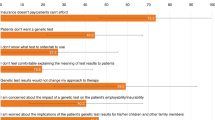
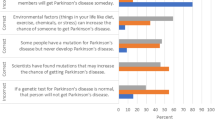
Abstract
Black patients are diagnosed with Parkinson’s disease (PD) at half the rate as White patients. The reasons for this large disparity are unknown. Here, we review evidence that practitioner bias may contribute. A key sign of PD is hypomimia or decreased facial expressivity. However, practitioner bias surrounding facial expressivity in Black people versus White people may lead practitioners to appraise Black patients with hypomimia as having higher levels of facial expressivity. Furthermore, practitioner bias may cause them to characterize reduced facial expressivity as being due to negative personality traits, as opposed to a medical sign, in Black patients with hypomimia. This racial bias in the evaluation of hypomimia in Black versus White patients could profoundly impact subsequent referral decisions and rates of diagnosis of PD. Therefore, exploring these differences is expected to facilitate addressing health care disparities through earlier and more accurate detection of PD in Black patients.
Similar content being viewed by others
Parkinson’s disease (PD) is a neurodegenerative disorder that leads to reduced quality of life, severe disability, and societal cost1,2. Parkinson’s disease affects approximately 1 million people in the United States (US), and that number is projected to double in the next 25 years2. There is a large racial disparity in the prevalence and incidence of PD in Black and White people3,4,5. “Black” is typically defined as a person having origins in any of the Black racial groups of Africa, whereas “White” is typically defined as a person having origins in any of the original peoples of Europe, the Middle East, or North Africa6,7. For example, the prevalence of PD is 50% lower in Black individuals as compared to White individuals, where Black people are half as likely as White people to receive a diagnosis of this disease3,5. In the US, the 4-year cumulative incidence of PD has been estimated at 54 per 100,000 among White individuals compared to 23 per 100,000 among Black individuals4.
Some factors that might contribute to this large racial disparity in the prevalence and incidence rates of PD in Black and White individuals are genetic or biological differences and systemic and structural factors in health care3,8. These factors include access to insurance, patient mistrust in health care, and under-reporting of symptoms. However, in the literature, no significant differences were found between Black and White people on genetic or biological factors3,9,10,11,12,13. Of note, these studies often had a low sample size of Black people which affected their generalizability. Hence, larger studies are needed. Regardless, this large disparity in the prevalence and incidence of PD still remains even after controlling for risk factors (demographic and structural) such as age, sex, geography, Medicaid eligibility, and average number of hospital visits4. One unexplored possibility is that health-care professionals evaluate motor signs of PD, such as hypomimia (reduced facial expressivity), differently in Black and White patients, a form of practitioner bias. Thus, in this perspective article, we hypothesize that practitioners are under-pathologizing reduced facial expressivity in Black patients with hypomimia. Further, practitioners could be viewing Black people as having higher levels of facial expressivity in comparison to White patients displaying the same level of facial expressivity/severity of hypomimia. In addition, practitioners are likely appraising reduced facial expressivity as being attributed to poor emotional engagement or attitudes, as opposed to a medical pathology, in Black patients with hypomimia in comparison to White patients with the same level of facial expressivity/severity of hypomimia. Indeed, practitioner bias may affect early evaluation of PD patients, leading to under-recognition of early or prodromal signs of PD in Black patients and further perpetuating disparities in diagnosis, treatment, and outcomes for these patients.
Epidemiology of PD and prodromal motor signs
PD is caused by dysfunction in midbrain dopaminergic neurons, which are important for the production and modulation of movement14. A reduction in dopamine neurons contributes to the development of the cardinal motor signs in PD; tremors, rigidity, postural instability, and bradykinesia15. Bradykinesia is a key part of many diagnostic criteria and is requisite for diagnosing PD; thus, it is crucial for practitioners to accurately identify this sign in patients15,16. Further, hypomimia is one manifestation of bradykinesia17,18. Of added importance, hypomimia can be one of the first motor manifestations and is a distinctive clinical feature in patients with PD, presenting as many as 10 years before a clinical diagnosis of PD is determined17,18,19. Hypomimia is present in most patients with PD (approximately 92%; race not reported)18,20. Therefore, accurately identifying signs such as hypomimia early in the course of PD could facilitate a timely diagnosis for patients. Further, failure to accurately identify this early sign of PD may result in a delay or false-negative error in the diagnosis of PD and could lead to an underestimation of the prevalence and incidence of this disease in the population, and could have other negative consequences as well.
