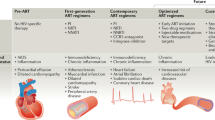
Abstract
The HIV epidemic has devastated millions of people globally, with approximately 40 million deaths since its start. The availability of antiretroviral therapy (ART) has transformed the prognosis of millions of individuals infected with HIV such that a diagnosis of HIV infection no longer automatically confers death. However, morbidity and mortality remain substantial among people living with HIV. HIV can directly infect the kidney to cause HIV-associated nephropathy (HIVAN) — a disease characterized by podocyte and tubular damage and associated with an increased risk of kidney failure. The reports of HIVAN occurring primarily in those of African ancestry led to the discovery of its association with APOL1 risk alleles. The advent of ART has led to a substantial decrease in the prevalence of HIVAN; however, reports have emerged of an increase in the prevalence of other kidney pathology, such as focal segmental glomerulosclerosis and pathological conditions associated with co-morbidities of ageing, such as hypertension and diabetes mellitus. Early initiation of ART also results in a longer cumulative exposure to medications, increasing the likelihood of nephrotoxicity. A substantial body of literature supports the use of kidney transplantation in people living with HIV, demonstrating significant survival benefits compared with that of people undergoing chronic dialysis, and similar long-term allograft and patient survival compared with that of HIV-negative kidney transplant recipients.
Key points
-
The widespread use of antiretroviral therapy has led to a change in the histological pattern of kidney disease in people living with HIV (PLWH) away from HIV-associated nephropathy and towards comorbid diseases of ageing and antiretroviral therapy-related nephrotoxicity.
-
Biopsy remains crucial for the appropriate management of an array of kidney diseases in PLWH; the current classification of kidney histology is based on the major tissue compartments affected, and is aimed at reducing heterogeneity in the biopsy definitions of HIV-related kidney disease and encouraging awareness of potential secondary causes of disease.
-
The new race-neutral creatinine-based equation for estimating the glomerular filtration rate identified more Black individuals with a lower estimated glomerular filtration rate than previously recognized, and exposed a subgroup of Black individuals living with HIV who are at an increased risk of chronic kidney disease progression.
-
The presence of APOL1 risk alleles influences the pattern of kidney disease in PLWH, its response to therapy and its outcomes; clinical trials are investigating therapeutic options to reduce APOL1 protein and/or its activity, which may ameliorate its potential harm.
-
Kidney transplantation in PLWH from both HIV-negative and HIV-positive donors is highly successful, and the donation of organs from PLWH may be an option for increasing the number of organs available for transplantation; however, this approach requires further evaluation before being adopted as standard clinical practice.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
UNAIDS. In Danger: UNAIDS Global AIDS Update 2022 https://www.unaids.org/en/resources/documents/2022/in-danger-global-aids-update (Joint United Nations Programme on HIV/AIDS, 2022).
Rao, T. K. et al. Associated focal and segmental glomerulosclerosis in the acquired immunodeficiency syndrome. N. Engl. J. Med. 310, 669–673 (1984).
D’Agati, V., Suh, J. I., Carbone, L., Cheng, J. T. & Appel, G. Pathology of HIV-associated nephropathy: a detailed morphologic and comparative study. Kidney Int. 35, 1358–1370 (1989).
Kudose, S. et al. The spectrum of kidney biopsy findings in HIV-infected patients in the modern era. Kidney Int. 97, 1006–1016 (2020).
Diana, N. E. et al. Clinicopathological correlation of kidney disease in HIV infection pre- and post-ART rollout. PLoS One 17, e0269260 (2022).
Achhra, A. C. et al. Impact of early versus deferred antiretroviral therapy of estimated glomerular filtration rate in HIV-positive individuals in the START trial. Int. J. Antimicrob. Agents 50, 453–460 (2017).
Stock, P. G. et al. Outcomes of kidney transplantation in HIV-infected recipients. N. Engl. J. Med. 18, 363 (2010). Erratum in: N Engl J Med. 364, 1082 (2011).
Muller, E. et al. Kidney transplantation in HIV-positive patients: current practice and management strategies. Transplantation 105, 1492–1501 (2021).
Kimmel, P. L., Barisoni, L. & Kopp, J. B. Pathogenesis and treatment of HIV-associated renal diseases: lessons from clinical and animal studies, molecular pathologic correlations, and genetic investigations. Ann. Intern. Med. 139, 214–226 (2003).
Lescure, F. X. et al. HIV-associated kidney glomerular diseases: changes with time and HAART. Nephrol. Dial. Transpl. 27, 2349–2355 (2012).
Swanepoel, C. R. et al. Kidney disease in the setting of HIV infection: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) controversies conference. Kidney Int. 93, 545–559 (2018).
Wearne, N. et al. The evolving spectrum of kidney histology in HIV positive patients in South Africa. Kidney Int. Rep. 8, 1087–1096 (2023).
Mallipattu, S. K., Salem, F. & Wyatt, C. M. The changing epidemiology of HIV-related chronic kidney disease in the era of antiretroviral therapy. Kidney Int. 86, 259–265 (2014).
