Abstract
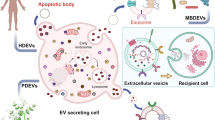
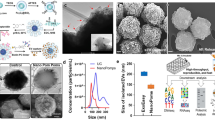
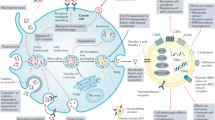
Extracellular vesicles (EVs) have an essential functional role in local tumour progression, metastatic spread and the emergence of drug resistance in bladder, kidney and prostate cancer. Thus, EVs could be diagnostic, prognostic and predictive biomarkers for these malignancies. Virtually all biomolecules (including DNA, mRNA, microRNA, long non-coding RNA, proteins and lipids) packaged into EVs have been tested as biomarkers in blood and urine samples. The results are very heterogeneous, but promising biomarker candidates have been identified. Differing methods of EV isolation, characterization and analysis of their content have been used owing to a lack of international consensus; hence, comparing study results is challenging. Furthermore, validation of potential biomarkers in independent cohorts or prospective trials has rarely been performed. Future efforts to establish EV-derived biomarkers need to adequately address these points. In addition, emerging technologies such as mass spectroscopy and chip-based approaches can identify surface markers specific for cancer-associated EVs and will enable specific separation from blood and urine EVs, which probably will improve their performance as biomarkers. Moreover, EVs could be harnessed as therapeutic drug delivery vehicles for precise and effective anticancer therapy.
Key points
-
Extracellular vesicles (EVs) isolated from different body fluids reflect the molecular profile of their parental cells and are, therefore, a new class of biomarkers in liquid biopsies from patients with urological cancer.
-
Biomarker studies are mainly focused on mRNAs and microRNAs as the most promising diagnostic tools using EVs in bladder, kidney and prostate cancer; data on prognostic and predictive EV markers are still limited.
-
Comparability and reproducibility of published data is complicated because different isolation techniques have been used; in order to introduce EVs into clinical management, standardized techniques for routine isolation and characterization need to be defined.
-
Identification of tumour-specific EV markers (such as prostate-specific membrane antigen in prostate cancer) will increase their diagnostic accuracy.
-
EVs regulate communication between tumour cells and the microenvironment supporting tumour development and prepare premetastatic niches via systemic circulation to distant sites; EVs stimulate angiogenesis, support immune evasion and affect therapy response in bladder, kidney and prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
18 December 2019
A Correction to this paper has been published: https://doi.org/10.1038/s41585-019-0279-y
References
Thery, C. et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 7, 1535750 (2018).
Akers, J. C., Gonda, D., Kim, R., Carter, B. S. & Chen, C. C. Biogenesis of extracellular vesicles (EV): exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies. J. Neurooncol. 113, 1–11 (2013).
Hessvik, N. P. & Llorente, A. Current knowledge on exosome biogenesis and release. Cell Mol. Life Sci. 75, 193–208 (2018).
Duijvesz, D., Luider, T., Bangma, C. H. & Jenster, G. Exosomes as biomarker treasure chests for prostate cancer. Eur. Urol. 59, 823–831 (2011).
van der Pol, E., Boing, A. N., Gool, E. L. & Nieuwland, R. Recent developments in the nomenclature, presence, isolation, detection and clinical impact of extracellular vesicles. J. Thromb. Haemost. 14, 48–56 (2016).
Kerr, J. F., Wyllie, A. H. & Currie, A. R. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer 26, 239–257 (1972).
Atkin-Smith, G. K. et al. A novel mechanism of generating extracellular vesicles during apoptosis via a beads-on-a-string membrane structure. Nat. Commun. 6, 7439 (2015).
Hauser, P., Wang, S. & Didenko, V. V. Apoptotic bodies: selective detection in extracellular vesicles. Methods Mol. Biol. 1554, 193–200 (2017).
Stridsberg, M., Fabiani, R., Lukinius, A. & Ronquist, G. Prostasomes are neuroendocrine-like vesicles in human semen. Prostate 29, 287–295 (1996).
Carlsson, L. et al. Characteristics of human prostasomes isolated from three different sources. Prostate 54, 322–330 (2003).
Aalberts, M., Stout, T. A. & Stoorvogel, W. Prostasomes: extracellular vesicles from the prostate. Reproduction 147, R1–R14 (2014).
Zijlstra, C. & Stoorvogel, W. Prostasomes as a source of diagnostic biomarkers for prostate cancer. J. Clin. Invest. 126, 1144–1151 (2016).
Gould, S. J. & Raposo, G. As we wait: coping with an imperfect nomenclature for extracellular vesicles. J. Extracell. Vesicles 2, 20389 (2013).
Mulcahy, L. A., Pink, R. C. & Carter, D. R. Routes and mechanisms of extracellular vesicle uptake. J. Extracell. Vesicles 3, 24641 (2014).
Abels, E. R. & Breakefield, X. O. Introduction to extracellular vesicles: biogenesis, RNA cargo selection, content, release, and uptake. Cell. Mol. Neurobiol. 36, 301–312 (2016).
O'Driscoll, L. Expanding on exosomes and ectosomes in cancer. N Engl. J. Med. 372, 2359–2362 (2015).