Epidemiological research has found that the observed prevalence rates of PD are approximately 50% lower in Black patients compared to White patients in the US3,4,5. It is possible that these statistics are an underestimation of the true prevalence and incidence rate of PD among these racial groups. Disease awareness has influenced the diagnosis of other neurological diseases; for instance, the prevalence of multiple sclerosis (MS) in Black people has increased as a result of education of healthcare professionals and the community21,22. In fact, the prevalence of other neurological diseases such as MS has increased drastically over the years in Black people – for MS, for example, the crude prevalence rate was 22 per 100,000 in 1998–2000 in comparison to the current age-standardized estimate of 521.4 per 100,00022. The low prevalence of MS in previous years could have been the result of underdiagnosis of the disease by practitioners due to bias, among other factors. A similar phenomenon might be occurring with PD.
Prevalence and incidence statistics can be affected by many factors including methodological issues and systemic factors within the health care system8,23,24,25,26. Schoenberg and colleagues (1985) found that the prevalence rate of PD was not significantly different between Black and White patients within a community in Copiah County, Mississippi (196 vs 280 per 100,000 respectively), after removing healthcare access from the inclusion criteria for their study26. Black people tend to have less access to healthcare and are less likely to see neurologists, in comparison to White people24,27. Therefore, by removing this factor, researchers were able to include Black patients that were typically underserved in healthcare, and in prevalence and incidence research on PD at that time. They also discovered that twice as many of Black patients with PD had no previous PD diagnoses recorded in their medical records in comparison to Whites, which could indicate that PD signs/symptoms were missed by physicians altogether in many Black patients26. Thus, this racial disparity in the evaluation and identification of PD motor signs resulted in a delay or missed diagnosis of PD in Black people, which led to gross underestimations of the true prevalence of this disease within Black patients.
More recent studies have shown that practitioners’ bias related to gender can delay referrals for PD, citing that the duration between the onset of PD signs/symptoms and a referral to a movement disorder specialist was 41% greater in women than in men, after controlling for factors such as time to first primary care visit following PD sign/symptom onset, stage of the disease, age, and family history28. Therefore, practitioner’s bias when evaluating motor signs like hypomimia in patients, could contribute to diagnostic delay for PD and, ultimately an underestimation of true prevalence of this disease for Black people.
Practitioner bias can influence the evaluation of patients with hypomimia
No work has compared the impact of general practitioners’ bias on their evaluations of hypomimia (severity) in Black and White patients with PD. One of the few studies that have investigated the impact of practitioners’ bias on their impressions of mood and traits in patients with PD and hypomimia, used a Caucasian and Asian Taiwanese sample29. They found that practitioners were biased in favor of White American patients when evaluating sociability and biased in favor of Asian Taiwanese patients when evaluating cognitive competence and social supportiveness. Practitioners perceived the White patients as being more sociable and Taiwanese patients as being more competent and having more social support. The Tickle-Degnen et. al (2011) study raises the possibility that racial stereotypes are influencing practitioners’ impression and evaluation of Black people with hypomimia29.
