Abstract
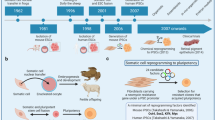
Patient-specific human-induced pluripotent stem cells (hiPSCs) hold great promise for the modelling of genetic disorders. However, these cells display wide intra- and interindividual variations in gene expression, which makes distinguishing true-positive and false-positive phenotypes challenging. Data from hiPSC phenotypes and human embryonic stem cells (hESCs) harbouring the same disease mutation are also lacking. Here, we report a comparison of the molecular, cellular and functional characteristics of three congruent patient-specific cell types—hiPSCs, hESCs and direct-lineage-converted cells—derived from currently available differentiation and direct-reprogramming technologies for use in the modelling of Charcot−Marie−Tooth 1A, a human genetic Schwann-cell disorder featuring a 1.4 Mb chromosomal duplication. We find that the chemokines C−X−C motif ligand chemokine-1 (CXCL1) and macrophage chemoattractant protein-1 (MCP1) are commonly upregulated in all three congruent models and in clinical patient samples. The development of congruent models of a single genetic disease using somatic cells from a common patient will facilitate the search for convergent phenotypes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the results of this study are available within the Article and the Supplementary Information. RNA sequencing data from CMT1A and control hESC−SCPs, hiPSC−SCPs and hiNC−Schwann cells have been uploaded to GEO under accession code GSE85598.
References
Park, I. H. et al. Disease-specific induced pluripotent stem cells. Cell 134, 877–886 (2008).
Lee, G. et al. Modelling pathogenesis and treatment of familial dysautonomia using patient-specific iPSCs. Nature 461, 402–406 (2009).
Mertens, J. et al. Directly reprogrammed human neurons retain aging-associated transcriptomic signatures and reveal age-related nucleocytoplasmic defects. Cell Stem Cell 17, 705–718 (2015).
van Paassen, B. W. et al. PMP22 related neuropathies: Charcot-Marie-Tooth disease type 1A and hereditary neuropathy with liability to pressure palsies. Orphanet J. Rare Dis. 9, 38 (2014).
Robaglia-Schlupp, A. et al. PMP22 overexpression causes dysmyelination in mice. Brain 125, 2213–2221 (2002).
Passage, E. et al. Ascorbic acid treatment corrects the phenotype of a mouse model of Charcot-Marie-Tooth disease. Nat. Med. 10, 396–401 (2004).
Burns, J. et al. Ascorbic acid for Charcot-Marie-Tooth disease type 1A in children: a randomised, double-blind, placebo-controlled, safety and efficacy trial. Lancet Neurol. 8, 537–544 (2009).
Verhamme, C. et al. Oral high dose ascorbic acid treatment for one year in young CMT1A patients: a randomised, double-blind, placebo-controlled Phase II trial. BMC Med. 7, 70 (2009).
Pareyson, D. et al. Ascorbic acid in Charcot-Marie-Tooth disease type 1A (CMT-TRIAAL and CMT-TRAUK): a double-blind randomised trial. Lancet Neurol. 10, 320–328 (2011).
Micallef, J. et al. Effect of ascorbic acid in patients with Charcot-Marie-Tooth disease type 1A: a multicentre, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 8, 1103–1110 (2009).
Lewis, R. A. et al. High-dosage ascorbic acid treatment in Charcot-Marie-Tooth disease type 1A: results of a randomized, double-masked, controlled trial. JAMA Neurol. 70, 981–987 (2013).
Suter, U. et al. Regulation of tissue-specific expression of alternative peripheral myelin protein-22 (PMP22) gene transcripts by two promoters. J. Biol. Chem. 269, 25795–25808 (1994).
Heine, W., Conant, K., Griffin, J. W. & Hoke, A. Transplanted neural stem cells promote axonal regeneration through chronically denervated peripheral nerves. Exp. Neurol. 189, 231–240 (2004).
Fu, S. Y. & Gordon, T. Contributing factors to poor functional recovery after delayed nerve repair: prolonged denervation. J. Neurosci. 15, 3886–3895 (1995).
Katona, I. et al. PMP22 expression in dermal nerve myelin from patients with CMT1A. Brain 132, 1734–1740 (2009).
Akdis, M. et al. Interleukins, from 1 to 37, and interferon-gamma: receptors, functions, and roles in diseases. J. Allergy Clin. Immunol. 127, 701–721 (2011).
Turner, M. D., Nedjai, B., Hurst, T. & Pennington, D. J. Cytokines and chemokines: at the crossroads of cell signalling and inflammatory disease. Biochim. Biophys. Acta 1843, 2563–2582 (2014).
Kim, Y. J. et al. Generation of multipotent induced neural crest by direct reprogramming of human postnatal fibroblasts with a single transcription factor. Cell Stem Cell 15, 497–506 (2014).
Germain, P. L. & Testa, G. Taming human genetic variability: transcriptomic meta-analysis guides the experimental design and interpretation of iPSC-based disease modeling. Stem Cell Rep. 8, 1784–1796 (2017).