Junker, K., Heinzelmann, J., Beckham, C., Ochiya, T. & Jenster, G. Extracellular vesicles and their role in urologic malignancies. Eur. Urol. 70, 323–331 (2016).
Nawaz, M. et al. The emerging role of extracellular vesicles as biomarkers for urogenital cancers. Nat. Rev. Urol. 11, 688–701 (2014).
Tkach, M. & Thery, C. Communication by extracellular vesicles: where we are and where we need to go. Cell 164, 1226–1232 (2016).
Kosaka, N., Yoshioka, Y., Fujita, Y. & Ochiya, T. Versatile roles of extracellular vesicles in cancer. J. Clin. Invest. 126, 1163–1172 (2016).
Madeo, M. et al. Cancer exosomes induce tumor innervation. Nat. Commun. 9, 4284 (2018).
Ringuette Goulet, C. et al. Exosomes induce fibroblast differentiation into cancer-associated fibroblasts through TGFbeta signaling. Mol. Cancer Res. 16, 1196–1204 (2018).
Hoshino, A. et al. Tumour exosome integrins determine organotropic metastasis. Nature 527, 329–335 (2015).
Zhang, L. et al. Microenvironment-induced PTEN loss by exosomal microRNA primes brain metastasis outgrowth. Nature 527, 100–104 (2015).
Roccaro, A. M. et al. BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. J. Clin. Invest. 123, 1542–1555 (2013).
Ono, M. et al. Exosomes from bone marrow mesenchymal stem cells contain a microRNA that promotes dormancy in metastatic breast cancer cells. Sci. Signal. 7, ra63 (2014).
Zhao, H. et al. Tumor microenvironment derived exosomes pleiotropically modulate cancer cell metabolism. eLife 5, e10250 (2016).
Lobb, R. J. et al. Exosomes derived from mesenchymal non-small cell lung cancer cells promote chemoresistance. Int. J. Cancer 141, 614–620 (2017).
Qu, L. et al. Exosome-transmitted lncARSR promotes sunitinib resistance in renal cancer by acting as a competing endogenous RNA. Cancer Cell 29, 653–668 (2016).
Tang, Y. T. et al. Comparison of isolation methods of exosomes and exosomal RNA from cell culture medium and serum. Int. J. Mol. Med. 40, 834–844 (2017).
Ding, M. et al. Comparison of commercial exosome isolation kits for circulating exosomal microRNA profiling. Anal. Bioanal. Chem. 410, 3805–3814 (2018).
Chiriaco, M. S. et al. Lab-on-chip for exosomes and microvesicles detection and characterization. Sensors 18, E3175 (2018).
Sharma, S., LeClaire, M. & Gimzewski, J. K. Ascent of atomic force microscopy as a nanoanalytical tool for exosomes and other extracellular vesicles. Nanotechnology 29, 132001 (2018).
Grasso, L. et al. Molecular screening of cancer-derived exosomes by surface plasmon resonance spectroscopy. Anal. Bioanal. Chem. 407, 5425–5432 (2015).
Padda, R. S. et al. Nanoscale flow cytometry to distinguish subpopulations of prostate extracellular vesicles in patient plasma. Prostate 79, 592–603 (2019).
Morales-Kastresana, A. et al. Labeling extracellular vesicles for nanoscale flow cytometry. Sci. Rep. 7, 1878 (2017).
Jimenez, L. et al. Quantitative proteomic analysis of small and large extracellular vesicles (EVs) reveals enrichment of adhesion proteins in small EVs. J. Proteome Res. 18, 947–959 (2019).
Kreimer, S. et al. Mass-spectrometry-based molecular characterization of extracellular vesicles: lipidomics and proteomics. J. Proteome Res. 14, 2367–2384 (2015).
McGranahan, N. & Swanton, C. Biological and therapeutic impact of intratumor heterogeneity in cancer evolution. Cancer Cell 27, 15–26 (2015).
Corcoran, R. B. & Chabner, B. A. Application of cell-free DNA analysis to cancer treatment. N. Engl. J. Med. 379, 1754–1765 (2018).
Gorin, M. A. et al. Circulating tumour cells as biomarkers of prostate, bladder, and kidney cancer. Nat. Rev. Urol. 14, 90–97 (2017).
Hille, C. & Pantel, K. Prostate cancer: circulating tumour cells in prostate cancer. Nat. Rev. Urol. 15, 265–266 (2018).
Werner, S., Keller, L. & Pantel, K. Epithelial keratins: biology and implications as diagnostic markers for liquid biopsies. Mol. Aspects Med. https://doi.org/10.1016/j.mam.2019.09.001 (2019).
Mader, S. & Pantel, K. Liquid biopsy: current status and future perspectives. Oncol. Res. Treat. 40, 404–408 (2017).
Volik, S., Alcaide, M., Morin, R. D. & Collins, C. Cell-free DNA (cfDNA): clinical significance and utility in cancer shaped by emerging technologies. Mol. Cancer Res. 14, 898–908 (2016).
Arroyo, J. D. et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl Acad. Sci. USA 108, 5003–5008 (2011).