Practitioners hold stereotypes surrounding facial expressivity in Black people, that can influence their ability to accurately appraise hypomimia in Black patients. White people (both laypersons and experts) have difficulty recognizing the type and magnitude of facial expressions in Black people. Specifically, White people have trouble distinguishing between genuine and false smiles on Black people’s faces30. These individuals are also more attuned to the expression and magnitude of anger or negatively valenced emotions on Black faces compared to those on White faces that show a similar intensity of emotion31,32,33. This finding is important because patients with PD and hypomimia appear to exhibit more negatively valenced emotions29. Practitioners with this type of bias could potentially view Black patients with PD as having higher levels of emotional expression than White patients with PD, when in fact the levels of expressivity are objectively the same. Consequentially, practitioners might assume that a White patient has more hypomimia impairment than a Black patient, which for the Black patient could lead to a delayed referral to a movement disorder specialist for expert evaluation of possible PD. Ultimately, delays like this could promote large disparities in treatment recommendations and outcomes between Black and White patients.
Delays in treatment of motor and non-motor symptoms in PD can lead to worse outcomes such as falls, decreased quality of life and faster disease progression34,35. Researchers have reported that Black patients with PD had higher mortality rates, presented at more advanced stages of PD, and had more motor impairment than White patients with PD after controlling for education and income36. These findings further highlight the importance of identifying whether practitioner’s racial bias is leading to a delay in or missed diagnosing of Black patients with hypomimia and PD.
Practitioner bias can contribute to negative impression of black patients with hypomimia
Bias may also influence practitioners’ impressions of mood and traits in Black patients which can impact their ability to appraise reduced facial expressivity as being a medical sign of PD in these patients. Further, practitioners may misappraise reduced facial expressivity as being a negative personality trait and contribute to negative impressions about attitudes and level of engagement in Black people37,38. It is possible that practitioners’ hold stereotypes that influence their impressions of Black people’s attitudes, during medical interviews. The preponderance of these stereotypes has the potential to ultimately impact practitioners’ ability to appraise reduced facial expressivity as a pathological sign of PD in Black people.
Practitioner bias can also affect non-verbal communication with non-white patients39. Practitioners view Black patients as being less medically cooperative, risky and more mistrustful than White patients, and reported a greater penchant for working with White patients over Black patients40,41. These findings further highlight the possibility that practitioners may form negative impressions about Black patients with hypomimia. This becomes even more significant given that such racial bias can produce differences in interview styles and can affect treatment of Black patients38,39. Practitioners may be more likely to characterize reduced facial expressivity as a negative personality trait, such as poor emotional engagement, as opposed to a medical pathology in Black patients than in White patients. Such misappraisal can cause a delay in referral to appropriate care for Black individuals with PD and hypomimia. Evaluating the impact that practitioners’ racial bias has on their appraisal and impressions of facial expressivity might help facilitate timely referrals to specialist care and accurate diagnosis of this disease and better treatment outcomes in Black patients.
Interventions, limitations, and future directions
Disparities in the prevalence and incidence of PD among Black and White people is multifactorial but may be driven in large part by practitioner bias. Specifically, practitioners’ racial bias could lead to misappraising and under-pathologizing of reduced facial expressivity in Black patients in comparison to White patients, with hypomimia. Exploring sources of health care biases (e.g., practitioner racial bias) in PD may reveal feasible points of intervention for reducing healthcare disparities in PD. For example, targeted intervention techniques such as implicit bias training can reduce biases in practitioners42,43. A previous study utilized a multifaceted intervention program, aimed at mitigating the disparities of cancer treatment in Black and White patients, as studies have shown that practitioner’s bias and poor communication leads to treatment variability42. However, interventions with training on implicit bias, gatekeeping, and institutional racism for practitioners reduced the disparity in outcomes; indeed, treatment completion in Black patients increased from being 10% lower to being almost equal to White patients42. Similar interventions aimed at addressing processes underlying faulty impression formation could attenuate racial disparities in the diagnosis and treatment of medical signs such as hypomimia, as seen in other chronic disorders such as PD29.
Another way to reduce health disparities is through fostering trust in healthcare and improving knowledge of PD signs in Black people. Medical mistrust is correlated with patient-perceived discrimination, which is related to their race, income, and insurance. Therefore, multilevel community- and theory-based training models aimed at increasing structural competency (the understanding of the impact of the social structure on a social group or individual) in practitioners can help to mitigate medical mistrust in Black patients and improve engagement in their health care44,45.
