multiple-ascending doses of a novel κ-opioid receptor antagonist LY2456302 and drug interaction with ethanol in healthy subjects. J. Clin. Pharmacol. 54, 968–978 (2014).
Zheng, M. Q. et al. Synthesis and evaluation of 11C-LY2795050 as a κ-opioid receptor antagonist radiotracer for PET imaging. J. Nucl. Med. 54, 455–463 (2013).
Carlezon, W. A. Jr, Béguin, C., Knoll, A. T. & Cohen, B. M. κ-Opioid ligands in the study and treatment of mood disorders. Pharmacol. Ther. 123, 334–343 (2009).
Knutson, B. & Gibbs, S. E. Linking nucleus accumbens dopamine and blood oxygenation. Psychopharmacology 191, 813–822 (2007).
Schott, B. H. et al. Mesolimbic functional magnetic resonance imaging activations during reward anticipation correlate with reward-related ventral striatal dopamine release. J. Neurosci. 28, 14311–14319 (2008).
Hedges, L. Distribution theory for Glass’s estimator of effect size and related estimators. J. Educat. Stat. 6, 107–128 (1981).
Green, I. W., Pizzagalli, D. A., Admon, R. & Kumar, P. Anhedonia modulates the effects of positive mood induction on reward-related brain activation. Neuroimage 193, 115–125 (2019).
Stoy, M. et al. Hyporeactivity of ventral striatum towards incentive stimuli in unmedicated depressed patients normalizes after treatment with escitalopram. J. Psychopharmacol. 26, 677–688 (2012).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences (Routledge, 1988).
Vrieze, E. et al. Reduced reward learning predicts outcome in major depressive disorder. Biol. Psychiatry 73, 639–645 (2013).
Kaiser, R. H. et al. Frontostriatal and dopamine markers of individual differences in reinforcement learning: a multi-modal investigation. Cereb. Cortex 28, 4281–4290 (2018).
Webb, C. A. et al. Neural correlates of three promising endophenotypes of depression: evidence from the EMBARC study. Neuropsychopharmacology 41, 454–463 (2016).
Pizzagalli, D. A., Jahn, A. L. & O’Shea, J. P. Toward an objective characterization of an anhedonic phenotype: a signal-detection approach. Biol. Psychiatry 57, 319–327 (2005).
Pizzagalli, D. A. et al. Single dose of a dopamine agonist impairs reinforcement learning in humans: behavioral evidence from a laboratory-based measure of reward responsiveness. Psychopharmacology 196, 221–232 (2008).
Pizzagalli, D. A., Iosifescu, D., Hallett, L. A., Ratner, K. G. & Fava, M. Reduced hedonic capacity in major depressive disorder: evidence from a probabilistic reward task. J. Psychiatr. Res. 43, 76–87 (2008).
Santesso, D. L. et al. Individual differences in reinforcement learning: behavioral, electrophysiological, and neuroimaging correlates. Neuroimage 42, 807–816 (2008).
Liu, W. et al. Deficits in sustaining reward responses in subsyndromal and syndromal major depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 35, 1045–1052 (2011).
Fletcher, K. et al. Anhedonia in melancholic and non-melancholic depressive disorders. J. Affect. Disord. 184, 81–88 (2015).
Morris, B. H., Bylsma, L. M., Yaroslavsky, I., Kovacs, M. & Rottenberg, J. Reward learning in pediatric depression and anxiety: preliminary findings in a high-risk sample. Depress. Anxiety 32, 373–381 (2015).
Jensen, J. et al. Direct activation of the ventral striatum in anticipation of aversive stimuli. Neuron 40, 1251–1257 (2003).
Carter, R. M., McInnes, J. J., Huettel, S. A. & Adcock, R. A. Activation in the VTA and nucleus accumbens increases in anticipation of both gains and losses. Front. Behav. Neurosci. 3, 1–15 (2009).
