Abstract
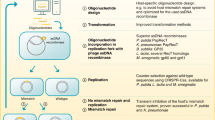
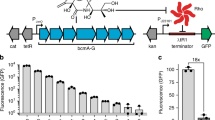
One key concept in the evolution of new functions is the ability of enzymes to perform promiscuous side-reactions that serve as a source of novelty that may become beneficial under certain conditions. Here, we identify a mechanism where a bacteriophage-encoded enzyme introduces novelty by inducing expression of a promiscuous bacterial enzyme. By screening for bacteriophage DNA that rescued an auxotrophic Escherichia coli mutant carrying a deletion of the ilvA gene, we show that bacteriophage-encoded S-adenosylmethionine (SAM) hydrolases reduce SAM levels. Through this perturbation of bacterial metabolism, expression of the promiscuous bacterial enzyme MetB is increased, which in turn complements the absence of IlvA. These results demonstrate how foreign DNA can increase the metabolic capacity of bacteria, not only by transfer of bona fide new genes, but also by bringing cryptic bacterial functions to light via perturbations of cellular physiology.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ochman, H., Lawrence, J. G. & Groisman, E. A. Lateral gene transfer and the nature of bacterial innovation. Nature 405, 299–304 (2000).
Leonard, G. & Richards, T. A. Genome-scale comparative analysis of gene fusions, gene fissions, and the fungal tree of life. Proc. Natl Acad. Sci. USA 109, 21402–21407 (2012).
Näsvall, J., Sun, L., Roth, J. R. & Andersson, D. I. Real-time evolution of new genes by innovation, amplification, and divergence. Science 338, 384–387 (2012).
Knowles, D. G. & Aoife, M. Recent de novo origin of human protein-coding genes. Genome Res. 19, 1752–1759 (2009).
Cai, J., Zhao, R., Jiang, H., & Wang, W. De novo origination of a new protein-coding gene in Saccharomyces cerevisiae. Genetics 179, 487–496 (2008).
Carvunis, A.-R. R. et al. Proto-genes and de novo gene birth. Nature 487, 370–374 (2012).
Copley, S. D. An evolutionary biochemist’s perspective on promiscuity. Trends Biochem. Sci. 40, 72–78 (2015).
Patrick, W. M., Quandt, E. M., Swartzlander, D. B. & Matsumura, I. Multicopy suppression underpins metabolic evolvability. Mol. Biol. Evol. 24, 2716–2722 (2007).
Bergmiller, T., Ackermann, M. & Silander, O. K. Patterns of evolutionary conservation of essential genes correlate with their compensability. PLoS Genet. 8, e1002803 (2012).
D’Ari, R. & Casadesús, J. Underground metabolism. Bioessays 20, 181–186 (1998).
Krahn, T. et al. Intraspecies transfer of the chromosomal Acinetobacter baumannii blaNDM-1 carbapenemase gene. Antimicrob. Agents Chemother. 60, 3032–3040 (2016).
Penadés, J. R. R., Chen, J., Nuria, Q.-P., Carpena, N. & Novick, R. P. Bacteriophage-mediated spread of bacterial virulence genes. Curr. Opin. Microbiol. 23, 171–178 (2015).
Kitagawa, M. et al. Complete set of ORF clones of Escherichia coli ASKA library (a complete set of E. coli K-12 ORF archive): unique resources for biological research. DNA Res. 12, 291–299 (2005).
Letunic, I., Doerks, T. & Bork, P. SMART: recent updates, new developments and status in 2015. Nucleic Acids Res. 43, D257–D260 (2015).
Söding, J., Biegert, A. & Lupas, A. N. The HHpred interactive server for protein homology detection and structure prediction. Nucleic Acids Res. 33, W244–W248 (2005).
Notredame, C., Higgins, D. & Heringa, J. T-Coffee: a novel method for fast and accurate multiple sequence alignment. J. Mol. Biol. 302, 205–217 (2000).
LaMonte, B. L. & Hughes, J. A. In vivo hydrolysis of S-adenosylmethionine induces the met regulon of Escherichia coli. Microbiol. Read. Engl. 152, 1451–1459 (2006).
van der Ploeg, J., Eichhorn, E. & Leisinger, T. Sulfonate-sulfur metabolism and its regulation in Escherichia coli. Arch. Microbiol. 176, 1–8 (2001).
Weissbach, H., & Brot, N. Regulation of methionine synthesis in Escherichia coli. Mol. Microbiol. 5, 1593–1597 (1991).
Spoerel, N. & Herrlich, P. Colivirus-T3-coded S-adenosylmethionine hydrolase. Eur. J. Biochem. 95, 227–233 (1979).
Hughes, J., Brown, L. & Ferro, A. Expression of the cloned coliphage T3 S-adenosylmethionine hydrolase gene inhibits DNA methylation and polyamine biosynthesis in Escherichia coli. J. Bacteriol. 169, 3625–3632 (1987).
Holbrook, E., Greene, R. & Krueger, J. Purification and properties of cystathionine gamma-synthase from overproducing strains of Escherichia coli. Biochem. 29, 435–442 (1990).
Mehta, P. K. & Christen, P. in Advances in Enzymology and Related Areas of Molecular Biology 74 (ed. Purich, D. L.) 129–184 (Wiley, New York, 2006).
Eliot, A. C. & Kirsch, J. F. Pyridoxal phosphate enzymes: mechanistic, structural, and evolutionary considerations. Annu. Rev. Biochem. 73, 383–415 (2004).
Born, T. & Blanchard, J. Enzyme-catalyzed acylation of homoserine: mechanistic characterization of the Escherichia coli metA-encoded homoserine transsuccinylase. Biochemistry 38, 14416–14423 (1999).
