Abstract
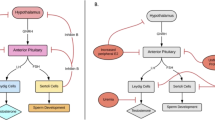
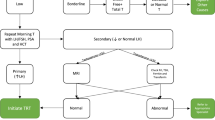
Testosterone deficiency is a prevalent condition that frequently affects individuals with end-stage renal disease (ESRD) and those who have undergone renal transplantation. While the etiology of this condition is complex, its implications in this population are far-reaching, impacting various domains such as endocrine profile, sexual and erectile function, bone mineral density (BMD), anemia, and graft survival following renal transplantation. Herein, we review the most recent literature exploring the pathophysiology of testosterone deficiency in ESRD and renal transplant patients, examining its diverse effects on this demographic, and assessing the advantages of testosterone replacement therapy (TRT). Existing evidence suggests that TRT is a safe intervention in ESRD and renal transplant patients, demonstrating improvements across multiple domains. Despite valuable insights from numerous studies, a critical need persists for larger, high-quality prospective studies to comprehensively grasp the nuances of TRT, especially in this vulnerable population. Proactive screening and treatment of testosterone deficiency may prove beneficial, emphasizing the urgency for further research in this area.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Araujo AB, O’Donnell AB, Brambilla DJ, Simpson WB, Longcope C, Matsumoto AM, et al. Prevalence and incidence of androgen deficiency in middle-aged and older men: estimates from the Massachusetts Male Aging Study. J Clin Endocrinol Metab. 2004;89:5920–6. https://doi.org/10.1210/jc.2003-031719
Mulligan T, Frick MF, Zuraw QC, Stemhagen A, McWhirter C. Prevalence of hypogonadism in males aged at least 45 years: the HIM study. Int J Clin Pract. 2006;60:762–9. https://doi.org/10.1111/j.1742-1241.2006.00992.x
Thirumavalavan N, Scovell JM, Link RE, Lamb DJ, Lipshultz LI. Does solid organ transplantation affect male reproduction? Eur Urol Focus. 2018;4:307–10. https://doi.org/10.1016/j.euf.2018.08.012
Carrero JJ, Qureshi AR, Parini P, Arver S, Lindholm B, Bárány P, et al. Low serum testosterone increases mortality risk among male dialysis patients. J Am Soc Nephrol. 2009;20:613–20. https://doi.org/10.1681/ASN.2008060664
Wachterman MW, O’Hare AM, Rahman O-K, Lorenz KA, Marcantonio ER, Alicante GK, et al. One-year mortality after dialysis initiation among older adults. JAMA Intern Med. 2019;179:987–90. https://doi.org/10.1001/jamainternmed.2019.0125
Kazemeini SM, Mogharabian N, Asadpour A, Naderi G, Kasaeian A, Mousavi A. The effect of renal transplant on hypogonadism and erectile dysfunction due to end-stage renal disease. Saudi J Kidney Dis Transplant. 2021;32:923–8. https://doi.org/10.4103/1319-2442.338303
Akbari F, Alavi M, Esteghamati A, Mehrsai A, Djaladat H, Zohrevand R, et al. Effect of renal transplantation on sperm quality and sex hormone levels. BJU Int. 2003;92:281–3. https://doi.org/10.1046/j.1464-410x.2003.04323.x
Reinhardt W, Kübber H, Dolff S, Benson S, Führer D, Tan S. Rapid recovery of hypogonadism in male patients with end stage renal disease after renal transplantation. Endocrine. 2018;60:159–66. https://doi.org/10.1007/s12020-018-1543-2
Saha M-T, Saha HHT, Niskanen LK, Salmela KT, Pasternack AI. Time course of serum prolactin and sex hormones following successful renal transplantation. Nephron. 2002;92:735–7. https://doi.org/10.1159/000064079
Shoskes DA, Kerr H, Askar M, Goldfarb DA, Schold J. Low testosterone at time of transplantation is independently associated with poor patient and graft survival in male renal transplant recipients. J Urol. 2014;192:1168–71. https://doi.org/10.1016/j.juro.2014.03.102
UNOS Data and Transplant Statistics | Organ Donation Data. UNOS. https://unos.org/data/. Accessed 3 February 2024.