Snaith, R. P. et al. A scale for the assessment of hedonic tone the Snaith–Hamilton Pleasure Scale. Br. J. Psychiatry 167, 99–103 (1995).
Franken, I. H., Rassin, E. & Muris, P. The assessment of anhedonia in clinical and non-clinical populations: further validation of the Snaith–Hamilton Pleasure Scale (SHAPS). J. Affect. Disord. 99, 83–89 (2007).
Nakonezny, P. A. et al. Evaluation of anhedonia with the Snaith–Hamilton Pleasure Scale (SHAPS) in adult outpatients with major depressive disorder. J. Psychiatr. Res. 65, 124–130 (2015).
Nakonezny, P. A., Carmody, T. J., Morris, D. W., Kurian, B. T. & Trivedi, M. H. Psychometric evaluation of the Snaith–Hamilton pleasure scale in adult outpatients with major depressive disorder. Int. Clin. Psychopharmacol. 25, 328–333 (2010).
Hamilton, M. Development of a rating scale for primary depressive illness. Br. J. Soc. Psychol. 6, 278–296 (1967).
Maier, W., Buller, R., Philipp, M. & Heuser, I. The Hamilton Anxiety Scale: reliability, validity and sensitivity to change in anxiety and depressive disorders. J. Affect. Disord. 14, 61–68 (1988).
Knutson, B., Westdorp, A., Kaiser, E. & Hommer, D. FMRI visualization of brain activity during a monetary incentive delay task. Neuroimage 12, 20–27 (2000).
Knutson, B., Bhanji, J. P., Cooney, R. E., Atlas, L. Y. & Gotlib, I. H. Neural responses to monetary incentives in major depression. Biol. Psychiatry 63, 686–692 (2008).
Knutson, B., Fong, G. W., Adams, C. M., Varner, J. L. & Hommer, D. Dissociation of reward anticipation and outcome with event-related fMRI. Neuroreport. 12, 3683–3687 (2001).
Knutson, B., Adams, C. M., Fong, G. W. & Hommer, D. Anticipation of increasing monetary reward selectively recruits nucleus accumbens. J. Neurosci. 21, RC159 (2001).
Knutson, B., Wimmer, G. E., Kuhnen, C. M. & Winkielman, P. Nucleus accumbens activation mediates the influence of reward cues on financial risk taking. Neuroreport. 19, 509–513 (2008).
Carlson, J. M., Foti, D., Mujica-Parodi, L. R., Harmon-Jones, E. & Hajcak, G. Ventral striatal and medial prefrontal BOLD activation is correlated with reward-related electrocortical activity: a combined ERP and fMRI study. Neuroimage 57, 1608–1616 (2011).
Dillon, D. G. et al. Dissociation of neural regions associated with anticipatory versus consummatory phases of incentive processing. Psychophysiology 45, 36–49 (2008).
Keedwell, P. A., Andrew, C., Williams, S. C., Brammer, M. J. & Phillips, M. L. The neural correlates of anhedonia in major depressive disorder. Biol. Psychiatry 58, 843–853 (2005).
Keller, J. et al. Trait anhedonia is associated with reduced reactivity and connectivity of mesolimbic and paralimbic reward pathways. J. Psychiatr. Res. 47, 1319–1328 (2013).
Li, Z. et al. Improving motivation through real-time fMRI-based self-regulation of the nucleus accumbens. Neuropsychology 32, 764–776 (2018).
Oldham, S. et al. The anticipation and outcome phases of reward and loss processing: a neuroimaging meta-analysis of the monetary incentive delay task. Hum. Brain Mapp. 39, 3398–3418 (2018).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders 4th edn (American Psychiatric Association, 2009).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59, 22–33 (1998).
Posner, K. Columbia–Suicide Severity Rating Scale (C-SSRS) http://www.cssrs.columbia.edu (2011).
Jenkinson, M., Bannister, P., Brady, M. & Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17, 825–841 (2002).
