Abstract
Hundreds of leucine-rich repeat receptor kinases (LRR-RKs) have evolved to control diverse processes of growth, development and immunity in plants, but the mechanisms that link LRR-RKs to distinct cellular responses are not understood. Here we show that two LRR-RKs, the brassinosteroid hormone receptor BRASSINOSTEROID INSENSITIVE 1 (BRI1) and the flagellin receptor FLAGELLIN SENSING 2 (FLS2), regulate downstream glycogen synthase kinase 3 (GSK3) and mitogen-activated protein (MAP) kinases, respectively, through phosphocoding of the BRI1-SUPPRESSOR1 (BSU1) phosphatase. BSU1 was previously identified as a component that inactivates GSK3s in the BRI1 pathway. We surprisingly found that the loss of the BSU1 family phosphatases activates effector-triggered immunity and impairs flagellin-triggered MAP kinase activation and immunity. The flagellin-activated BOTRYTIS-INDUCED KINASE 1 (BIK1) phosphorylates BSU1 at serine 251. Mutation of serine 251 reduces BSU1’s ability to mediate flagellin-induced MAP kinase activation and immunity, but not its abilities to suppress effector-triggered immunity and interact with GSK3, which is enhanced through the phosphorylation of BSU1 at serine 764 upon brassinosteroid signalling. These results demonstrate that BSU1 plays an essential role in immunity and transduces brassinosteroid–BRI1 and flagellin–FLS2 signals using different phosphorylation sites. Our study illustrates that phosphocoding in shared downstream components provides signalling specificities for diverse plant receptor kinases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The RNA-seq data that support the findings of this study have been deposited in GEO with the accession code GSE140037. The MS proteomics data are available via ProteomeXchange with the identifiers PXD016283 (username, reviewer67695@ebi.ac.uk; password, pCDR9H2R, for DDA mode) and PXD016257 (username, reviewer88035@ebi.ac.uk; password, IQKH8jha, for PRM mode). Source data are provided with this paper.
References
Shiu, S. H. et al. Comparative analysis of the receptor-like kinase family in Arabidopsis and rice. Plant Cell 16, 1220–1234 (2004).
Li, J. et al. BAK1, an Arabidopsis LRR receptor-like protein kinase, interacts with BRI1 and modulates brassinosteroid signaling. Cell 110, 213–222 (2002).
Nam, K. H. & Li, J. BRI1/BAK1, a receptor kinase pair mediating brassinosteroid signaling. Cell 110, 203–212 (2002).
Chinchilla, D. et al. A flagellin-induced complex of the receptor FLS2 and BAK1 initiates plant defence. Nature 448, 497–500 (2007).
Tang, W. et al. BSKs mediate signal transduction from the receptor kinase BRI1 in Arabidopsis. Science 321, 557–560 (2008).
Kim, T. W., Guan, S. H., Burlingame, A. L. & Wang, Z. Y. The CDG1 kinase mediates brassinosteroid signal transduction from BRI1 receptor kinase to BSU1 phosphatase and GSK3-like kinase BIN2. Mol. Cell 43, 561–571 (2011).
Kim, T. W. et al. Brassinosteroid signal transduction from cell-surface receptor kinases to nuclear transcription factors. Nat. Cell Biol. 11, 1254–U1233 (2009).
Tang, W. Q. et al. PP2A activates brassinosteroid-responsive gene expression and plant growth by dephosphorylating BZR1. Nat. Cell Biol. 13, 124–U149 (2011).
Kim, T. W. & Wang, Z. Y. Brassinosteroid signal transduction from receptor kinases to transcription factors. Annu. Rev. Plant Biol. 61, 681–704 (2010).
Lu, D. et al. A receptor-like cytoplasmic kinase, BIK1, associates with a flagellin receptor complex to initiate plant innate immunity. Proc. Natl Acad. Sci. USA 107, 496–501 (2010).
Zhang, J. et al. Receptor-like cytoplasmic kinases integrate signaling from multiple plant immune receptors and are targeted by a Pseudomonas syringae effector. Cell Host Microbe 7, 290–301 (2010).
DeFalco, T. A. & Zipfel, C. Molecular mechanisms of early plant pattern-triggered immune signaling. Mol. Cell 81, 3449–3467 (2021).
Lin, W. et al. Inverse modulation of plant immune and brassinosteroid signaling pathways by the receptor-like cytoplasmic kinase BIK1. Proc. Natl Acad. Sci. USA 110, 12114–12119 (2013).
Shi, H. et al. BR-SIGNALING KINASE1 physically associates with FLAGELLIN SENSING2 and regulates plant innate immunity in Arabidopsis. Plant Cell 25, 1143–1157 (2013).