Lotan, Y. & Roehrborn, C. G. Sensitivity and specificity of commonly available bladder tumor markers versus cytology: results of a comprehensive literature review and meta-analyses. Urology 61, 109–118 (2003).
Schmitz-Drager, B. J. et al. Molecular markers for bladder cancer screening, early diagnosis, and surveillance: the WHO/ICUD consensus. Urol. Int. 94, 1–24 (2015).
Mowatt, G. et al. Systematic review of the clinical effectiveness and cost-effectiveness of photodynamic diagnosis and urine biomarkers (FISH, ImmunoCyt, NMP22) and cytology for the detection and follow-up of bladder cancer. Health Technol. Assess. 14, 1–331 (2010).
Armstrong, D. A., Green, B. B., Seigne, J. D., Schned, A. R. & Marsit, C. J. MicroRNA molecular profiling from matched tumor and bio-fluids in bladder cancer. Mol. Cancer 14, 194 (2015).
Baumgart, S. et al. Invasion-associated miRNAs S as possible diagnostic biomarkers of muscle invasive bladder cancer in tumor tissues and urinary exosomes. J. Urol. 199, E1038 (2018).
Andreu, Z. et al. Extracellular vesicles as a source for non-invasive biomarkers in bladder cancer progression. Eur. J. Pharm. Sci. 98, 70–79 (2017).
De Long, J. et al. A non-invasive miRNA based assay to detect bladder cancer in cell-free urine. Am. J. Transl. Res. 7, 2500–250 (2015).
Royo, F. et al. Comparative miRNA analysis of urine extracellular vesicles isolated through five different methods. Cancers 8, E112 (2016).
Berrondo, C. et al. Expression of the long non-coding RNA HOTAIR correlates with disease progression in bladder cancer and is contained in bladder cancer patient urinary exosomes. PLOS ONE 11, e0147236 (2016).
Sanger, H. L., Klotz, G., Riesner, D., Gross, H. J. & Kleinschmidt, A. K. Viroids are single-stranded covalently closed circular RNA molecules existing as highly base-paired rod-like structures. Proc. Natl Acad. Sci. USA 73, 3852–3856 (1976).
Chen, X. et al. PRMT5 circular RNA promotes metastasis of urothelial carcinoma of the bladder through sponging miR-30c to induce epithelial-mesenchymal transition. Clin. Cancer Res. 24, 6319–6330 (2018).
Katsuda, T., Kosaka, N. & Ochiya, T. The roles of extracellular vesicles in cancer biology: toward the development of novel cancer biomarkers. Proteomics 14, 412–425 (2014).
Murakami, T. et al. Bladder cancer detection by urinary extracellular vesicle mRNA analysis. Oncotarget 9, 32810–32821 (2018).
Afsharpad, M. et al. Cancer-testis antigens as new candidate diagnostic biomarkers for transitional cell carcinoma of bladder. Pathol. Oncol. Res. 25, (191–199 (2019).
Esfandiary, A. & Ghafouri-Fard, S. New York esophageal squamous cell carcinoma-1 and cancer immunotherapy. Immunotherapy 7, 411–439 (2015).
Yazarlou, F. et al. Urine exosome gene expression of cancer-testis antigens for prediction of bladder carcinoma. Cancer Manag. Res. 10, 5373–5381 (2018).
Lee, J. et al. Altered proteome of extracellular vesicles derived from bladder cancer patients urine. Mol. Cell 41, 179–187 (2018).
Silvers, C. R., Miyamoto, H., Messing, E. M., Netto, G. J. & Lee, Y. F. Characterization of urinary extracellular vesicle proteins in muscle-invasive bladder cancer. Oncotarget 8, 91199–91208 (2017).
Lin, S. Y. et al. Proteome profiling of urinary exosomes identifies alpha 1-antitrypsin and H2B1K as diagnostic and prognostic biomarkers for urothelial carcinoma. Sci. Rep. 6, 34446 (2016).
Silvers, C. R. et al. Identification of extracellular vesicle-borne periostin as a feature of muscle-invasive bladder cancer. Oncotarget 7, 23335–23345 (2016).
Zhang, S. et al. Evaluation of serum exosomal lncRNA-based biomarker panel for diagnosis and recurrence prediction of bladder cancer. J. Cell. Mol. Med. 23, 1396–1405 (2018).
Xue, M. et al. Hypoxic exosomes facilitate bladder tumor growth and development through transferring long non-coding RNA-UCA1. Mol. Cancer 16, 143 (2017).
Marconi, L. et al. Systematic review and meta-analysis of diagnostic accuracy of percutaneous renal tumour biopsy. Eur. Urol. 69, 660–673 (2016).
Zhang, W. et al. MicroRNAs in serum exosomes as potential biomarkers in clear-cell renal cell carcinoma. Eur. Urol. Focus 4, 412–419 (2018).
Zimpfer, A. et al. Prognostic and diagnostic implications of epithelial cell adhesion/activating molecule (EpCAM) expression in renal tumours: a retrospective clinicopathological study of 948 cases using tissue microarrays. BJU Int. 114, 296–302 (2014).
Wang, X. et al. Serum exosomal miR-210 as a potential biomarker for clear cell renal cell carcinoma. J. Cell. Biochem. https://doi.org/10.1002/jcb.27347 (2018).