Jenkinson, M. & Smith, S. A global optimisation method for robust affine registration of brain images. Med. Image Anal. 5, 143–156 (2001).
Andersson, J. L. R., Jenkinson, M. & Smith, S. Non-linear registration, aka Spatial normalisation. FMRIB technical report TR07JA2 FMRIB Centre, 2007.
Smith, S. M. Fast robust automated brain extraction. Hum. Brain Mapp. 17, 143–155 (2002).
Woolrich, M. W., Ripley, B. D., Brady, M. & Smith, S. M. Temporal autocorrelation in univariate linear modeling of fMRI data. Neuroimage 14, 1370–1386 (2001).
Woolrich, M. W., Behrens, T. E. J., Beckmann, C. F., Jenkinson, M. & Smith, S. M. Multilevel linear modelling for fMRI group analysis using Bayesian inference. Neuroimage 21, 1732–1747 (2004).
Woolrich, M. W. et al. Bayesian analysis of neuroimaging data in FSL. Neuroimage 45, S173–S186 (2009).
Friedman, L. & Glover, G. H. Report on a multicenter fMRI quality assurance protocol. J. Magn. Reson. Imaging 23, 827–839 (2006).
Di Giannantonio, M. & Martinotti, G. Anhedonia and major depression: the role of agomelatine. Eur. Neuropsychopharmacol. 22, S505–S510 (2012).
Martinotti, G. et al. Acetyl-l-carnitine in the treatment of anhedonia, melancholic and negative symptoms in alcohol dependent subjects. Prog. Neuropsychopharmacol. Biol. Psychiatry 35, 953–958 (2011).
Wacker, J., Dillon, D. G. & Pizzagalli, D. A. The role of the nucleus accumbens and rostral anterior cingulate cortex in anhedonia: integration of resting EEG, fMRI, and volumetric techniques. Neuroimage 46, 327–337 (2009).
Treadway, M. T., Buckholtz, J. W., Schwartzman, A. N., Lambert, W. E. & Zald, D. H. Worth the ‘EEfRT’? The effort expenditure for rewards task as an objective measure of motivation and anhedonia. PLoS One 12, e6598 (2009).
Gard, D. E., Gard-Germans, M., Kring, A. M. & Oliver, J. P. Anticipatory and consummatory components of the experience of pleasure: a scale development study. J. Res. Personality 40, 1086–1102 (2006).
Fava, M., Iosifescu, D. V., Pedrelli, P. & Baer, L. Reliability and validity of the Massachusetts General Hospital cognitive and physical functioning questionnaire. Psychother. Psychosom. 78, 91–97 (2009).
Wilson, R. P. et al. The neural substrate of reward anticipation in health: a meta-analysis of fMRI findings in the monetary incentive delay task. Neuropsychol. Rev. 28, 496–506 (2018).
Kraemer, H. C. & Blasey, C. M. Centering in regression analyses: a strategy to prevent errors in statistical inference. Int. J. Methods Psychiatr. Res. 13, 141–151 (2004).
Azur, M. J., Stuart, E. A., Frangakis, C. & Leaf, P. J. Multiple imputation by chained equations: what is it and how does it work? Int. J. Methods Psychiatr. Res. 20, 40–49 (2011).
Acknowledgements
This project was supported by contract HHS-N271-2012-000006-I from the National Institute of Mental Health awarded to A.D.K. D.A.P. was partially supported by R37 MH068376. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the US government.