Lincoff AM, Bhasin S, Flevaris P, Mitchell LM, Basaria S, Boden WE, et al. Cardiovascular safety of testosterone-replacement therapy. N Engl J Med. 2023;389:107–17. https://doi.org/10.1056/NEJMoa2215025
Antonucci M, Palermo G, Recupero SM, Bientinesi R, Presicce F, Foschi N, et al. Male sexual dysfunction in patients with chronic end-stage renal insufficiency and in renal transplant recipients. Arch Ital Urol Androl. 2016;87:299–305. https://doi.org/10.4081/aiua.2015.4.299
Miner MM, Khera M, Bhattacharya RK, Blick G, Kushner H. Baseline data from the TRiUS registry: symptoms and comorbidities of testosterone deficiency. Postgrad Med. 2011;123:17–27. https://doi.org/10.3810/pgm.2011.05.2280
Cha J, Han D. Health-related quality of life based on comorbidities among patients with end-stage renal disease. Osong Public Health Res Perspect. 2020;11:194–200. https://doi.org/10.24171/j.phrp.2020.11.4.08
Albaaj F, Sivalingham M, Haynes P, McKinnon G, Foley RN, Waldek S, et al. Prevalence of hypogonadism in male patients with renal failure. Postgrad Med J. 2006;82:693–6. https://doi.org/10.1136/pgmj.2006.045963
Balcázar-Hernández L, Mendoza-Zubieta V, González-Virla B, González-García B, Osorio-Olvera M, Peñaloza-Juarez JU, et al. Hypothalamic-pituitary-gonadal axis disturbance and its association with insulin resistance in kidney transplant recipients. J Bras Nefrol. 2023;45:77–83. https://doi.org/10.1590/2175-8239-JBN-2021-0250en
Stenvinkel P. Inflammation in end-stage renal disease: the hidden enemy. Nephrology. 2006;11:36–41. https://doi.org/10.1111/j.1440-1797.2006.00541.x
Wibullaksanakul S, Handelsman DJ. Regulation of hypothalamic gonadotropin-releasing hormone secretion in experimental uremia: in vitro studies. Neuroendocrinology. 1991;54:353–8. https://doi.org/10.1159/000125913
Dong QH, Handelsman DJ. Regulation of pulsatile luteinizing hormone secretion in experimental uremia. Endocrinology. 1991;128:1218–22. https://doi.org/10.1210/endo-128-3-1218
Lim VS, Henriquez C, Sievertsen G, Frohman LA. Ovarian function in chronic renal failure: evidence suggesting hypothalamic anovulation. Ann Intern Med. 1980;93:21–7. https://doi.org/10.7326/0003-4819-93-1-21
Prem AR, Punekar SV, Kalpana M, Kelkar AR, Acharya VN. Male reproductive function in uraemia: efficacy of haemodialysis and renal transplantation. Br J Urol. 1996;78:635–8. https://doi.org/10.1046/j.1464-410x.1996.14624.x
Levitan D, Moser SA, Goldstein DA, Kletzky OA, Lobo RA, Massry SG. Disturbances in the hypothalamic-pituitary-gonadal axis in male patients with acute renal failure. Am J Nephrol. 1984;4:99–106. https://doi.org/10.1159/000166785
Distiller LA, Morley JE, Sagel J, Pokroy M, Rabkin R. Pituitary-gonadal function in chronic renal failure: the effect of luteinizing hormone-releasing hormone and the influence of dialysis. Metabolism. 1975;24:711–20. https://doi.org/10.1016/0026-0495(75)90039-6
Biasioli S, Mazzali A, Foroni R, D’Andrea G, Feriani M, Chiaramonte S, et al. Chronobiological variations of prolactin (PRL) in chronic renal failure (CRF). Clin Nephrol. 1988;30:86–92.
Carrero JJ, Kyriazis J, Sonmez A, Tzanakis I, Qureshi AR, Stenvinkel P, et al. Prolactin levels, endothelial dysfunction, and the risk of cardiovascular events and mortality in patients with CKD. Clin J Am Soc Nephrol. 2012;7:207–15. https://doi.org/10.2215/CJN.06840711
Adachi N, Lei B, Deshpande G, Seyfried FJ, Shimizu I, Nagaro T, et al. Uraemia suppresses central dopaminergic metabolism and impairs motor activity in rats. Intensive Care Med. 2001;27:1655–60. https://doi.org/10.1007/s001340101067
Sievertsen GD, Lim VS, Nakawatase C, Frohman LA. Metabolic clearance and secretion rates of human prolactin in normal subjects and in patients with chronic renal failure. J Clin Endocrinol Metab. 1980;50:846–52. https://doi.org/10.1210/jcem-50-5-846
Carrero JJ, Qureshi AR, Nakashima A, Arver S, Parini P, Lindholm B, et al. Prevalence and clinical implications of testosterone deficiency in men with end-stage renal disease. Nephrol Dial Transplant. 2011;26:184–90. https://doi.org/10.1093/ndt/gfq397