Wyatt, C. M., Klotman, P. E. & DÁgati, V. D. HIV-associated nephropathy: clinical presentation, pathology and epidemiology in the era of antiretroviral therapy. Semin. Nephrol. 28, 513–522 (2008).
Alfano, G. et al. Kidney disease in HIV infection. J. Clin. Med. 8, 1254 (2019).
Remark, R. et al. In-depth tissue profiling using multiplexed immunohistochemical consecutive staining on single slide. Sci. Immunol. 1, aaf6925 (2016).
Xu, G. J. et al. Viral immunology. Comprehensive serological profiling of human populations using a synthetic human virome. Science 348, aaa0698 (2015).
Kimmel, P. L. et al. Brief report: idiotypic IgA nephropathy in patients with human immunodeficiency virus infection. N. Engl. J. Med. 327, 702–706 (1992).
Kimmel, P. L. et al. HIV-associated immune-mediated renal disease. Kidney Int. 44, 1327–1340 (1993).
Gerntholtz, T. E., Goetsch, S. J. & Katz, I. HIV-related nephropathy: a South African perspective. Kidney Int. 69, 1885–1891 (2006).
Vermeulen, A. et al. Patterns of renal disease: a 30-year renal biopsy study at Chris Hani Baragwanath Academic Hospital, Soweto, Johannesburg, South Africa. S Afr. Med. J. 109, 486–492 (2019).
Nair, R., Walker, P. D. & Is, I. G. A nephropathy the commonest primary glomerulopathy among your adults in the USA? Kidney Int. 69, 1455–1458 (2006).
Fabrizi, F. et al. Hepatitis C virus increases the risk of kidney disease among HIV-positive patients: systematic review and meta-analysis. J. Med. Virol. 88, 487–497 (2016).
Mocroft, A. et al. Hepatitis B and co-infection are independent predictors of progressive kidney disease in HIV-positive, antiretroviral-treated adults. PLoS One 7, e40245 (2012).
Davidson, B. et al. Granulomatous interstitial nephritis on renal biopsy in human immunodeficiency virus positive patients: prevalence and causes in Cape Town, South Africa. Nephrology 24, 681–688 (2019).
Kearns, A., Gordon, J., Burdo, T. H. & Qin, X. HIV-1-associated atherosclerosis: unraveling the missing link. J. Am. Coll. Cardiol. 69, 3084–3098 (2017).
Ipp, H. & Zemlin, A. The paradox of the immune response in HIV infection: when inflammation becomes harmful. Clin. Chim. Acta 416, 96–99 (2013).
Kelesidis, T., Kendall, M. A., Yang, O. O., Hodis, H. N. & Currier, J. S. Biomarkers of microbial translocation and macrophage activation: association with progression of subclinical atherosclerosis in HIV-1 infection. J. Infect. Dis. 206, 1558–1567 (2012).
Alcaide, M. L. et al. Immune activation in HIV-infected aging women on antiretrovirals-implications for age-associated comorbidities: a cross-sectional pilot study. PLoS One 8, e63804 (2013).
Ma, R., Yang, L., Niu, F. & Buch, S. HIV Tat-mediated induction of human brain microvascular endothelial cell apoptosis involves endoplasmic reticulum stress and mitochondrial dysfunction. Mol. Neurobiol. 53, 132–142 (2016).
Guo, H., Gao, J., Taxman, D. J., Ting, J. P. & Su, L. HIV-1 infection induces interleukin-1β production via TLR8 protein-dependent and NLRP3 inflammasome mechanisms in human monocytes. J. Biol. Chem. 289, 21716–21726 (2014).
Hernandez, J. C., Latz, E. & Urcuqui-Inchima, S. HIV-1 induces the first signal to activate the NLRP3 inflammasome in monocyte-derived macrophages. Intervirology 57, 36–42 (2014).
Brown, T. T. et al. Antiretroviral therapy and the prevalence and incidence of diabetes mellitus in the multicenter AIDS cohort study. Arch. Intern. Med. 165, 1179–1184 (2005).
Heron, J. E. et al. The prevalence and risk of non-infectious comorbidities in HIV infected and non-HIV infected men attending general practice in Australia. PLoS One 14, e0223224 (2019).
Høgh, J. et al. HIV infection as associated with type 2 diabetes mellitus. J. Acquir. Immune Defic. Syndr. 88, e32–e35 (2021).
Noubissi, E. C., Katte, J. C. & Sobngwi, E. Diabetes and HIV. Curr. Diab Rep. 18, 125 (2018).
Mallipattu, S. K. et al. Expression of HIV transgene aggravates kidney injury in diabetic mice. Kidney Int. 83, 626–634 (2013).
Gooden, T. E. et al. A matched cohort study investigating premature, accentuated, and accelerated aging in people living with HIV. HIV Med. 24, 640–647 (2023).
Cohen, S. & Kimmel, P. Renal biopsy is necessary for the diagnosis of HIV-associated renal diseases. Nat. Rev. Nephrol. 5, 22–23 (2009).