Author information
Authors and Affiliations
Contributions
A.D.K. designed the study, provided oversight for all aspects of the study, analyzed the data and wrote the manuscript. W.Z.P. provided critical intellectual input for designing the study and contributed to writing the manuscript. D.A.P. provided critical intellectual input for designing the study, provided training, oversight and data analysis for the aspects of the study related to the behavioral tests, and co-wrote the manuscript. A.E.W. provided training, oversight and data analysis for the aspects of the study related to the behavioral tests. S.J.M., S.H.L., J.R.C., G.S. and W.G. contributed to designing the study, provided oversight for a study site where they led efforts in training and managing study personnel, recruiting participants and completing all protocol-specified study procedures including data collection with the participants, and contributed to writing the manuscript. R.D.W. and J.N. provided oversight for a study site where they led efforts in training and managing study personnel, recruiting participants and completing all protocol-specified study procedures including data collection with the participants and contributed to writing the manuscript. A.S. and M.S. provided oversight for collection and analysis of the MRI data and contributed to writing the manuscript. H.Y. served as statistician for the study, contributing to study design, carrying out data analysis and contributing to writing the manuscript. R.S.E.K. contributed to designing the study, provided data quality oversight and contributed to writing the manuscript. D.I., S.T.S., G.H. and K.G. participated in recruiting participants, completing all protocol-specified study procedures including data collection with the participants, and contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
A.D.K. has been a consultant for Adare, Eisai, Ferring, Galderma, Idorsia, Jazz, Janssen, Takeda, Merck, Neurocrine, Pernix, Physician’s Seal, Evecxia and Sage Research and received support from the NIH, the Ray and Dagmar Dolby Family Fund, Janssen, Jazz. Axsome and Reveal Biosensors. D.A.P. has consulted for Akili Interactive Labs, BlackThorn Therapeutics, Boehringer Ingelheim, Compass Pathway, Otsuka Pharmaceuticals, Posit Science and Takeda Pharmaceuticals and received honoraria from Alkermes. D.A.P. has a financial interest in BlackThorn Therapeutics, which has licensed the copyright to the PRT through Harvard University. The interests of D.A.P. were reviewed and are managed by McLean Hospital and Partners HealthCare in accordance with their conflict-of-interest policies. No funding from these entities was used to support the current work, and all views expressed are solely those of the authors. S.J.M. has been a consultant for Allergan, Alkermes, Greenwich Biosciences, Clexio Biosciences, Intra-Cellular Therapies, Janssen, Perception Neuroscience, Sage Therapeutics, Signant Health and Seelos Therapeutics and received research support from Biohaven, Janssen, the NIH, NeuroRx and VistaGen Therapeutics, drug from Biohaven for NIMH-funded study and support from the Michael E. Debakey VA Medical Center (Houston, TX) for use of resources and facilities. G.S. has consulted for Allergan, Alkermes, AstraZeneca, Avanier Pharmaceuticals, Axsome Therapeutics, Biohaven Pharmaceuticals, Boehringer Ingelheim, Bristol-Myers Squibb, Clexio Biosciences, EMA-Wellness, Hoffman La-Roche, Intra-Cellular Therapies, Janssen, Merck, Naurex, Navitor Pharmaceuticals, Novartis, Noven Pharmaceuticals, Otsuka, Praxis Therapeutics, Sage Pharmaceuticals, Servier Pharmaceuticals, Taisho Pharmaceuticals, Teva, Valeant and Vistagen Therapeutics and received research funding from AstraZeneca, Bristol-Myers Squibb, Eli Lilly, Johnson & Johnson, Hoffman La-Roche, Merck, Naurex and Servier. G.S. has equity interests in Biohaven Pharmaceuticals and receives patent royalties from Biohaven. J.W.M. has consulted for Boehringer Ingelheim, Sage Therapeutics, Novartis, Allergan, Fortress Biotech, Janssen Research and Development, Medavante-Prophase and Global Medical Education (GME) and