Increasing health literacy in Black people with PD signs and symptoms will improve self-advocacy, and is another way to help further reduce racial disparities in outcomes for these patients. Health literacy is the ability to obtain and understand health information in order to make informed decisions regarding healthcare and tend to be lower in Black people46,47. Further, low health literacy is associated with greater disease severity and morbidity in patients with PD and, in general, is moderated by education and medical mistrust in Black people46,47. Therefore, increasing health literacy in these individuals through adopting culturally tailored, interactive, and community-engaged health literacy approaches, would increase Black patients’ ability to advocate for necessary medical resource/services. This is especially important in the context of PD since Black people are less likely to see neurologists, in comparison to White people24,27. Overall, improving health literacy in Black patients will aid in their ability to understand and advocate for their health, especially when practitioner’s bias is impacting the medical encounter.
Limitation
One limitation to this viewpoint is that there may be genetic or biological factors that are driving differences in PD prevalence between Black and White people. As noted, there are few studies on this topic with an adequate sample size. Therefore, PD research equity may result from increased patient awareness of clinical research opportunities, increased availability of culturally diverse research materials, and partnership with community organizations48. Additionally, targeted training may reduce racial disparities in practitioners’ referrals of Black patients to clinical trials48. Identifying the sources of racial disparities in PD will be highly clinically meaningful.
Future direction
Future research will explore ways in which practitioner bias impacts their evaluation of advanced motor and non-motor signs of PD in patients from other racially diverse groups, such as Asian people, given the lower prevalence of PD in this population in comparison to their White counterparts5. In addition, researchers can investigate whether factors such as practitioners’ race or years of experience in healthcare moderate their ability to accurately appraise PD signs, such as hypomimia, in Black patients. This line of work is significant as it will aid in the development of intervention techniques and policies aimed at reducing health disparities for Black individuals with PD and other movement disorders.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed for the current article.
References
Chen, J. J. Parkinson’s disease: Health-related quality of life, economic cost, and implications of early treatment. Am. J. Manag Care. 16, S87–S93 (2010).
Yang, W. et al. Current and projected future economic burden of Parkinson’s disease in the U.S. NPJ Park Dis. 6, 15 (2020).
Bailey, M., Anderson, S. & Hall, D. A. Parkinson’s disease in African Americans: a review of the current literature. J. Parkinsons Dis. 10, 831–841 (2020).
Dahodwala, N. et al. Racial differences in the diagnosis of Parkinson’s disease. Mov. Disord. 24, 1200–1205 (2009a).
Wright Willis, A., Evanoff, B. A., Lian, M., Criswell, S. R. & Racette, B. A. Geographic and ethnic variation in Parkinson disease: a population-based study of US Medicare beneficiaries. Neuroepidemiology 34, 143–151 (2010).
Bhopal, R. & Donaldson, L. White, European, Western, Caucasian, or what? Inappropriate labeling in research on race, ethnicity, and health. Am. J. Public Health 88, 1303–1307 (1998).
Flanagin, A., Frey, T. & Christiansen, S. L., AMA Manual of Style Committee. Updated guidance on the reporting of race and ethnicity in medical and science journals. JAMA 326, 621–627 (2021).
Dahodwala, N., Xie, M., Noll, E., Siderowf, A. & Mandell, D. S. Treatment disparities in Parkinson’s disease. Ann. Neurol. 66, 142–145 (2009b).
Jain, S. et al. The risk of Parkinson disease associated with urate in a community-based cohort of older adults. Neuroepidemiology 36, 223–229 (2011).
Maynard, J. W. et al. Racial differences in gout incidence in a population-based cohort: atherosclerosis risk in communities study. Am. J. Epidemiol. 179, 576–583 (2014).
McGuire, V. et al. Association of DRD2 and DRD3 polymorphisms with Parkinson’s disease in a multiethnic consortium. J. Neurol. Sci. 307, 22–29 (2011).
McInerney-Leo, A., Gwinn-Hardy, K. & Nussbaum, R. L. Prevalence of Parkinson’s disease in populations of African ancestry: a review. J. Natl Med. Assoc. 96, 974–979 (2004).