Soler-García, A. A., Rakhmanina, N. Y., Mattison, P. C. & Ray, P. E. A urinary biomarker profile for children with HIV-associated renal diseases. Kidney Int. 76, 207–214 (2009).
Paragas, N. et al. Urinary NGAL marks cystic disease in HIV-associated nephropathy. J. Am. Soc. Nephrol. 20, 1687–1692 (2009).
Sola-Del Valle, D. A. et al. Urinary NGAL is a useful clinical biomarker of HIV-associated nephropathy. Nephrol. Dial. Transpl. 26, 2387–2390 (2011).
Naicker, S. et al. Profiling biomarkers in HIV glomerular disease — potential for the non-invasive diagnosis of HIVAN? Int. J. Nephrol. Renovasc Dis. 14, 427–440 (2021).
Griffin, B. R., Faubel, S. & Edelstein, C. L. Biomarkers of drug-induced kidney toxicity. Ther. Drug. Monit. 41, 213–226 (2019).
Fiseha, T. & Gebreweld, A. Urinary markers of tubular injury in HIV-infected patients. Biochem. Res. Int. 2016, 1501785 (2016).
Shlipak, M. G. et al. Urinary markers of kidney injury and kidney function decline in HIV-infected women. J. Acquir. Immune Defic. Syndr. 61, 565–573 (2012).
Inker, L. A. et al. Performance of creatinine and cystatin C GFR estimating equations in an HIV population on antiretrovirals. J. Acquir. Immune Defic. Syndr. 61, 302–309 (2012).
Gagneux-Brunon, A. et al. Performance of creatinine and cystatin C-based glomerular filtration rate estimation equations in a European HIV-positive cohort. AIDS 27, 1573–1581 (2013).
European AIDS Clinical Society guidelines Oct 2021. https://www.eacsociety.org/guidelines/eacs-guidelines/ (accessed January 2023).
Levey, A. S. et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med 150, 604–612 (2009).
Degado, C. et al. Reassessing the inclusion of race in diagnosing kidney diseases: an interim report from the NKF-ASN Task Force. Am. J. Kidney Dis. 78, 103–115 (2021).
Degado, C. et al. A unifying approach for GFR estimation: recommendations of the NKF-ASN task force on reassessing the inclusion of race in diagnosing kidney disease. Am. J. Kidney Dis. 79, 268–288.e1 (2022).
Muiru, A. N. et al. Effect of adopting the new race-free 2021 chronic kidney disease epidemiology collaboration estimated glomerular filtration rate creatinine equation on racial differences in kidney disease progression among people with human immunodeficiency virus: an observational study. Clin. Infect. Dis. 76, 461–468 (2023).
Fabian, J. et al. Measurement of kidney function in Malawi, South Africa, and Uganda: a multicenter cohort study. Lancet Glob. Health 10, e1159–e1169 (2022).
Inker, L. A. et al. New creatinine-and cystatin C–based equations to estimate GFR without race. N. Engl. J. Med. 385, 1737–1749 (2021).
Bhasin, B. et al. Correction: HIV viremia and T-cell activation differentially affect the performance of glomerular filtration rate equations based on creatinine and cystatin C. PLoS One 14, e0215630 (2019).
Bruggeman, L. A. et al. Renal epithelium is a previously unrecognized site of HIV-1 infection. J. Am. Soc. Nephrol. 11, 2079–2087 (2000).
Marras, D. et al. Replication and compartmentalization of HIV-1 in kidney epithelium of patients with HIV-associated nephropathy. Nat. Med. 8, 522–526 (2002).
Payne, E. H., Ramalingam, D., Fox, D. T. & Klotman, M. E. Polyploidy and mitotic cell death are two distinct HIV-1 Vpr-driven outcomes in renal tubule epithelial cells. J. Virol. 92, e01718–17 (2018).
Bruggeman, L. A. et al. Nephropathy in human immunodeficiency virus-1 transgenic mice is due to renal transgene expression. J. Clin. Invest. 100, 84–92 (1997).
Husain, M. et al. HIV-1 Nef induces proliferation and anchorage-independent growth in podocytes. J. Am. Soc. Nephrol. 13, 1806–1815 (2002).
Dickie, P. et al. HIV-associated nephropathy in transgenic mice expressing HIV-1 genes. Virology 185, 109–119 (1991).
Zuo, Y. et al. HIV-1 genes vpr and nef synergistically damage podocytes, leading to glomerulosclerosis. J. Am. Soc. Nephrol. 17, 2832–2843 (2006).
Conaldi, P. G. et al. Human immunodeficiency virus-1 tat induces hyperproliferation and dysregulation of renal glomerular epithelial cells. Am. J. Pathol. 61, 53–61 (2002).
Winston, J. A. et al. Nephropathy and establishment of a renal reservoir of HIV type 1 during primary infection. N. Engl. J. Med. 344, 1979–1984 (2001).
Pierson, T., McArthur, J. & Siliciano, R. F. Reservoirs for HIV-1: mechanisms for viral persistence in the presence of antiviral immune responses and antiretroviral therapy. Annu. Rev. Immunol. 18, 665–708 (2000).