Foresta C, Caretta N, Lana A, De Toni L, Biagioli A, Ferlin A, et al. Reduced number of circulating endothelial progenitor cells in hypogonadal men. J Clin Endocrinol Metab. 2006;91:4599–602. https://doi.org/10.1210/jc.2006-0763
Nilsson E, Stenvinkel P, Liu S, Stedman MR, Chertow GM, Floege J. Serum testosterone concentrations and outcomes in hemodialysis patients enrolled in the EVOLVE trial. Nephrol Dial Transplant. 2023;38:1519–27. https://doi.org/10.1093/ndt/gfac278
Rymarz A, Matyjek A, Gomółka M, Niemczyk S. Lean tissue index and body cell mass can be predictors of low free testosterone levels in men on hemodialysis. J Ren Nutr. 2019;29:529–35. https://doi.org/10.1053/j.jrn.2019.03.078
Cobo G, Gallar P, Di Gioia C, García Lacalle C, Camacho R, Rodriguez I, et al. Hypogonadism associated with muscle atrophy, physical inactivity and ESA hyporesponsiveness in men undergoing haemodialysis. Nefrologia. 2017;37:54–60. https://doi.org/10.1016/j.nefro.2016.04.009
Park MG, Koo HS, Lee B. Characteristics of testosterone deficiency syndrome in men with chronic kidney disease and male renal transplant recipients: a cross-sectional study. Transplant Proc. 2013;45:2970–4. https://doi.org/10.1016/j.transproceed.2013.08.087
Skiba R, Rymarz A, Matyjek A, Dymus J, Woźniak-Kosek A, Syryło T, et al. Testosterone replacement therapy in chronic kidney disease patients. Nutrients. 2022;14:3444. https://doi.org/10.3390/nu14163444
Mulhall JP, Trost LW, Brannigan RE, Kurtz EG, Redmon JB, Chiles KA, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200:423–32. https://doi.org/10.1016/j.juro.2018.03.115
Brock G, Heiselman D, Maggi M, Kim SW, Rodríguez Vallejo JM, Behre HM, et al. Effect of testosterone solution 2% on testosterone concentration, sex drive and energy in hypogonadal men: results of a placebo controlled study. J Urol. 2016;195:699–705. https://doi.org/10.1016/j.juro.2015.10.083
Maggi M, Heiselman D, Knorr J, Iyengar S, Paduch DA, Donatucci CF. Impact of testosterone solution 2% on ejaculatory dysfunction in hypogonadal men. J Sex Med. 2016;13:1220–6. https://doi.org/10.1016/j.jsxm.2016.05.012
Roy CN, Snyder PJ, Stephens-Shields AJ, Artz AS, Bhasin S, Cohen HJ, et al. Association of testosterone levels with anemia in older men: a controlled clinical trial. JAMA Intern Med. 2017;177:480–90. https://doi.org/10.1001/jamainternmed.2016.9540
Svartberg J, Agledahl I, Figenschau Y, Sildnes T, Waterloo K, Jorde R. Testosterone treatment in elderly men with subnormal testosterone levels improves body composition and BMD in the hip. Int J Impot Res. 2008;20:378–87. https://doi.org/10.1038/ijir.2008.19
Cherrier MM, Anderson K, Shofer J, Millard S, Matsumoto AM. Testosterone treatment of men with mild cognitive impairment and low testosterone levels. Am J Alzheimers Dis Other Demen. 2015;30:421–30. https://doi.org/10.1177/1533317514556874
Vaughan C, Goldstein FC, Tenover JL. Exogenous testosterone alone or with finasteride does not improve measurements of cognition in healthy older men with low serum testosterone. J Androl. 2007;28:875–82. https://doi.org/10.2164/jandrol.107.002931
Grossmann M, Hoermann R, Wittert G, Yeap BB. Effects of testosterone treatment on glucose metabolism and symptoms in men with type 2 diabetes and the metabolic syndrome: a systematic review and meta-analysis of randomized controlled clinical trials. Clin Endocrinol. 2015;83:344–51. https://doi.org/10.1111/cen.12664
Barton CH, Mirahmadi MK, Vaziri ND. Effects of long-term testosterone administration on pituitary-testicular axis in end-stage renal failure. Nephron. 1982;31:61–4. https://doi.org/10.1159/000182618
van Coevorden A, Stolear JC, Dhaene M, van Herweghem JL, Mockel J. Effect of chronic oral testosterone undecanoate administration on the pituitary-testicular axes of hemodialyzed male patients. Clin Nephrol. 1986;26:48–54.