Wu, J. et al. Insertional mutagenesis identifies a STAT3/Arid1b/beta-catenin pathway driving neurofibroma initiation. Cell Rep. 14, 1979–1990 (2016).
Woodhoo, A. et al. Notch controls embryonic Schwann cell differentiation, postnatal myelination and adult plasticity. Nat. Neurosci. 12, 839–847 (2009).
D'Antonio, M. et al. TGFbeta type II receptor signaling controls Schwann cell death and proliferation in developing nerves. J. Neurosci. 26, 8417–8427 (2006).
Clements, M. P. et al. The wound microenvironment reprograms Schwann cells to invasive mesenchymal-like cells to drive peripheral nerve regeneration. Neuron 96, 98–114(2017).
Lee, S. M., Chin, L. S. & Li, L. Dysregulation of ErbB receptor trafficking and signaling in demyelinating Charcot-Marie-Tooth disease. Mol. Neurobiol. 54, 87–100 (2017).
Nickols, J. C., Valentine, W., Kanwal, S. & Carter, B. D. Activation of the transcription factor NF-kappaB in Schwann cells is required for peripheral myelin formation. Nat. Neurosci. 6, 161–167 (2003).
Tang, W. et al. Expression of Nrf2 promotes Schwann cell-mediated sciatic nerve recovery in diabetic peripheral neuropathy. Cell Physiol. Biochem. 46, 1879–1894 (2018).
Mey, J., Schrage, K., Wessels, I. & Vollpracht-Crijns, I. Effects of inflammatory cytokines IL-1beta, IL-6, and TNFalpha on the intracellular localization of retinoid receptors in Schwann cells. Glia 55, 152–164 (2007).
Zhang, G. et al. Hypothalamic programming of systemic ageing involving IKK-β, NF-κB and GnRH. Nature 497, 211–216 (2013).
Trujillo, G. et al. Neutrophil recruitment to the lung in both C5a- and CXCL1-induced alveolitis is impaired in vitamin D-binding protein-deficient mice. J. Immunol. 191, 848–856 (2013).
Vries, M. H. et al. CXCL1 promotes arteriogenesis through enhanced monocyte recruitment into the peri-collateral space. Angiogenesis 18, 163–171 (2015).
Deshmane, S. L., Kremlev, S., Amini, S. & Sawaya, B. E. Monocyte chemoattractant protein-1 (MCP-1): an overview. J. Interf. Cytok. Res. 29, 313–326 (2009).
Kohl, B., Fischer, S., Groh, J., Wessig, C. & Martini, R. MCP-1/CCL2 modifies axon properties in a PMP22-overexpressing mouse model for Charcot-Marie-Tooth 1A neuropathy. Am. J. Pathol. 176, 1390–1399 (2010).
Kobsar, I., Hasenpusch-Theil, K., Wessig, C., Muller, H. W. & Martini, R. Evidence for macrophage-mediated myelin disruption in an animal model for Charcot-Marie-Tooth neuropathy type 1A. J. Neurosci. Res. 81, 857–864 (2005).
Lehmann, H. C. et al. Human Schwann cells retain essential phenotype characteristics after immortalization. Stem Cells Dev. 21, 423–431 (2012).
Monk, K. R. et al. A G protein-coupled receptor is essential for Schwann cells to initiate myelination. Science 325, 1402–1405 (2009).
Wainger, B. J. et al. Modeling pain in vitro using nociceptor neurons reprogrammed from fibroblasts. Nat. Neurosci. 18, 17–24 (2015).
Meyer Zu Horste, G. & Nave, K. A. Animal models of inherited neuropathies. Curr. Opin. Neurol. 19, 464–473 (2006).
Chittoor, V. G. et al. Biochemical characterization of protein quality control mechanisms during disease progression in the C22 mouse model of CMT1A. ASN Neuro. 5, e00128 (2013).
Misko, A., Ferguson, T. & Notterpek, L. Matrix metalloproteinase mediated degradation of basement membrane proteins in Trembler J neuropathy nerves. J. Neurochem. 83, 885–894 (2002).
Chambers, S. M. et al. Combined small-molecule inhibition accelerates developmental timing and converts human pluripotent stem cells into nociceptors. Nat. Biotechnol. 30, 715–720 (2012).
Jessen, K. R. & Mirsky, R. The origin and development of glial cells in peripheral nerves. Nat. Rev. Neurosci. 6, 671–682 (2005).
Kim, D. et al. TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol. 14, R36 (2013).
Anders, S., Pyl, P. T. & Huber, W. HTSeq-a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Choi, I. Y., . & LimH.. & Lee, G. Efficient generation human induced pluripotent stem cells from human somatic cells with Sendai-virus. J. Vis. Exp. 86, e51406 (2014).
Koyanagi-Aoi, M. et al. Differentiation-defective phenotypes revealed by large-scale analyses of human pluripotent stem cells. Proc. Natl Acad. Sci. USA 110, 20569–20574 (2013).