Naveed, Z. A., Wei, X., Chen, J., Mubeen, H. & Ali, G. S. The PTI to ETI continuum in Phytophthora–plant interactions. Front. Plant Sci. 11, 593905 (2020).
Ngou, B. P. M., Ahn, H. K., Ding, P. & Jones, J. D. G. Mutual potentiation of plant immunity by cell-surface and intracellular receptors. Nature 592, 110–115 (2021).
Yuan, M. et al. Pattern-recognition receptors are required for NLR-mediated plant immunity. Nature 592, 105–109 (2021).
Zhai, K. et al. NLRs guard metabolism to coordinate pattern- and effector-triggered immunity. Nature https://doi.org/10.1038/s41586-021-04219-2 (2021).
Wu, Y. J. et al. Loss of the common immune coreceptor BAK1 leads to NLR-dependent cell death. Proc. Natl Acad. Sci. USA 117, 27044–27053 (2020).
Veronese, P. et al. The membrane-anchored BOTRYTIS-INDUCED KINASE1 plays distinct roles in Arabidopsis resistance to necrotrophic and biotrophic pathogens. Plant Cell 18, 257–273 (2006).
Mora-Garcia, S. et al. Nuclear protein phosphatases with Kelch-repeat domains modulate the response to brassinosteroids in Arabidopsis. Genes Dev. 18, 448–460 (2004).
Maselli, G. A. et al. Revisiting the evolutionary history and roles of protein phosphatases with Kelch-like domains in plants. Plant Physiol. 164, 1527–1541 (2014).
Kim, T. W., Michniewicz, M., Bergmann, D. C. & Wang, Z. Y. Brassinosteroid regulates stomatal development by GSK3-mediated inhibition of a MAPK pathway. Nature 482, 419–422 (2012).
Turnbull, D. et al. AVR2 targets BSL family members, which act as susceptibility factors to suppress host immunity. Plant Physiol. 180, 571–581 (2019).
Zipfel, C. et al. Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium-mediated transformation. Cell 125, 749–760 (2006).
Asai, T. et al. MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415, 977–983 (2002).
Bi, G. et al. Receptor-like cytoplasmic kinases directly link diverse pattern recognition receptors to the activation of mitogen-activated protein kinase cascades in Arabidopsis. Plant Cell 30, 1543–1561 (2018).
Kadota, Y. et al. Direct regulation of the NADPH oxidase RBOHD by the PRR-associated kinase BIK1 during plant immunity. Mol. Cell 54, 43–55 (2014).
Kong, Q. et al. The MEKK1–MKK1/MKK2–MPK4 kinase cascade negatively regulates immunity mediated by a mitogen-activated protein kinase kinase kinase in Arabidopsis. Plant Cell 24, 2225–2236 (2012).
Lukowitz, W., Roeder, A., Parmenter, D. & Somerville, C. A MAPKK kinase gene regulates extra-embryonic cell fate in Arabidopsis. Cell 116, 109–119 (2004).
Guo, X., Park, C. H., Wang, Z. Y., Nickels, B. E. & Dong, J. A spatiotemporal molecular switch governs plant asymmetric cell division. Nat. Plants 7, 667–680 (2021).
Kim, E. J. et al. Oligomerization between BSU1 family members potentiates brassinosteroid signaling in Arabidopsis. Mol. Plant 9, 178–181 (2016).
Curtis, M. D. & Grossniklaus, U. A gateway cloning vector set for high-throughput functional analysis of genes in planta. Plant Physiol. 133, 462–469 (2003).
Hartwig, T. et al. Propiconazole is a specific and accessible brassinosteroid (BR) biosynthesis inhibitor for Arabidopsis and maize. PLoS ONE https://doi.org/10.1371/journal.pone.0036625 (2012).
Earley, K. W. et al. Gateway-compatible vectors for plant functional genomics and proteomics. Plant J. 45, 616–629 (2006).
Goff, S. A. et al. The iPlant collaborative: cyberinfrastructure for plant biology. Front. Plant Sci. https://doi.org/10.3389/fpls.2011.00034 (2011).
Saeed, A. I. et al. TM4: a free, open-source system for microarray data management and analysis. Biotechniques 34, 374–378 (2003).
Tian, T. et al. agriGO v2.0: a GO analysis toolkit for the agricultural community, 2017 update. Nucleic Acids Res. 45, W122–W129 (2017).
Fan, M. et al. The bHLH transcription factor HBI1 mediates the trade-off between growth and pathogen-associated molecular pattern-triggered immunity in Arabidopsis. Plant Cell 26, 828–841 (2014).
Ishiga, Y., Ishiga, T., Uppalapati, S. R. & Mysore, K. S. Arabidopsis seedling flood-inoculation technique: a rapid and reliable assay for studying plant–bacterial interactions. Plant Methods https://doi.org/10.1186/1746-4811-7-32 (2011).