Olsen, J. H., Friis, S. & Frederiksen, K. Malignant melanoma and other types of cancer preceding Parkinson disease. Epidemiology 17, 582–587 (2006).
Wichmann, T. & Dostrovsky, J. O. Pathological basal ganglia activity in movement disorders. Neuroscience 198, 232–244 (2011).
Postuma, R. B. et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 30, 1591–1601 (2015).
Tarakad, A. & Jankovic, J. Diagnosis and management of Parkinson’s disease. Semin. Neurol. 37, 118–126 (2017).
Maycas-Cepeda, T. et al. Hypomimia in Parkinson’s disease: what is it telling us. Front. Neurol. 11, 603582 (2021).
Novotny, M. et al. Automated video-based assessment of facial bradykinesia in de-novo Parkinson’s disease. NPJ Digit. Med. 5, 98 (2022).
Fereshtehnejad, S. M., Skogar, Ö. & Lökk, J. Evolution of orofacial symptoms and disease progression in idiopathic Parkinson’s disease: longitudinal data from the Jönköping Parkinson Registry. Parkinson’s Dis. 2017, 7802819 (2017).
Ricciardi, L. et al. Hypomimia in Parkinson’s disease: an axial sign responsive to levodopa. Eur. J. Neurol. 27, 2422–2429 (2020).
Amezcua, L., Rivera, V. M., Vazquez, T. C., Baezconde-Garbanati, L. & Langer-Gould, A. Health disparities, inequities, and social determinants of health in multiple sclerosis and related disorders in the US: a review. JAMA Neurol. 78, 1515–1524 (2021).
Langer-Gould, A. M., Gonzales, E. G., Smith, J. B., Li, B. H. & Nelson, L. M. Racial and ethnic disparities in multiple sclerosis prevalence. Neurology 98, e1818–e1827 (2022).
Dahodwala, N., Willis, A. W., Li, P. & Doshi, J. A. Prevalence and correlates of anti-Parkinson drug use in a nationally representative sample. Mov. Disord. Clin. Pract. 4, 335–341 (2016).
Saadi, A., Himmelstein, D. U., Woolhandler, S. & Mejia, N. I. Racial disparities in neurologic health care access and utilization in the United States. Neurology 88, 2268–2275 (2017).
Schneider, M. G. et al. Minority enrollment in Parkinson’s disease clinical trials. Parkinsonism Relat. Disord. 15, 258–262 (2009).
Schoenberg, B. S., Anderson, D. W. & Haerer, A. F. Prevalence of Parkinson’s disease in the biracial population of Copiah County, Mississippi. Neurology 35, 841–845 (1985).
Shi, L., Chen, C. C., Nie, X., Zhu, J. & Hu, R. Racial and socioeconomic disparities in access to primary care among people with chronic conditions. J. Am. Board Fam. Med. 27, 189–198 (2014).
Saunders-Pullman, R., Wang, C., Stanley, K. & Bressman, S. B. Diagnosis and referral delay in women with Parkinson’s disease. Gend. Med. 8, 209–217 (2011).
Tickle-Degnen, L., Zebrowitz, L. A. & Ma, H. I. Culture, gender and health care stigma: practitioners’ response to facial masking experienced by people with Parkinson’s disease. Soc. Sci. Med. 73, 95–102 (2011).
Friesen, J. P. et al. Perceiving happiness in an intergroup context: the role of race and attention to the eyes in differentiating between true and false smiles. J. Pers. Soc. Psychol. 116, 375–395 (2019).
Hugenberg, K. & Bodenhausen, G. V. Facing prejudice: Implicit prejudice and the perception of facial threat. Psychol. Sci. 14, 640–643 (2003).
Hugenberg, K. & Bodenhausen, G. V. Ambiguity in social categorization: the role of prejudice and facial affect in race categorization. Psychol. Sci. 15, 342–345 (2004).