Cohen, A. H., Sun, N. C., Shapshak, P. & Imagawa, D. T. Demonstration of human immunodeficiency virus in renal epithelium in HIV-associated nephropathy. Mod. Pathol. 2, 125–128 (1989).
Blasi, M. et al. Identification of HIV-1 genitourinary tract compartmentalization by analyzing the env gene sequences in urine. AIDS 29, 1651–1657 (2015).
Mzingwane, M. L. et al. Detection and molecular characterization of urinary tract HIV-1 populations. Ann. Clin. Microbiol. Antimicrob. 18, 27 (2019).
Canaud, G. et al. The kidney as a reservoir for HIV-1 after renal transplantation. J. Am. Soc. Nephrol. 25, 407–419 (2014).
Blasi, M. et al. Detection of donor’s HIV strain in HIV-positive kidney transplant recipient. N. Engl. J. Med. 382, 195–197 (2020).
Dufour, C. et al. Near full-length HIV sequencing in multiple tissues collected postmortem reveals shared clonal expansions across distinct reservoirs during ART. Cell Rep. 42, 113053 (2023).
Genovese, G. et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 329, 841–845 (2010).
Tzur, S. et al. Missense mutations in the APOL1 gene are highly associated with end stage kidney disease risk previously attributed to the MYH9 gene. Hum. Genet. 128, 345–350 (2010).
Daneshpajouhnejad, P., Kopp, J. B., Winkler, C. A. & Rosenberg, A. Z. The evolving story of apolipoprotein L1 nephropathy: the end of the beginning. Nat. Rev. Nephrol. 18, 307–320 (2022).
Dummer, P. D. et al. APOL1 kidney disease risk variants: an evolving landscape. Semin. Nephrol. 35, 222–236 (2015).
Kopp, J. B. et al. APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J. Am. Soc. Nephrol. 22, 2129–2137 (2011).
Kasembeli, A. N. et al. APOL1 risk variants are strongly associated with HIV-associated nephropathy in black South Africans. J. Am. Soc. Nephrol. 26, 2882–2890 (2015).
Atta, M. G. et al. Association of APOL1 genotype with renal histology among black HIV-positive patients undergoing kidney biopsy. Clin. J. Am. Soc. Nephrol. 11, 262–270 (2016).
Abdu, A. et al. High risk APOL1 genotypes and kidney disease among treatment naïve HIV patients at Kano, Nigeria. PLoS One 17, e0275949 (2022).
Hung, R. K. Y. et al. Genetic variants of APOL1 are major determinants of kidney failure in people of African ancestry with HIV. Kidney Int. Rep. 7, 786–796 (2022).
Waziri, B., Raji, Y. E., Ekrikpo, U. E. & Naicker, S. Apolipoprotein L1 gene variants and kidney disease in patients with HIV: a systematic review and meta-analysis. J. Nephrol. 36, 1119–1134 (2023).
Fine, D. M. et al. APOL1 risk variants predict histopathology and progression to ESRD in HIV-related kidney disease. J. Am. Soc. Nephrol. 23, 343–350 (2012).
Purswani et al. Pediatric HIVAIDS Cohort Study. Brief report: APOL1 renal risk variants are associated with chronic kidney disease in children and youth with perinatal HIV infection. J. Acquir. Immune Defic. Syndr. 73, 63–68 (2016).
Ekulu, P. M. et al. APOL1 risk genotypes are associated with early kidney damage in children in Sub-Saharan Africa. Kidney Int. Rep. 4, 930–938 (2019).
Behar, D. M. et al. Absence of APOL1 risk variants protects against HIV-associated nephropathy in the Ethiopian population. Am. J. Nephrol. 34, 452–459 (2011).
Rosset, S., Tzur, S., Behar, D. M., Wasser, W. G. & Skorecki, K. The population genetics of chronic kidney disease: insights from the MYH9-APOL1 locus. Nat. Rev. Nephrol. 7, 313–326 (2011).
Johnstone, D. B. et al. APOL1 null alleles from a rural village in India do not correlate with glomerulosclerosis. PLoS One 7, e51546 (2012).
Friedman, D. J. A brief history of APOL1: a gene evolving. Semin. Nephrol. 37, 508–513 (2017).
Olabisi, O. A. et al. APOL1 kidney disease risk variants cause cytotoxicity by depleting cellular potassium and inducing stress-activated protein kinases. Proc. Natl Acad. Sci. USA 113, 830–837 (2016).
Rednor, S. J. & Ross, M. J. Molecular mechanisms of injury in HIV-associated nephropathy. Front. Med. 5, 177 (2018).
Beckerman, P. et al. Transgenic expression of human APOL1 risk variants in podocytes induces kidney disease in mice. Nat. Med. 23, 429–438 (2017).
McCarthy, G. M. et al. Recessive, gain-of-function toxicity in an APOL1 BAC transgenic mouse model mirrors human APOL1 kidney disease. Dis. Model. Mech. 14, dmm048952 (2021).