Guan, S. H., Price, J. C., Prusiner, S. B., Ghaemmaghami, S. & Burlingame, A. L. A data processing pipeline for mammalian proteome dynamics studies using stable isotope metabolic labeling. Mol. Cell. Proteomics https://doi.org/10.1074/mcp.M111.010728 (2011).
MacLean, B. et al. Skyline: an open source document editor for creating and analyzing targeted proteomics experiments. Bioinformatics 26, 966–968 (2010).
He, P. et al. Specific bacterial suppressors of MAMP signaling upstream of MAPKKK in Arabidopsis innate immunity. Cell 125, 563–575 (2006).
Oh, E., Zhu, J. Y. & Wang, Z. Y. Interaction between BZR1 and PIF4 integrates brassinosteroid and environmental responses. Nat. Cell Biol. 14, 802–U864 (2012).
Acknowledgements
We thank J.-M. Zhou for sharing the BIK1::BIK1–HA and MEKK1–FLAG seeds, and L. Shan and P. He for the GST–BIK1 and GST–BIK1Km constructs. We thank A. V. Reyes for generating the bee swarm box plot figures. This research was supported by grants from the NIH (no. R01GM066258 to Z.-Y.W. and no. R01GM135706 to S.-L.X.), the National Research Foundation of Korea funded by the Ministry of Science, ICT, Future Planning (nos NRF-2021R1A2C1006617 and 2020R1A6A1A06046728 to T.-W.K. and no. 2021R1A2C1007516 to S.-K.K.), the NSF (no. NSF-IOS 2026368 to M.B.M.), the Howard Hughes Medical Institute and NIH (no. P41GM103481 to A.L.B.), and the Carnegie Institution for Science endowment fund to the Carnegie MS facility.
Author information
Authors and Affiliations
Contributions
C.H.P., Y.B., T.-W.K. and Z.-Y.W. designed the research. C.H.P. and Y.B. performed most of the experiments. C.H.P. and Y.B. performed the PAMP treatment and immunoblotting. C.H.P. performed RNA-seq. C.H.P. and Y.B. analysed the RNA-seq data. C.H.P., Y.B., J.-H.Y., S.-H.K., S.-K.K. and T.-W.K. performed and analysed the yeast two-hybrid assay and in vitro pull-down assay. S.-H.K., S.-K.K. and T.-W.K. performed the SA quantification. C.H.P., S.-L.X., A.L.B. and R.S. performed the MS analysis. C.H.P., J.-G.K. and M.B.M. performed and analysed the bacterial growth assay. C.H.P., Y.B. and N.Y.X. performed the immunoblot and seedling flood inoculation assay. C.H.P., Y.B., S.-K.K., T.-W.K., M.B.M. and Z.-Y.W. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Gene ontology (GO) analysis of genes differentially expressed in bsu-q.
(a, b) Enrichment of GO annotation in categories of biological process for genes increased (a) and decreased (b) in bsu-q. Blue and orange columns indicate the percent of genes of input (Supplementary Table 2) and reference (TAIR10), respectively. Bold with underlines indicate enriched GO categories of biological process.
Extended Data Fig. 2 Expression of flagellin-induced genes are altered in bsu-q.
(a, b) The bsu-q mutant has defects in flg22-induced gene expression. Heat map (a) and scatter plot (b) of log2 fold change (FC, flg22/mock) values of flg22-responsive genes in wild type (WT) and bsu-q from RNA-seq data. (c) Quantitative RT-PCR of flg22-induced FRK1, At2g17740, ROF2 and CRK13 expression in WT and bsu-q. The relative expression levels are normalized to the PP2A gene expression and to the wild type sample. Asterisks indicate statistically significant difference between mock and flg22 treated samples (two-tail t-test, p = 6.3014E-7, 0.02168, 0.002650, 0.0006797, 1.9289E-5 and 0.003883 from left to right). The graph shows average expression level ± SEM of six biological repeats for FRK1 (n = 6) and three biological repeats for other genes (n = 3).
Extended Data Fig. 3 Loss of BSU1 family impairs flg22 induction of ROS and increases SA level.
(a) Flg22-induced ROS burst in WT and bsu-q. are indicated. Each data point represents the average of relative light unit (RLU) ± SEM of at least 12 replicates from 2 biological repeats (6 samples for each repeat). Each repeat showed similar results. (b) The bsu-i mutant accumulates increased levels of salicylic acid. The SA levels (microgram per gram fresh tissue) of wild type and bsu-i were measured from 0.1 g of 11-day-old seedlings grown on 10 µM estradiol in two biological repeats.
Extended Data Fig. 4 The flg22- and elf18-induced MPK3/6 phosphorylation in the BSU1 family mutants.