received research support from Avanir Pharmaceuticals. R.S.E.K. has consulted for or been a speaker or advisory board member for Abbvie, Acadia, Aeglea, Akebia, Akili, Alkermes, Allergan, ArmaGen, Astellas, Avanir, Axovant, Biogen, Boehringer Ingelheim, Cerecor, CoMentis, Critical Path Institute, FORUM, Gammon Howard & Zeszotarski, Global Medical Education (GME), GW Pharmaceuticals, Intracellular Therapeutics, Janssen, Kempharm, Lundbeck, Lysogene, MedScape, Mentis Cura, Merck, Merrakris Therapetics, Minerva Neurosciences, Mitsubishi, Montana State University, Monteris, Moscow Research Institute of Psychiatry, Neuralstem, Neuronix, Novartis, the New York State Office of Mental Health, Orygen, Otsuka, Paradigm Testing, Percept Solutions, Pfizer, Pharm-Olam, Regenix Bio, Reviva, Roche, Sangamo, Sanofi, SOBI, Six Degrees Medical, Sunovion, Takeda, Targacept, Teague Rotenstreich Stanaland Fox & Holt, Thrombosis Research Institute, the University of Moscow, the University of Southern California, the University of Texas Southwestern Medical Center, WebMD and Wilson Therapeutics. R.S.E.K. has received research funding from the National Institute of Mental Health and Boehringer Ingelheim and royalties from Versions of the BAC testing battery, the MATRICS Battery (BACS Symbol Coding) and the Virtual Reality Functional Capacity Assessment Tool (VRFCAT). R.S.E.K. is a shareholder in VeraSci and Sengenix. S.H.L. contributed to this article while at Duke University, before joining the National Institute of Mental Health. The views expressed are her own and do not necessarily represent the views of the National Institutes of Health or the US government. S.H.L. is a co-inventor on a patent for TMS Technology, unrelated to this manuscript. J.N. has received research funding from Janssen and Assurex. A.S. has received research support from the NIH and GE Healthcare. W.G. has consulted for Biohaven Pharmaceuticals and received research funding from the NIH, the Simons Foundation and Biohaven Pharmaceuticals Device Donation: Medtronic. S.T.S. has consulted for Otsuka Pharmaceuticals, Neurocrine Biosciences, Jazz, Teva and the Centers of Psychiatric Excellence and Continuous Precision Medicine and received research support from Otsuka Pharmaceuticals and Veterans’ Affairs grant IK2CX001397. K.G. has been on the advisory board of Sunovion and Otsuka Pharmaceuticals and received research funding from AstraZeneca, the Cleveland Foundation and the Brain and Behavioral Research Foundation. K.G. has been a speaker for AstraZeneca, Pfizer and Sunovion. W.Z.P. has been on the advisory board and/or consulted for Takeda, Lilly, Praxis, Astellas, Otsuka and Noven. W.Z.P. has been on the DSMB for Agene-Bio and has stock ownership in Merck.
Additional information
Peer review information Kate Gao was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Consort diagram.
Study consort diagram.
Supplementary information
Supplementary Information
Supplementary Note and Supplementary Tables 1–3.
Rights and permissions
About this article
Cite this article
Krystal, A.D., Pizzagalli, D.A., Smoski, M. et al. A randomized proof-of-mechanism trial applying the ‘fast-fail’ approach to evaluating κ-opioid antagonism as a treatment for anhedonia. Nat Med 26, 760–768 (2020). https://doi.org/10.1038/s41591-020-0806-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-020-0806-7
This article is cited by
-
Psychosis superspectrum II: neurobiology, treatment, and implications
Molecular Psychiatry (2024)
-
Advancing preclinical chronic stress models to promote therapeutic discovery for human stress disorders
Neuropsychopharmacology (2024)
-
Will the promise of translational neuropsychopharmacology research ever deliver? The lion’s roar; the kitten’s purr
NPP—Digital Psychiatry and Neuroscience (2024)
-
Efficacy and safety of aticaprant, a kappa receptor antagonist, adjunctive to oral SSRI/SNRI antidepressant in major depressive disorder: results of a phase 2 randomized, double-blind, placebo-controlled study
Neuropsychopharmacology (2024)
-
Finding new and better treatments for psychiatric disorders
Neuropsychopharmacology (2024)