Kalayjian, R. C. The treatment of HIV-associated nephropathy. Adv. Chronic Kidney Dis. 17, 59–71 (2010).
Estrella, M. M. et al. The association between APOL1 risk alleles and longitudinal kidney function differs by HIV viral suppression status. Clin. Infect. Dis. 60, 646–652 (2015).
Freedman, B. I. & Skorecki, K. Gene-gene and gene-environment interactions in apolipoprotein L1 gene-associated nephropathy. Clin. J. Am. Soc. Nephrol. 9, 2006–2013 (2014).
Danwang, C., Noubiap, J. J., Robert, A. & Yombi, J. C. Outcomes of patients with HIV and COVID-19 co-infection: a systematic review and meta-analysis. AIDS Res. Ther. 19, 3 (2022).
Bertagnolio, S. et al. Clinical features of, and risk factors for, severe or fatal COVID-19 among people living with HIV admitted to hospital: analysis of data from the WHO Global Clinical Platform of COVID-19. Lancet HIV. 9, e486–e495 (2022).
May, R. M. et al. A multi-center retrospective cohort study defines the spectrum of kidney pathology in Coronavirus 2019 Disease (COVID-19). Kidney Int. 100, 1303–1315 (2021).
Velez, J. C., Caza, T. & Larsen, C. COVAN is the new HIVAN: the re-emergence of collapsing glomerulopathy with COVID-19. Nat. Rev. Nephrol. 16, 565–567 (2020).
Shetty, A. A. et al. COVID-19-associated glomerular disease. J. Am. Soc. Nephrol. 32, 33 (2021).
Wu, H. L. et al. Acute kidney injury and collapsing glomerulopathy associated with COVID-19 and APOL1 high risk genotype. J. Am. Soc. Nephrol. 31, 1688–1695 (2020).
Giannini, G. et al. Renal prognosis of COVID-19 associated nephropathy. Kidney Int. Rep. 7, 2722–2725 (2022).
Kudose, S. et al. Longitudinal outcomes of COVID-19-associated collapsing glomerulopathy and other podocytopathies. J. Am. Soc. Nephrol. 32, 2958–2969 (2021).
World Health Organization. Consolidated guidelines on HIV prevention, testing, treatment, service delivery and monitoring: recommendations for a public health approach. https://www.who.int/publications/i/item/9789240031593 (World Health Organization, 2021; accessed January 2023).
Acquah, R., Graham, H. & Winter, A. Quantifying polypharmacy in a large HIV-infected cohort. HIV Med. 16, 583–584 (2015).
Bastida, C. et al. Polypharmacy and potential drug-drug interactions in an HIV-infected elderly population. Farm. Hosp. 41, 618–624 (2017).
Cohen, S. D., Kopp, J. B. & Kimmel, P. L. Kidney diseases associated with human immunodeficiency virus infection. N. Engl. J. Med. 14, 377 (2017). Erratum in: N. Engl. J. Med. 378, 1657 (2018).
Hamzah, L. et al. Treatment-limiting renal tubulopathy in patients treated with tenofovir disoproxil fumarate. J. Infect. 74, 492–500 (2017).
Irizarry-Alvarado, J. M., Dwyer, J. P., Brumble, L. M., Alvarez, S. & Mensez, J. C. Proximal tubular dysfunction associated with tenofovir and didanosine causing Fanconi syndrome and diabetes insipidus: a report of 3 cases. AIDS Read. 19, 114–121 (2009).
Iwata, K., Nagata, M., Watanabe, S. & Nishi, S. Distal renal tubular acidosis without renal impairment after use of tenofovir: a case report. BMC Pharm. Toxicol. 17, 52 (2016).
Fernandez-Fernandez, B. et al. Tenofovir nephrotoxicity: 2011 update. AIDS Res. Treat. 2011, 354908 (2011).
Mocroft, A. et al. Cumulative and current exposure to potentially nephrotoxic antiretrovirals and development of chronic kidney disease in HIV-positive individuals with a normal baseline estimated glomerular filtration rate: a prospective international Cohort Study. Lancet HIV. 3, e23–e32 (2016).
Cooper, R. D. et al. Systematic review and meta-analysis: renal safety of tenofovir disoproxil fumarate in HIV-infected patients. Clin. Infect. Dis. 51, 496–505 (2010).
Stray, K. M. et al. Tenofovir alafenamide (TAF) does not deplete mitochondrial DNA in human T-cell lines at intracellular concentrations exceeding clinically relevant drug exposures. Antivir. Res. 140, 116–120 (2017).
Novick, T. K. et al. Tenofovir alafenamide nephrotoxicity in an HIV-positive patient: a case report. Medicine 96, e8046 (2017).
Bahr, N. C. & Yarlagadda, S. G. Fanconi syndrome and tenofovir alafenamide: a case report. Ann. Intern. Med. 170, 814 (2019).