(a) Flg22-induced MPK3/6 phosphorylation in wild-type (WT) and bsl2;bsl3. Ten-day-old seedlings were treated with 100 nM flg22 for 10 min. Immunoblot was performed using anti-phospho-p44/42 MAPK and anti-BSL2/3 antibodies. Ponceau S staining shows equal protein loading. (b) Elf18-induced MPK3/6 phosphorylation in bsu-i. Twelve-day-old seedlings grown on 20 µM estradiol containing media were treated with 1 µM elf18 for 10 min. Immunoblot was performed using anti-phospho-p44/42 MAPK and anti-BSL2/3 antibodies. Ponceau S staining shows equal protein loading.
Extended Data Fig. 5 Yeast-two-hybrid assays for interactions between BIK1 family RLCKs and BSU1/BSLs proteins.
Yeast-two-hybrid assay for interaction between BIK1/PBLs and BSU1/BSLs proteins. Yeast cells co-expressing AD-BIK1/PBLs and BD-BSU1/BSLs were grown on synthetic dropout medium containing 2.5 mM 3-amino-1, 2, 4-triazole (3AT).
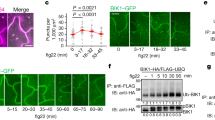
Extended Data Fig. 6 SILIA IP-MS PRM analysis of flg22-induced BSU1 phosphorylation.
(a) Chromatograms of fragment ions from the replicate SILIA IP-MS PRM experiment of Fig. 3c using reciprocal labeling. (b) The ratio of phosphorylated and non-phosphorylated MDSDNVWTPVPAVAPpSPR peptides. The values of peak area were used for quantification. The ratios of S251 phosphorylation between flg22-treated (+flg22) and mock-treated (-flg22) samples are shown.
Extended Data Fig. 7 Alignment of sequence around S251 of BSU1 with those of paralogs.
Amino acid sequences of the indicated species were obtained from Maselli et al., 2014 (22). Asterisks indicate BSU1 S251.
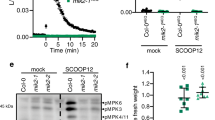
Extended Data Fig. 8 Relative band intensity of Fig. 5a, c.
(a) Relative band intensities of immunoblot images of flg22-treated samples using anti-phospho-p44/42 MAPK antibody (anti-p-MPK) in Fig. 5a were measured by ImageJ. The band intensity of each bsu-i sample was set to 1. The graph shows the average band intensity ± SEM from three repeat experiments. One-tail t-tests show that the band intensities of BSU1;i-1 and BSU1;i-2 are significantly stronger than that of S251A;i (p = 0.0268 and p = 0.0157). (b) Band intensities of immunoblot images, represented by Fig. 5c, were measured by ImageJ. The band intensity of GST-BIN2 was divided by that of MBP-BSU1, and then the ratio between +GST-CDG1 and -GST-CDG1 is presented to show the effect of CDG1 on BSU1-BIN2 binding. The graph shows the average binding activity ± SEM from three repeat experiments. One-tail t-test shows that the binding activity of S251A is significantly higher than that of S764A. (p = 0.041983).
Extended Data Fig. 9 BSU1 protein level of seedlings in Fig. 5d, e.
Total protein was extracted from 7-day old light-grown seedlings. BSU1-YFP protein level was analyzed by immunoblotting using anti-GFP antibody. Ponceau S staining image shows equal protein loading. These transgenic lines were used in Fig. 5d, e.
Supplementary information
Supplementary Data 1
Supplementary Tables 1–6.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 1
Unprocessed gels or blots.
Source Data Fig. 2
Unprocessed gels or blots.
Source Data Fig. 4
Unprocessed gels or blots.
Source Data Fig. 5
Unprocessed gels or blots.
Source Data Fig. 5
Statistical source data.
Extended Data Fig. 2
Statistical source data.
Extended Data Fig. 3
Statistical source data.
Extended Data Fig. 4
Unprocessed gels or blots.
Extended Data Fig. 8
Statistical source data.
Extended Data Fig. 9
Unprocessed gels or blots.
Rights and permissions
About this article
Cite this article
Park, C.H., Bi, Y., Youn, JH. et al. Deconvoluting signals downstream of growth and immune receptor kinases by phosphocodes of the BSU1 family phosphatases. Nat. Plants 8, 646–655 (2022). https://doi.org/10.1038/s41477-022-01167-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-022-01167-1
This article is cited by
-
Brassinosteroids and Salicylic Acid Mutually Enhance Endogenous Content and Signaling to Show a Synergistic Effect on Pathogen Resistance in Arabidopsis thaliana
Journal of Plant Biology (2023)
-
How a single receptor-like kinase exerts diverse roles: lessons from FERONIA
Molecular Horticulture (2022)