Hutchings, P. B. & Haddock, G. Look Black in anger: the role of implicit prejudice in the categorization and perceived emotional intensity of racially ambiguous faces. J. Exp. Soc. Psychol. 44, 1418–1420 (2008).
Merola, A. et al. Earlier versus later subthalamic deep brain stimulation in Parkinson’s disease. Parkinsonism Relat. Disord. 21, 972–975 (2015).
Parkinson Study Group. A controlled, randomized, delayed-start study of Rasagiline in early Parkinson disease. Arch. Neurol. 61, 561–566 (2004).
Dahodwala, N., Karlawish, J., Siderowf, A., Duda, J. E. & Mandell, D. S. Delayed Parkinson’s disease diagnosis among African-Americans: the role of reporting of disability. Neuroepidemiology 36, 150–154 (2011).
Hagiwara, N., Dovidio, J. F., Eggly, S. & Penner, L. A. The effects of racial attitudes on affect and engagement in racially discordant medical interactions between non-Black physicians and Black patients. Group Process Intergroup Relat. 19, 509–527 (2016).
Hall, W. J. et al. Implicit racial/ethnic bias among health care professionals and its influence on health care outcomes: A systematic review. Am. J. Public Health 105, e60–e76 (2015).
Levine, C. S. & Ambady, N. The role of non-verbal behaviour in racial disparities in health care: implications and solutions. Med Edu. 47, 867–876 (2013).
Oliver, M. N., Wells, K. M., Joy-Gaba, J. A., Hawkins, C. B. & Nosek, B. A. Do physicians’ implicit views of African Americans affect clinical decision making? J. Am. Board Fam. Med. 27, 177–188 (2014).
van Ryn, M. & Burke, J. The effect of patient race and socio-economic status on physicians’ perceptions of patients. Soc. Sci. Med. 50, 813–828 (2000).
Cykert, S. et al. A multi-faceted intervention aimed at Black-White disparities in the treatment of early stage cancers: the ACCURE pragmatic quality improvement trial. J. Natl Med Assoc. 112, 468–477 (2020).
Kawakami, K., Dovidio, J. F., Moll, J., Hermsen, S. & Russin, A. Just say no (to stereotyping): effects of training in the negation of stereotypic associations on stereotype activation. J. Pers. Soc. Psychol. 78, 871–888 (2000).
Bazargan, M., Cobb, S. & Assari, S. Discrimination and medical mistrust in a racially and ethnically diverse sample of California adults. Ann. Fam. Med. 19, 4–15 (2021).
Wang, E. E. Structural Competency: What is it, why do we need it, and what does the structurally competent emergency physician look like? AEM Educ. Train. 4, S140–S142 (2019).
Fleisher, J. E., Shah, K., Fitts, W. & Dahodwala, N. A. Associations and implications of low health literacy in Parkinson’s Disease. Mov. Disord. Clin. Pract. 3, 250–256 (2016).
Muvuka, B. et al. Health literacy in African-American communities: barriers and strategies. Health Lit. Res Pract. 4, e138–e143 (2020).
Adrissi, J. & Fleisher, J. Moving the dial toward equity in Parkinson’s disease clinical research: a review of current literature and future directions in diversifying PD clinical trial participation. Curr. Neurol. Neurosci. Rep. 22, 475–483 (2022).
Acknowledgements
This work was supported in part by the National Institute of Health T32 pre-doctoral training grant: [T32GM108540] (S.H). The funder played no role in the study design, data collection, analysis, and interpretation of data, or the writing of this manuscript.
Author information
Authors and Affiliations
Contributions
S.H. conceptualized and organized the research idea. S.H. was the major contributor to writing and revising the manuscript. N.S.N. contributed to the conceptualization of the research idea and editing the manuscript. D.T. contributed to the conceptualization of the research idea and editing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Harris, S., Narayanan, N.S. & Tranel, D. Does Black vs. White race affect practitioners’ appraisal of Parkinson’s disease?. npj Parkinsons Dis. 9, 106 (2023). https://doi.org/10.1038/s41531-023-00549-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-023-00549-2