Post, F. A. et al. Brief report: switching to tenofovir alafenamide, coformulated with elvitegravir, cobicistat, and emtricitabine, in HIV-infected adults with renal impairment: 96-week results from a single-arm, multicenter, open-label phase 3 study. J. Acquir. Immune Defic. Syndr. 74, 180 (2017).
Pilkington, V. et al. Tenofovir alafenamide vs. tenofovir disoproxil fumarate: an updated meta-analysis of 14894 patients across 14 trials. AIDS 34, 2259–2268 (2020).
Burgos, J., Ribera, E. & Falco, V. Antiretroviral therapy in advanced HIV disease: which is the best regimen? AIDS Rev. 20, 3–13 (2018).
Achhra, A. C., Nugent, M., Mocroft, A., Ryom, L. & Wyatt, C. M. Chronic kidney disease and antiretroviral therapy in HIV-positive individuals: recent developments. Curr. HIV/AIDS Rep. 13, 149–157 (2016).
Chughlay, M. F. et al. Acute interstitial nephritis caused by lopinavir/ritonavir in a surgeon receiving antiretroviral postexposure prophylaxis. AIDS 29, 503–504 (2015).
Hamada, Y. et al. High incidence of renal stones among HIV-infected patients on ritonavir-boosted atazanavir than in those receiving other protease inhibitor-containing antiretroviral therapy. Clin. Infect. Dis. 55, 1262–1269 (2012).
Schmid, S. et al. Acute interstitial nephritis of HIV positive patients under atazanavir and tenofovir therapy in a retrospective analysis of kidney biopsies. Virchows Arch. 450, 665–670 (2007).
Jose, S. et al. Improved kidney function in patients who switch their protease inhibitor from atazanavir or lopinavir to darunavir. AIDS 31, 485–492 (2017).
Gupta, S. K., Mi, D., Moe, S. M., Dubé, M. P. & Liu, Z. Effects of switching from efavirenz to raltegravir on endothelial function, bone mineral metabolism, inflammation, and renal function: a randomized, controlled trial. J. Acquir. Immune Defic. Syndr. 64, 279–283 (2013).
Milburn, J., Jones, R. & Levy, J. B. Renal effects of novel antiretroviral drugs. Nephrol. Dial. Transpl. 32, 434–439 (2016).
Deeks, E. D. Cobicistat: a review of its use as a pharmacokinetic enhancer of atazanavir and darunavir in patients with HIV-1 infection. Drugs 74, 195–206 (2014).
Spagnuolo, V., Castagna, A. & Lazzarin, A. Bictegravir. Curr. Opin. HIV AIDS 13, 326–333 (2018).
Yin, J. & Wang, J. Renal drug transporters and their significance in drug–drug interactions. Acta Pharm. Sin. B 6, 363–373 (2016).
Raffi, F. et al. Once-daily dolutegravir versus raltegravir in antiretroviral-naive adults with HIV-1 infection: 48 week results from the randomised, double-blind, non-inferiority SPRING-2 study. Lancet 381, 735–743 (2013).
Galizzi, N. et al. Glomerular filtration rate estimated by cystatin C formulas in HIV-1 patients treated with dolutegravir, rilpivirine or cobicistat. N. Microbiol. 41, 256–261 (2018).
Gallant, J. E. et al. Tenofovir DF, emtricitabine, and efavirenz vs. zidovudine, lamivudine, and efavirenz for HIV. N. Engl. J. Med. 354, 251–260 (2006).
Cutrell, J. & Bedimo, R. Single-tablet regimens in the treatment of HIV-1 infection. Fed. Pract. 33, 24S–30S (2016).
Truong, W. R., Schafer, J. J. & Short, W. R. Once-daily, single-tablet regimens for the treatment of HIV-1 infection. P T 40, 44–55 (2015).
Michienzi, S. M., Schriever, C. A. & Badowski, M. E. Abacavir/lamivudine/dolutegravir single tablet regimen in patients with human immunodeficiency virus and end-stage renal disease on hemodialysis. Int. J. STD AIDS 30, 181–187 (2019).
Eron, J. J. et al. Safety of elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide in HIV-1-infected adults with end-stage renal disease on chronic haemodialysis: an open-label, single-arm, multicentre, phase 3B trial. Lancet HIV. 6, e15–e24 (2019).
Guidelines for the use of antiretroviral agents in adults and adolescents with HIV. https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-arv/whats-new (accessed March 2023).
Cahn, P. et al. Durable efficacy of dolutegravir plus lamivudine in antiretroviral treatment-naive adults with HIV-1 infection: 96-week results from the GEMINI-1 and GEMINI-2 randomized clinical trials. J. Acquir. Immune Defic. Syndr. 83, 310–318 (2020). Erratum in: J Acquir Immune Defic Syndr. 84, e21 (2020).
van Welzen, B. J., Oomen, P. G. A. & Hoepelman, A. I. M. Dual antiretroviral therapy-all quiet beneath the surface? Front. Immunol. 12, 12 (2021).
Pérez-González, A., Suárez-García, I., Ocampo, A. & Poveda, E. Two-drug regimens for HIV-current evidence, research gaps and future challenges. Microorganisms 10, 433 (2022).