Usuda, Y. & Kurahashi, O. Effects of deregulation of methionine biosynthesis on methionine excretion in Escherichia coli. Appl. Env. Microbiol. 71, 3228–3234 (2005).
Mordukhova, E. A., Lee, H.-S. S. & Pan, J.-G. G. Improved thermostability and acetic acid tolerance of Escherichia coli via directed evolution of homoserine o-succinyltransferase. Appl. Env. Microbiol. 74, 7660–7668 (2008).
Laber, B. et al. Mechanisms of interaction of Escherichia coli threonine synthase with substrates and inhibitors. Biochemistry 33, 3413–3423 (1994).
Soo, V. W., Paulina, H.-M. & Patrick, W. M. Artificial gene amplification reveals an abundance of promiscuous resistance determinants in Escherichia coli. Proc. Natl Acad. Sci. USA 108, 1484–1489 (2011).
Kim, J., Kershner, J. P., Novikov, Y., Shoemaker, R. K. & Copley, S. D. Three serendipitous pathways in E. coli can bypass a block in pyridoxal-5’-phosphate synthesis. Mol. Syst. Biol. 6, 436 (2010).
Downs, D. M. Understanding microbial metabolism. Annu Rev. Microbiol. 60, 533–559 (2006).
Palmer, L. D., Paxhia, M. D. & Downs, D. M. Induction of the sugar-phosphate stress response allows Saccharomyces cerevisiae 2-methyl-4-amino-5-hydroxymethylpyrimidine phosphate synthase to function in Salmonella enterica. J. Bacteriol. 197, 3554–3562 (2015).
Notebaart, R. A. et al. Network-level architecture and the evolutionary potential of underground metabolism. Proc. Natl Acad. Sci. USA 111, 11762–11767 (2014).
Sévin, D. C., Fuhrer, T., Zamboni, N. & Sauer, U. Nontargeted in vitro metabolomics for high-throughput identification of novel enzymes in Escherichia coli. Nat. Methods 14, 187–194 (2017).
Canchaya, C., Fournous, G. & Brüssow, H. The impact of prophages on bacterial chromosomes. Mol. Microbiol. 53, 9–18 (2004).
Youle, M., Haynes, M. & Rohwer, F. in Viruses: Essential Agents of Life (ed. Witzany, G.) 61–81 (Springer, Dordrecht, 2012).
Studier, F. & Movva, N. SAMase gene of bacteriophage T3 is responsible for overcoming host restriction. J. Virol. 19, 136–145 (1976).
Bazurto, J. V., Farley, K. R. & Downs, D. M. An unexpected route to an essential cofactor: Escherichia coli relies on threonine for thiamine biosynthesis. mBio 7, e01840-15 (2016).
John, S. G. et al. A simple and efficient method for concentration of ocean viruses by chemical flocculation. Env. Microbiol. Rep. 3, 195–202 (2011).
Baba, T. et al. Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: the Keio collection. Mol. Syst. Biol. 2, 2006.0008 (2006).
Cherepanov, P. & Wackernagel, W. Gene disruption in Escherichia coli: TcR and KmR cassettes with the option of Flp-catalyzed excision of the antibiotic-resistance determinant. Gene 158, 9–14 (1995).
Zheng, L., Baumann, U. & Reymond, J.-L. L. An efficient one-step site-directed and site-saturation mutagenesis protocol. Nucleic Acids Res. 32, e115 (2004).
Datsenko, K. & Wanner, B. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl Acad. Sci. USA 97, 6640–6645 (2000).
Knopp, M. & Andersson, D. I. Amelioration of the fitness costs of antibiotic resistance due to reduced outer membrane permeability by upregulation of alternative porins. Mol. Biol. Evol. 32, 3252–3263 (2015).
Wiśniewski, J. R., Zougman, A., Nagaraj, N. & Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 6, 359–362 (2009).
Bailey, T. L. et al. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 37, W202–W208 (2009).
Vizcaíno, J. A. et al. 2016 update of the PRIDE database and its related tools. Nucleic Acids Res. 44, D447–D456 (2016).
Szklarczyk, D. et al. STRINGv10: protein–protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 43, D447–D452 (2015).
Acknowledgements
Enriched viral DNA from the CF6C, CF7LLP and Por1LT samples were graciously provided by F. L. Rohwer and S. D. Quistad. We acknowledge the assistance of S. Bertilsson in phage purification. The Proteomics Core Facility at the University of Gothenburg is grateful to IngaBritt och Arne Lundbergs Forskningsstiftelse for donating the Orbitrap Fusion Tribrid MS instrument. This work was supported by grants from the Swedish Research Council and the Knut and Alice Wallenberg foundation (to M.S. and D.I.A.).
Author information
Authors and Affiliations
Contributions
J.J.H, O.W., A.S., M.S. and D.I.A. designed the experiments. J.J.H., O.W., M.K., A.S. and M.S. carried out the experiments. U.E. analysed activity data. J.J.H, O.W., M.S. and D.I.A. wrote the manuscript with input from all authors. E.V. carried out the proteomic analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–8; Legends for Supplementary Tables 1–4; Supplementary References
Dataset 1
Supplementary Table 1: Data and lists of hits in screen
Dataset 2
Supplementary Table 2: HHpred searches with inserts sequences directing ΔilvA prototrophy
Dataset 3
Supplementary Table 3: Hierarchical clustering of TMT and RNA-seq data
Dataset 4
Supplementary Table 4: Strains and primers list
Rights and permissions
About this article
Cite this article
Jerlström Hultqvist, J., Warsi, O., Söderholm, A. et al. A bacteriophage enzyme induces bacterial metabolic perturbation that confers a novel promiscuous function. Nat Ecol Evol 2, 1321–1330 (2018). https://doi.org/10.1038/s41559-018-0568-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0568-5