Liu, H. et al. Identifying mRNA targets of microRNA dysregulated in cancer: with application to clear cell renal cell carcinoma. BMC Syst. Biol. 4, 51 (2010).
Fujii, N. et al. Extracellular miR-224 as a prognostic marker for clear cell renal cell carcinoma. Oncotarget 8, 109877–109888 (2017).
Du, M. J. et al. Plasma exosomal miRNAs-based prognosis in metastatic kidney cancer. Oncotarget 8, 63703–63714 (2017).
Butz, H. et al. Exosomal microRNAs are diagnostic biomarkers and can mediate cell–cell communication in renal cell carcinoma. Eur. Urol. Focus 2, 210–218 (2016).
Crentsil, V. C., Liu, H. & Sellitti, D. F. Comparison of exosomal microRNAs secreted by 786-O clear cell renal carcinoma cells and HK-2 proximal tubule-derived cells in culture identifies microRNA-205 as a potential biomarker of clear cell renal carcinoma. Oncol. Lett. 16, 1285–1290 (2018).
De Palma, G. et al. The three-gene signature in urinary extracellular vesicles from patients with clear cell renal cell carcinoma. J. Cancer 7, 1960–1967 (2016).
Raimondo, F. et al. Differential protein profiling of renal cell carcinoma urinary exosomes. Mol. Biosyst. 9, 1220–1233 (2013).
Del Boccio, P. et al. A hyphenated microLC-Q-TOF-MS platform for exosomal lipidomics investigations: application to RCC urinary exosomes. Electrophoresis 33, 689–696 (2012).
Boyd, L. K., Mao, X. & Lu, Y. J. The complexity of prostate cancer: genomic alterations and heterogeneity. Nat. Rev. Urol. 9, 652–664 (2012).
Grasso, C. S. et al. The mutational landscape of lethal castration-resistant prostate cancer. Nature 487, 239–243 (2012).
Fraser, M. et al. Genomic hallmarks of localized, non-indolent prostate cancer. Nature 541, 359–364 (2017).
Fitzpatrick, J. M., Banu, E. & Oudard, S. Prostate-specific antigen kinetics in localized and advanced prostate cancer. BJU Int. 103, 578–587 (2009).
Mottet, N. et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur. Urol. 71, 618–629 (2017).
Cornford, P. et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part II: treatment of relapsing, metastatic, and castration-resistant prostate cancer. Eur. Urol. 71, 630–642 (2017).
Heijnsdijk, E. A. M. et al. Summary statement on screening for prostate cancer in Europe. Int. J. Cancer 142, 741–746 (2018).
Catalona, W. J. Prostate cancer screening. Med. Clin. North. Am. 102, 199–214 (2018).
Antonarakis, E. S. et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl. J. Med. 371, 1028–1038 (2014).
Mateo, J. et al. DNA-repair defects and olaparib in metastatic prostate cancer. N. Engl. J. Med. 373, 1697–1708 (2015).
de Bono, J. S. et al. Randomized phase II study evaluating Akt blockade with ipatasertib, in combination with abiraterone, in patients with metastatic prostate cancer with and without PTEN loss. Clin. Cancer Res. 25, 928–936 (2019).
Luo, J. et al. Role of androgen receptor variants in prostate cancer: report from the 2017 Mission Androgen Receptor Variants Meeting. Eur. Urol. 73, 715–723 (2018).
Fletcher, C. AR-v7 liquid biopsy for treatment stratification in prostate cancer: how close are we? Curr. Opin. Urol. 27, 500–509 (2017).
Pan, J., Ding, M., Xu, K., Yang, C. & Mao, L. J. Exosomes in diagnosis and therapy of prostate cancer. Oncotarget 8, 97693–97700 (2017).
Valentino, A. et al. Exosomal microRNAs in liquid biopsies: future biomarkers for prostate cancer. Clin. Transl. Oncol. 19, 651–657 (2017).
Donovan, M. J. et al. A molecular signature of PCA3 and ERG exosomal RNA from non-DRE urine is predictive of initial prostate biopsy result. Prostate Cancer Prostatic Dis. 18, 370–375 (2015).
Tomlins, S. A. et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310, 644–648 (2005).
Mehra, R. et al. Comprehensive assessment of TMPRSS2 and ETS family gene aberrations in clinically localized prostate cancer. Mod. Pathol. 20, 538–544 (2007).
Berg, K. D. et al. The predictive value of ERG protein expression for development of castration-resistant prostate cancer in hormone-naive advanced prostate cancer treated with primary androgen deprivation therapy. Prostate 75, 1499–1509 (2015).
de Kok, J. B. et al. DD3(PCA3), a very sensitive and specific marker to detect prostate tumors. Cancer Res. 62, 2695–2698 (2002).
Hessels, D. et al. DD3(PCA3)-based molecular urine analysis for the diagnosis of prostate cancer. Eur. Urol. 44, 8–15; discussion 15–16 (2003).
Haese, A. et al. Clinical utility of the PCA3 urine assay in European men scheduled for repeat biopsy. Eur. Urol. 54, 1081–1088 (2008).