Palmer, C. S. et al. Emerging role and characterization of immunometabolism: relevance to HIV pathogenesis, serious non-AIDS events, and a cure. J. Immunol. 196, 4437–4444 (2016).
Greene, M., Steinman, M. A., McNicholl, I. R. & Valcour, V. Polypharmacy, drug-drug interactions, and potentially inappropriate medications in older adults with human immunodeficiency virus infection. J. Am. Geriatr. Soc. 62, 447–453 (2014).
Wei, A. et al. Long-term renal survival in HIV-associated nephropathy with angiotensin-converting enzyme inhibition. Kidney Int. 64, 1462–1471 (2003).
Wheeler, D. C. et al. Safety and efficacy of dapagliflozin in patients with focal segmental glomerulosclerosis: a prespecified analysis of the dapagliflozin and prevention of adverse outcomes in chronic kidney disease (DAPA-CKD) trial. Nephrol. Dial. Transpl. 37, 1647–1656 (2022).
Lipkowitz, M. S. et al. Apolipoprotein L1 gene variants associate with hypertension-attributed nephropathy and the rate of kidney function decline in African Americans. Kidney Int. 83, 114–120 (2013).
Freedman, B. I. & Cohen, A. H. Hypertension-attributed nephropathy: what’s in a name? Nat. Rev. Nephrol. 12, 27–36 (2016).
Friedman, D. J., Ma, L. & Freedman, B. I. Treatment potential in APOL1-associated nephropathy. Curr. Opin. Nephrol. Hypertens. 31, 442–448 (2022).
Zimmerman B., Dakin L., Fortier A. Small molecule APOL1 inhibitors block APOL1 pore function and reduce proteinuria in an APOL1-mediated kidney disease mouse model. J. Am. Soc. Nephrol. 2021. (Kidney Week Edition Abstract):419.
Egbuna, O. et al. Inaxaplin for proteinuric kidney disease in persons with two APOL1 variants. N. Engl. J. Med. 388, 969–979 (2023).
Falk, R. et al. Design of a phase 2/3 adaptive trail, AMPLITUDE, evaluating Inaxaplin in APOL1-mediated kidney disease. J. Am. Soc. Nephrol. (Suppl.) 33, Th-PO480 (2022).
Nichols, B. et al. Innate immunity pathways regulate the nephropathy gene apolipoprotein L1. Kidney Int. 87, 332–342 (2015).
Bruggeman, L. A., Sedor, J. R. & O’Toole, J. F. Apolipoprotein L1 and mechanisms of kidney disease susceptibility. Curr. Opin. Nephrol. Hypertens. 30, 317–323 (2021).
Singh, J. A. Filgotinib, a JAK1 inhibitor, for treatment-resistant rheumatoid arthritis. JAMA 322, 309–311 (2019).
Aghajan, M. et al. Antisense oligonucleotide treatment ameliorates IFN-g-induced proteinuria in APOL1-transgenic mice. JCI Insight 4, e126124 (2019).
Health Resources and Services Administration (HRSA), Department of Health and Human Services (HHS). Organ procurement and transplantation: implementation of the HIV Organ Policy Equity Act. Final rule. Fed. Regist. 80, 26464–26467 (2015).
Roland, M. E. et al. Survival in HIV-positive transplant recipients compared with transplant candidates and with HIV-negative controls. AIDS 30, 435–444 (2016).
Zheng, X. et al. Kidney transplant outcomes in HIV-positive patients: a systematic review and meta-analysis. AIDS Res. Ther. 16, 37 (2019).
Zarinsefat et al. Long-term outcomes following kidney and liver transplant in recipients with HIV. JAMA Surg. 157, 240–247 (2022). Erratum in: JAMA Surg. (2022).
Xia, Y. et al. Effect of HCV, HIV and coinfection in kidney transplant recipients: mate kidney analyses. Am. J. Transpl. 14, 2037–2047 (2014).
Locke, J. E. et al. A national study of outcomes among HIV-infected kidney transplant recipients. J. Am. Soc. Nephrol. 26, 2222–2229 (2015) .
Camargo, J. F. et al. Clinical outcomes in HIV+/HCV+ coinfected kidney transplant recipients in the pre- and post-direct-acting antiviral therapy eras: 10-Year single center experience. Clin. Transpl. 33, e13532 (2019).
Locke, J. E. et al. Survival benefit of kidney transplantation in HIV-infected patients. Ann. Surg. 265, 604–608 (2017).
Muller, E., Kahn, D. & Mendelson, M. Renal transplantation between HIV-positive donors and recipients. N. Engl. J. Med. 362, 2336–2337 (2010).
Selhorst, P. et al. Longer-term outcomes of HIV-positive-to-HIV-positive renal transplantation. N. Engl. J. Med. 381, 1387–1389 (2019).
Muller, E. & Barday, Z. HIV-positive kidney donor selection for HIV-positive transplant recipients. J. Am. Soc. Nephrol. 29, 1090–1095 (2018).