Kim, H. et al. miR-371-3 expression predicts neural differentiation propensity in human pluripotent stem cells. Cell Stem Cell 8, 695–706 (2011).
Sareen, D. et al. Targeting RNA foci in iPSC-derived motor neurons from ALS patients with a C9ORF72 repeat expansion. Sci. Transl. Med. 5, 208ra149 (2013).
Acknowledgements
The work in the Lee lab was supported by grants from the Robertson Investigator Award from the New York Stem Cell Foundation (G.L.), the CMT Association (G.L.), the National Institutes of Health through grant no. R01NS093213 (G.L.), the Muscular Dystrophy Association (G.L.) and MSCRF/TEDCO (G.L.). We also acknowledge salary support from the Johns Hopkins MD/PhD program (B.M.-C.), the FARMS Fellowship (B.M.-C.), the Adrienne Helis Malvin Medical Research Foundation (G.L., Y.O.) and the GRDC Programme through the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (2017K1A4A3014959). The work in the Kim lab was supported by grants from Kyung Hee University in 2016 (KHU-20160535), the Korea Health Technology R&D Project through the KHIDI funded by the Ministry of Health & Welfare, the Republic of Korea (HI16C2216) and NRF grants funded by the Korean government (NRF-2017R1C1B3009321, NRF-2017M3C7A1047640 and NRF-2017M3A9E4047243). The work in the Baloh lab was supported by grant nos. RN3-06530 (California Institute for Regenerative Medicine) and NS097545 (National Institutes of Health). The work in the Studer lab was supported by the New York State Stem Cell Fund (G.L., K.E. and L.S.) and New York state stem cell science program (NYSTEM, contract C32599GG). The work in the Hoke lab was supported by MSCRF/TEDCO and the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation. The work in the Brandacher lab was supported by MSCRF/TEDCO.
Author information
Authors and Affiliations
Contributions
R.H.B., G.B., A.H., L.S. and G.L. conceived the study. B.M.-C., Y.J.K., K.E., R.H.B., G.B., A.H. and L.S. designed the study. B.M.-C., Y.J.K., R.M., B. K., I.Y.C., H.L., Y.O., B.L., K.J.K., S.B., J.K.H., W.H., O.H., Y.H.C. and G.L. performed experiments. B.M.-C., Y.J.K., R.M., B. K., I.Y.C., H.L., Y.O., B.L., K.J.K., S.B., J.K.H., W.H., O.H., Y.H.C., L.S. and G.L. analysed the data. B.M.-C., Y.J.K. and G.L. contributed to the data assembly. B.M.-C., Y.J.K. and G.L. interpreted the results. B.M.-C., Y.J.K. and G.L. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures and table captions.
Supplementary Table 1
Unfiltered microarray results for CMT1A versus control hiPSC-derived Schwann cells.
Supplementary Table 2
Detailed data characterizing the original patient and embryonic sources of the fibroblasts and hESCs used.
Supplementary Table 3
RNA-sequencing data from control hESC-derived, hiPSC-derived and hiNC-derived Schwann cells, analysed with conventional methodologies and DAVID.
Supplementary Table 4
RNA-sequencing data from control hESC-derived, hiPSC-derived and hiNC-derived Schwann cells, analyzed with ‘power kit’ methodology and DAVID.
Supplementary Table 5
RNA-sequencing data evaluated for differentially expressed genes between CMT1A versus control cells from hESC-derived, hiPSC-derived and hiNC-derived Schwann cells. Analysed with DAVID and filtered for inflammation-related categories.
Supplementary Table 6
RNA-sequencing data evaluated for differentially expressed genes between CMT1A versus control cells from hESC-derived, hiPSC-derived and hiNC-derived Schwann cells. Analysed with Ingenuity Pathway Analysis in search of candidate pathologic pathways.
Supplementary Table 7
Cytokine-array results from hESC-derived, hiPSC-derived and hiNC-derived Schwann cells.
Supplementary Table 8
List of qRT-PCR primers used.
Supplementary Table 9
List of primary antibodies used.
Rights and permissions
About this article
Cite this article
Mukherjee-Clavin, B., Mi, R., Kern, B. et al. Comparison of three congruent patient-specific cell types for the modelling of a human genetic Schwann-cell disorder. Nat Biomed Eng 3, 571–582 (2019). https://doi.org/10.1038/s41551-019-0381-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0381-8
This article is cited by
-
Enhancing Viability of Human Embryonic Stem Cells during Cryopreservation via RGD-REP-Mediated Activation of FAK/AKT/FoxO3a Signaling Pathway
Tissue Engineering and Regenerative Medicine (2023)
-
OCT4-induced oligodendrocyte progenitor cells promote remyelination and ameliorate disease
npj Regenerative Medicine (2022)
-
Approaches to characterize the transcriptional trajectory of human myogenesis
Cellular and Molecular Life Sciences (2021)
-
Mechanisms and Treatments in Demyelinating CMT
Neurotherapeutics (2021)