McKiernan, J. et al. A novel urine exosome gene expression assay to predict high-grade prostate cancer at initial biopsy. JAMA Oncol. 2, 882–889 (2016).
McKiernan, J. et al. A prospective adaptive utility trial to validate performance of a novel urine exosome gene expression assay to predict high-grade prostate cancer in patients with prostate-specific antigen 2-10ng/ml at initial biopsy. Eur. Urol. 74, 731–738 (2018).
Leyten, G. H. et al. Prospective multicentre evaluation of PCA3 and TMPRSS2-ERG gene fusions as diagnostic and prognostic urinary biomarkers for prostate cancer. Eur. Urol. 65, 534–542 (2014).
Tomlins, S. A. et al. Urine TMPRSS2:ERG plus PCA3 for individualized prostate cancer risk assessment. Eur. Urol. 70, 45–53 (2016).
Antonarakis, E. S. et al. Androgen receptor splice variant 7 and efficacy of taxane chemotherapy in patients with metastatic castration-resistant prostate cancer. OJAMA Oncol. 1, 582–591 (2015).
Del, Re,M. et al. The detection of androgen receptor splice variant 7 in plasma-derived exosomal RNA strongly predicts resistance to hormonal therapy in metastatic prostate cancer patients. Eur. Urol. 71, 680–687 (2017).
Woo, H. K. et al. Urine-based liquid biopsy: non-invasive and sensitive AR-V7 detection in urinary EVs from patients with prostate cancer. Lab. Chip 19, 87–97 (2018).
Cucchiara, V. et al. Genomic markers in prostate cancer decision making. Eur. Urol. 73, 572–582 (2018).
Zeuschner, P., Linxweiler, J. & Junker, K. Non-coding RNAs as biomarkers in liquid biopsies with a special emphasis on extracellular vesicles in urological malignancies. Expert. Rev. Mol. Diagn. https://doi.org/10.1080/14737159.2019.1665998 (2019).
Huang, X. et al. Exosomal miR-1290 and miR-375 as prognostic markers in castration-resistant prostate cancer. Eur. Urol. 67, 33–41 (2015).
Bhagirath, D. et al. MicroRNA-1246 is an exosomal biomarker for aggressive prostate cancer. Cancer Res. 78, 1833–1844 (2018).
Endzelins, E. et al. Detection of circulating miRNAs: comparative analysis of extracellular vesicle-incorporated miRNAs and cell-free miRNAs in whole plasma of prostate cancer patients. BMC Cancer 17, 730 (2017).
Foj, L. et al. Exosomal and non-exosomal urinary miRNAs in prostate cancer detection and prognosis. Prostate 77, 573–583 (2017).
Li, Z. et al. Exosomal microRNA-141 is upregulated in the serum of prostate cancer patients. Onco Targets Ther. 9, 139–148 (2016).
Bryant, R. J. et al. Changes in circulating microRNA levels associated with prostate cancer. Br. J. Cancer 106, 768–774 (2012).
Khan, S. et al. Plasma-derived exosomal survivin, a plausible biomarker for early detection of prostate cancer. PLOS ONE 7, e46737 (2012).
Bijnsdorp, I. V. et al. Exosomal ITGA3 interferes with non-cancerous prostate cell functions and is increased in urine exosomes of metastatic prostate cancer patients. J. Extracell. Vesicles 2, 22097 (2013).
Wang, Y. H. et al. Tumor-derived exosomal long noncoding RNAs as promising diagnostic biomarkers for prostate cancer. Cell Physiol. Biochem. 46, 532–545 (2018).
Clos-Garcia, M. et al. Metabolic alterations in urine extracellular vesicles are associated to prostate cancer pathogenesis and progression. J. Extracell. Vesicles 7, 1470442 (2018).
Skotland, T. et al. Molecular lipid species in urinary exosomes as potential prostate cancer biomarkers. Eur. J. Cancer 70, 122–132 (2017).
Shah, R., Patel, T. & Freedman, J. E. Circulating extracellular vesicles in human disease. N Engl. J. Med. 379, 958–966 (2018).
Namee, N. M. & O'Driscoll, L. Extracellular vesicles and anti-cancer drug resistance. Biochim. Biophys. Acta Rev. Cancer 1870, 123–136 (2018).
Webber, J., Yeung, V. & Clayton, A. Extracellular vesicles as modulators of the cancer microenvironment. Semin. Cell Dev. Biol. 40, 27–34 (2015).
Xu, H. et al. Exosome-transmitted PSMA3 and PSMA3-AS1 promote proteasome inhibitor resistance in multiple myeloma. Clin. Cancer Res. 25, 1923–1935 (2019).
Binenbaum, Y. et al. Transfer of miRNA in macrophage-derived exosomes induces drug resistance in pancreatic adenocarcinoma. Cancer Res. 78, 5287–5299 (2018).
Yang, L., Wu, X. H., Wang, D., Luo, C. L. & Chen, L. X. Bladder cancer cell-derived exosomes inhibit tumor cell apoptosis and induce cell proliferation in vitro. Mol. Med. Rep. 8, 1272–1278 (2013).
Beckham, C. J. et al. Bladder cancer exosomes contain EDIL-3/Del1 and facilitate cancer progression. J. Urol. 192, 583–592 (2014).