Durand, C. M. et al. A prospective multicenter pilot study of HIV-positive deceased donor to HIV-positive recipient kidney transplantation: HOPE in action. Am. J. Transpl. 21, 1754–1764 (2021).
Frassetto, L. et al. Changes in clearance, volume and bioavailability of immunosuppressants when given with HAART in HIV-1 infected liver and kidney transplant recipients. Biopharm. Drug. Dispos. 34, 442–451 (2013).
Flentge, C. A. et al. Synthesis and evaluation of inhibitors of cytochrome P450 3A (CYP3A) for pharmacokinetic enhancement of drugs. Bioorg. Med. Chem. Lett. 19, 5444–5448 (2009).
Hearps, A. C. et al. HIV infection and aging of the innate immune system. Sex. Health 8, 453–464 (2011).
Touzot, M. et al. Renal transplantation in HIV-infected patients: the Paris experience. Am. J. Transpl. 10, 2263–2269 (2010).
Rodrigo, E. et al. Heterogeneity of induction therapy in Spain: changing patterns according to year, centre, indications and results. NDT Plus 3, ii9–ii14 (2010).
Chin-Hong, P. V. & Kwak, E. J. AST infectious diseases community of practice. human papillomavirus in solid organ transplantation. Am. J. Transpl. 13, 189–200 (2013).
Jotwani, V., Li, Y., Grunfeld, C., Choi, A. I. & Shlipak, M. G. Risk factors for ESRD in HIV-infected individuals: traditional and HIV-related factors. Am. J. Kidney Dis. 59, 628–635 (2012).
Ryom, L. et al. Advanced chronic kidney disease, end-stage renal disease and renal death among HIV-positive individuals in Europe. HIV Med. 14, 503–508 (2013).
Muzaale, A. D. et al. Risk of end-stage renal disease in HIV-positive potential live kidney donors. Am. J. Transpl. 17, 1823–1832 (2017).
Grupper, A. et al. In sickness and in health: living HIV positive kidney donation from a wife to her husband, with 7 years’ post-transplant follow-up. Transpl. Infect. Dis. 21, e13171 (2019).
Johns Hopkins Medicine. First living donor HIV-to-HIV kidney transplant in the U.S. https://www.hopkinsmedicine.org/news/newsroom/news-releases/first-ever-living-donor-hiv-to-hiv-kidney-transplant-in-the-US (2019).
Kapuriya, M., Vaidya, A. & Rajkumar, V. Successful HIV-positive, live renal donor transplant. a unique method of expanding the donor pool. Transplantation 104, e140–e141 (2020).
Saldanha, N. et al. A preemptive living donor renal transplant in an HIV positive patient from an HIV positive donor. Indian J. Nephrol. 32, 375–377 (2022).
Doshi, M. D. et al. APOL1 genotype and renal function of black living donors. J. Am. Soc. Nephrol. 29, 1309–1316 (2018).
Reeves-Daniel, A. M. et al. The APOL1 gene and allograft survival after kidney transplantation. Am. J. Transpl. 11, 1025–1030 (2011).
Okumi, M. et al. ABO-incompatible living kidney transplants: evolution of outcomes and immunosuppressive management. Am. J. Transpl. 16, 886–896 (2016).
Katou, S. et al. ABO-incompatible living donor kidney transplantation in a human immunodeficiency virus-positive recipient from a human immunodeficiency virus-positive donor: a case report. Transpl. Proc. 52, 2739–2741 (2020).
Campara, M. et al. ABO incompatible renal transplantation in an HIV-seropositive patient. Transplantation 86, 176–178 (2008).
Thornton, J. Expanding HIV-positive organ donation. Lancet 397, 184–185 (2021).
Botha, J. et al. Living donor liver transplant from an HIV-positive mother to her HIV-negative child: opening up new therapeutic options. AIDS 32, F13–F19 (2018).
Pereira, M. R. et al. HIV transmission through living donor kidney transplant: an 11-year follow-up on the recipient and donor. Transpl. Infect. Dis. 23, e13691 (2021).
Lin, S.-N. et al. Outcomes of solid organ transplantation from an HIV positive donor to negative recipients. Am J Transplant. https://atcmeetingabstracts.com/abstract/outcomes-of-solid-organ-transplantation-from-an-hiv-positive-donor-to-negative-recipients (2016).
Mukhopadhyay, P. et al. Transmission of human immunodeficiency virus infection by renal transplantation. Indian. J. Nephrol. 22, 133–135 (2012).
Borchi, B. et al. Case report: HIV infection from a kidney transplant. Transpl. Proc. 42, 2267–2269 (2010).
Fischetti, B. et al. Real-world experience with higher-than-recommended doses of lamivudine in patients with varying degrees of renal impairment. Open. Forum Infect. Dis. 5, ofy225 (2018).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Nephrology thanks John He, Robert Kalyesubula and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Diana, N.E., Naicker, S. The changing landscape of HIV-associated kidney disease. Nat Rev Nephrol 20, 330–346 (2024). https://doi.org/10.1038/s41581-023-00801-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-023-00801-1