King, H. W., Michael, M. Z. & Gleadle, J. M. Hypoxic enhancement of exosome release by breast cancer cells. BMC Cancer 12, 421 (2012).
Kucharzewska, P. et al. Exosomes reflect the hypoxic status of glioma cells and mediate hypoxia-dependent activation of vascular cells during tumor development. Proc. Natl Acad. Sci. USA 110, 7312–7317 (2013).
Xue, M., Li, X., Li, Z. & Chen, W. Urothelial carcinoma associated 1 is a hypoxia-inducible factor-1alpha-targeted long noncoding RNA that enhances hypoxic bladder cancer cell proliferation, migration, and invasion. Tumour Biol. 35, 6901–6912 (2014).
Franzen, C. A. et al. Urothelial cells undergo epithelial-to-mesenchymal transition after exposure to muscle invasive bladder cancer exosomes. Oncogenesis 4, e163 (2015).
Ostenfeld, M. S. et al. Cellular disposal of miR23b by RAB27-dependent exosome release is linked to acquisition of metastatic properties. Cancer Res. 74, 5758–5771 (2014).
Goulet, C. R. et al. Exosomes induce fibroblast differentiation into cancer-associated fibroblasts through TGF beta signaling. Mol. Cancer Res. 16, 1196–1204 (2018).
Zhang, L. et al. The 786-0 renal cancer cell-derived exosomes promote angiogenesis by downregulating the expression of hepatocyte cell adhesion molecule. Mol. Med. Rep. 8, 272–276 (2013).
Jiang, X. L., Zhang, Y., Tan, B., Luo, C. L. & Wu, X. H. Renal tumor-derived exosomes inhibit hepaCAM expression of renal carcinoma cells in a p-AKT-dependent manner. Neoplasma 61, 416–423 (2014).
Horie, K. et al. Exosomes expressing carbonic anhydrase 9 promote angiogenesis. Biochem. Biophys. Res. Commun. 492, 356–361 (2017).
Logozzi, M. et al. Prostate cancer cells and exosomes in acidic condition show increased carbonic anhydrase IX expression and activity. J. Enzyme Inhib. Med. Chem. 34, 272–278 (2019).
Grange, C. et al. Microvesicles released from human renal cancer stem cells stimulate angiogenesis and formation of lung premetastatic niche. Cancer Res. 71, 5346–5356 (2011).
Jingushi, K. et al. Extracellular vesicles isolated from human renal cell carcinoma tissues disrupt vascular endothelial cell morphology via azurocidin. Int. J. Cancer 142, 607–617 (2018).
Lindoso, R. S., Collino, F. & Camussi, G. Extracellular vesicles derived from renal cancer stem cells induce a pro-tumorigenic phenotype in mesenchymal stromal cells. Oncotarget 6, 7959–7969 (2015).
Du, T. et al. Microvesicles derived from human Wharton's jelly mesenchymal stem cells promote human renal cancer cell growth and aggressiveness through induction of hepatocyte growth factor. PLOS ONE 9, e96836 (2014).
Xia, Y. et al. Negatively regulation of tumor-infiltrating NK cell in clear cell renal cell carcinoma patients through the exosomal pathway. Oncotarget 8, 37783–37795 (2017).
Diao, J. et al. Exosomal Hsp70 mediates immunosuppressive activity of the myeloid-derived suppressor cells via phosphorylation of Stat3. Med. Oncol. 32, 453 (2015).
Webber, J., Steadman, R., Mason, M. D., Tabi, Z. & Clayton, A. Cancer exosomes trigger fibroblast to myofibroblast differentiation. Cancer Res. 70, 9621–9630 (2010).
Chowdhury, R. et al. Cancer exosomes trigger mesenchymal stem cell differentiation into pro-angiogenic and pro-invasive myofibroblasts. Oncotarget 6, 715–731 (2015).
Lu, H. et al. Exosomal alphavbeta6 integrin is required for monocyte M2 polarization in prostate cancer. Matrix Biol. 70, 20–35 (2018).
Lazaro-Ibanez, E. et al. Metastatic state of parent cells influences the uptake and functionality of prostate cancer cell-derived extracellular vesicles. J. Extracell. Vesicles 6, 1354645 (2017).
Singh, A. et al. Exosome-mediated transfer of alphavbeta3 integrin from tumorigenic to nontumorigenic cells promotes a migratory phenotype. Mol. Cancer Res. 14, 1136–1146 (2016).
Sanchez, C. A. et al. Exosomes from bulk and stem cells from human prostate cancer have a differential microRNA content that contributes cooperatively over local and pre-metastatic niche. Oncotarget 7, 3993–4008 (2016).
Bilusic, M., Madan, R. A. & Gulley, J. L. Immunotherapy of prostate cancer: facts and hopes. Clin. Cancer Res. 23, 6764–6770 (2017).
Gao, J. et al. VISTA is an inhibitory immune checkpoint that is increased after ipilimumab therapy in patients with prostate cancer. Nat. Med. 23, 551–555 (2017).
Lu, X. et al. Effective combinatorial immunotherapy for castration-resistant prostate cancer. Nature 543, 728–732 (2017).
Salimu, J. et al. Dominant immunosuppression of dendritic cell function by prostate-cancer-derived exosomes. J. Extracell. Vesicles 6, 1368823 (2017).
Clayton, A., Al-Taei, S., Webber, J., Mason, M. D. & Tabi, Z. Cancer exosomes express CD39 and CD73, which suppress T cells through adenosine production. J. Immunol. 187, 676–683 (2011).
Clayton, A., Mitchell, J. P., Court, J., Mason, M. D. & Tabi, Z. Human tumor-derived exosomes selectively impair lymphocyte responses to interleukin-2. Cancer Res. 67, 7458–7466 (2007).
Clayton, A. et al. Human tumor-derived exosomes down-modulate NKG2D expression. J. Immunol. 180, 7249–7258 (2008).
Lundholm, M. et al. Prostate tumor-derived exosomes down-regulate NKG2D expression on natural killer cells and CD8+ T cells: mechanism of immune evasion. PLOS ONE 9, e108925 (2014).
Hashimoto, K. et al. Cancer-secreted hsa-miR-940 induces an osteoblastic phenotype in the bone metastatic microenvironment via targeting ARHGAP1 and FAM134A. Proc. Natl Acad. Sci. USA 115, 2204–2209 (2018).
Ye, Y. et al. Exosomal miR-141-3p regulates osteoblast activity to promote the osteoblastic metastasis of prostate cancer. Oncotarget 8, 94834–94849 (2017).
Karlsson, T., Lundholm, M., Widmark, A. & Persson, E. Tumor cell-derived exosomes from the prostate cancer cell line TRAMP-C1 impair osteoclast formation and differentiation. PLOS ONE 11, e0166284 (2016).
Li, J. et al. Exosome-derived microRNAs contribute to prostate cancer chemoresistance. Int. J. Oncol. 49, 838–846 (2016).
Ramteke, A. et al. Exosomes secreted under hypoxia enhance invasiveness and stemness of prostate cancer cells by targeting adherens junction molecules. Mol. Carcinog. 54, 554–565 (2015).
Kumar, D., Gupta, D., Shankar, S. & Srivastava, R. K. Biomolecular characterization of exosomes released from cancer stem cells: possible implications for biomarker and treatment of cancer. Oncotarget 6, 3280–3291 (2015).
Hosseini-Beheshti, E. et al. Exosomes confer pro-survival signals to alter the phenotype of prostate cells in their surrounding environment. Oncotarget 7, 14639–14658 (2016).
Kharmate, G., Hosseini-Beheshti, E., Caradec, J., Chin, M. Y. & Tomlinson Guns, E. S. Epidermal growth factor receptor in prostate cancer derived exosomes. PLOS ONE 11, e0154967 (2016).
Corcoran, C. et al. Docetaxel-resistance in prostate cancer: evaluating associated phenotypic changes and potential for resistance transfer via exosomes. PLOS ONE 7, e50999 (2012).
Kharaziha, P. et al. Molecular profiling of prostate cancer derived exosomes may reveal a predictive signature for response to docetaxel. Oncotarget 6, 21740–21754 (2015).
Kawakami, K. et al. Integrin beta4 and vinculin contained in exosomes are potential markers for progression of prostate cancer associated with taxane-resistance. Int. J. Oncol. 47, 384–390 (2015).
Kato, T. et al. Serum exosomal P-glycoprotein is a potential marker to diagnose docetaxel resistance and select a taxoid for patients with prostate cancer. Urol. Oncol. 33, 385.e15–e20 (2015).
Vagner, T. et al. Large extracellular vesicles carry most of the tumour DNA circulating in prostate cancer patient plasma. J. Extracell. Vesicles 7, 1505403 (2018).
Becker, A. et al. Extracellular vesicles in cancer: cell-to-cell mediators of metastasis. Cancer Cell 30, 836–848 (2016).
Schwarzenbach, H., da Silva, A. M., Calin, G. & Pantel, K. Data normalization strategies for microRNA quantification. Clin. Chem. 61, 1333–1342 (2015).
Anand, S., Samuel, M., Kumar, S. & Mathivanan, S. Ticket to a bubble ride: cargo sorting into exosomes and extracellular vesicles. Biochim. Biophys. Acta Proteins Proteom. 1867, 140203 (2019).
Villarroya-Beltri, C., Baixauli, F., Gutierrez-Vazquez, C., Sanchez-Madrid, F. & Mittelbrunn, M. Sorting it out: regulation of exosome loading. Semin. Cancer Biol. 28, 3–13 (2014).
Hisey, C. L., Dorayappan, K. D. P., Cohn, D. E., Selvendiran, K. & Hansford, D. J. Microfluidic affinity separation chip for selective capture and release of label-free ovarian cancer exosomes. Lab. Chip 18, 3144–3153 (2018).
Kanwar, S. S., Dunlay, C. J., Simeone, D. M. & Nagrath, S. Microfluidic device (ExoChip) for on-chip isolation, quantification and characterization of circulating exosomes. Lab. Chip 14, 1891–1900 (2014).
Brett, S. I. et al. Immunoaffinity based methods are superior to kits for purification of prostate derived extracellular vesicles from plasma samples. Prostate 77, 1335–1343 (2017).
Biggs, C. N. et al. Prostate extracellular vesicles in patient plasma as a liquid biopsy platform for prostate cancer using nanoscale flow cytometry. Oncotarget 7, 8839–8849 (2016).
Mizutani, K. et al. Isolation of prostate cancer-related exosomes. Anticancer Res. 34, 3419–3423 (2014).
Welton, J. L. et al. Proteomics analysis of bladder cancer exosomes. Mol. Cell Proteom. 9, 1324–1338 (2010).
Jin, D. et al. ExoAPP: exosome-oriented, aptamer nanoprobe-enabled surface proteins profiling and detection. Anal. Chem. 90, 14402–14411 (2018).
Castillo, J. et al. Surfaceome profiling enables isolation of cancer-specific exosomal cargo in liquid biopsies from pancreatic cancer patients. Ann. Oncol. 29, 223–229 (2018).
Das, C. K. et al. Exosome as a novel shuttle for delivery of therapeutics across biological barriers. Mol. Pharm. 16, 24–40 (2018).
Hood, J. L. Post isolation modification of exosomes for nanomedicine applications. Nanomedicine 11, 1745–1756 (2016).
Ferguson, S. W. & Nguyen, J. Exosomes as therapeutics: the implications of molecular composition and exosomal heterogeneity. J. Control. Release 228, 179–190 (2016).
Dijkstra, S. et al. Prostate cancer biomarker profiles in urinary sediments and exosomes. J. Urol. 191, 1132–1138 (2014).
Nilsson, J. et al. Prostate cancer-derived urine exosomes: a novel approach to biomarkers for prostate cancer. Br. J. Cancer 100, 1603–1607 (2009).
Royo, F. et al. Transcriptomic profiling of urine extracellular vesicles reveals alterations of CDH3 in prostate cancer. Oncotarget 7, 6835–6846 (2016).
Xu, Y. et al. MiR-145 detection in urinary extracellular vesicles increase diagnostic efficiency of prostate cancer based on hydrostatic filtration dialysis method. Prostate 77, 1167–1175 (2017).
Koppers-Lalic, D. et al. Noninvasive prostate cancer detection by measuring miRNA variants (isomiRs) in urine extracellular vesicles. Oncotarget 7, 22566–22578 (2016).
Wani, S., Kaul, D., Mavuduru, R. S., Kakkar, N. & Bhatia, A. Urinary-exosomal miR-2909: a novel pathognomonic trait of prostate cancer severity. J. Biotechnol. 259, 135–139 (2017).
Rodriguez, M. et al. Identification of non-invasive miRNAs biomarkers for prostate cancer by deep sequencing analysis of urinary exosomes. Mol. Cancer 16, 156 (2017).
Hessvik, N. P., Sandvig, K. & Llorente, A. Exosomal miRNAs as biomarkers for prostate cancer. Front. Genet. 4, 36 (2013).
Moltzahn, F. et al. Microfluidic-based multiplex qRT-PCR identifies diagnostic and prognostic microRNA signatures in the sera of prostate cancer patients. Cancer Res. 71, 550–560 (2011).
Lodes, M. J. et al. Detection of cancer with serum miRNAs on an oligonucleotide microarray. PLOS ONE 4, e6229 (2009).
Duijvesz, D. et al. Immuno-based detection of extracellular vesicles in urine as diagnostic marker for prostate cancer. Int. J. Cancer 137, 2869–2878 (2015).
Overbye, A. et al. Identification of prostate cancer biomarkers in urinary exosomes. Oncotarget 6, 30357–30376 (2015).
Duijvesz, D. et al. Proteomic profiling of exosomes leads to the identification of novel biomarkers for prostate cancer. PLOS ONE 8, e82589 (2013).
Li, S. et al. Exosomal ephrinA2 derived from serum as a potential biomarker for prostate cancer. J. Cancer 9, 2659–2665 (2018).
Logozzi, M. et al. Increased PSA expression on prostate cancer exosomes in in vitro condition and in cancer patients. Cancer Lett. 403, 318–329 (2017).
Kawakami, K. et al. Gamma-glutamyltransferase activity in exosomes as a potential marker for prostate cancer. BMC Cancer 17, 316 (2017).
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, made substantial contributions to the discussion of content, and wrote and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Urology thanks H. Leong, T. Ochiya and A. Zijlstra for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Linxweiler, J., Junker, K. Extracellular vesicles in urological malignancies: an update. Nat Rev Urol 17, 11–27 (2020). https://doi.org/10.1038/s41585-019-0261-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41585-019-0261-8
This article is cited by
-
Targeting cancer-derived extracellular vesicles by combining CD147 inhibition with tissue factor pathway inhibitor for the management of urothelial cancer cells
Cell Communication and Signaling (2024)
-
Diagnostic liquid biopsy biomarkers in renal cell cancer
Nature Reviews Urology (2024)
-
Tumor promoting effect of spheroids in an orthotopic prostate cancer mouse model
Scientific Reports (2024)
-
Current knowledge on the role of extracellular vesicles in endometrial receptivity
European Journal of Medical Research (2023)
-
Extracellular vesicle analysis
Nature Reviews Methods Primers (2023)