Pechersky AV, Mazurov VI, Semiglazov VF, Karpischenko AI, Mikhailichenko VV, Udintsev AV. Androgen administration in middle-aged and ageing men: effects of oral testosterone undecanoate on dihydrotestosterone, oestradiol and prostate volume. Int J Androl. 2002;25:119–25. https://doi.org/10.1046/j.1365-2605.2002.00335.x
Pizzol D, Xiao T, Yang L, Demurtas J, McDermott D, Garolla A, et al. Prevalence of erectile dysfunction in patients with chronic kidney disease: a systematic review and meta-analysis. Int J Impot Res. 2021;33:508–15. https://doi.org/10.1038/s41443-020-0295-8
Lawrence IG, Price DE, Howlett TA, Harris KP, Feehally J, Walls J. Correcting impotence in the male dialysis patient: experience with testosterone replacement and vacuum tumescence therapy. Am J Kidney Dis. 1998;31:313–9. https://doi.org/10.1053/ajkd.1998.v31.pm9469503
Chatterjee R, Wood S, McGarrigle HH, Lees WR, Ralph DJ, Neild GH. A novel therapy with testosterone and sildenafil for erectile dysfunction in patients on renal dialysis or after renal transplantation. J Fam Plann Reprod Health Care. 2004;30:88–90. https://doi.org/10.1783/147118904322995438
Cunningham G, Belkoff L, Brock G, Efros M, Gittelman M, Carrara D, et al. Efficacy and safety of a new topical testosterone replacement gel therapy for the treatment of male hypogonadism. Endocr Pract. 2017;23:557–65. https://doi.org/10.4158/EP161665.OR
Gronski MA, Grober ED, Gottesman IS, Ormsby RW, Bryson N. Efficacy of nasal testosterone gel (Natesto®) stratified by baseline endogenous testosterone levels. J Endocr Soc. 2019;3:1652–62. https://doi.org/10.1210/js.2019-00183
Raynaud J-P, Legros J-J, Rollet J, Augès M, Bunouf P, Sournac M, et al. Efficacy and safety of a new testosterone-in-adhesive matrix patch applied every 2 days for 1 year to hypogonadal men. J Steroid Biochem Mol Biol. 2008;109:168–76. https://doi.org/10.1016/j.jsbmb.2007.10.010
Kresch E, Lima TFN, Molina M, Deebel NA, Reddy R, Patel M, et al. Efficacy and safety outcomes of a compounded testosterone pellet versus a branded testosterone pellet in men with testosterone deficiency: a single-center, open-label, randomized trial. Sex Med. 2023;11:qfad007. https://doi.org/10.1093/sexmed/qfad007
Dinsmore WW, Wyllie MG. The long‐term efficacy and safety of a testosterone mucoadhesive buccal tablet in testosterone‐deficient men. BJU Int. 2012;110:162–9. https://doi.org/10.1111/j.1464-410X.2011.10837.x
Cangüven O, Aykose G, Albayrak S, Goktas C, Horuz R, Yencilek F. Efficacy of testosterone gel in the treatment of erectile dysfunction in hypogonadal hemodialysis patients: a pilot study. Int J Impot Res. 2010;22:140–5. https://doi.org/10.1038/ijir.2009.55
Cervi A, Balitsky AK. Testosterone use causing erythrocytosis. CMAJ. 2017;189:E1286–8. https://doi.org/10.1503/cmaj.170683
Brockenbrough AT, Dittrich MO, Page ST, Smith T, Stivelman JC, Bremner WJ. Transdermal androgen therapy to augment EPO in the treatment of anemia of chronic renal disease. Am J Kidney Dis. 2006;47:251–62. https://doi.org/10.1053/j.ajkd.2005.10.022
Inoue Y, Nakamura K, Kuwahara Y, Lu Y, Masuda N, Horie S, et al. Efficacy of testosterone treatment in hemodialysis patients as assessed by aging males’ symptoms scores: a pilot study. Am J Mens Health. 2018;12:1541–7. https://doi.org/10.1177/1557988318772734
Pencina KM, Travison TG, Artz AS, Lincoff AM, Nissen SE, Flevaris P, et al. Efficacy of testosterone replacement therapy in correcting anemia in men with hypogonadism: a randomized clinical trial. JAMA Netw Open. 2023;6:e2340030 https://doi.org/10.1001/jamanetworkopen.2023.40030
Aramwit P, Palapinyo S, Wiwatniwong S, Supasyndh O. The efficacy of oxymetholone in combination with erythropoietin on hematologic parameters and muscle mass in CAPD patients. Int J Clin Pharmacol Ther. 2010;48:803–13. https://doi.org/10.5414/cpp48803
Gaughan WJ, Liss KA, Dunn SR, Mangold AM, Buhsmer JP, Michael B, et al. A 6-month study of low-dose recombinant human erythropoietin alone and in combination with androgens for the treatment of anemia in chronic hemodialysis patients. Am J Kidney Dis. 1997;30:495–500. https://doi.org/10.1016/s0272-6386(97)90307-3
Dhindsa S, Nargunan V, Reddy A, Clubb C, Haider A, Cheng F, et al. Testosterone decreases erythropoietin stimulating agent use in men on hemodialysis. Androg Clin Res Ther. 2020;1:32–9. https://doi.org/10.1089/andro.2020.0003
Yeo S, Holl K, Peñaherrera N, Wissinger U, Anstee K, Wyn R. Burden of male hypogonadism and major comorbidities, and the clinical, economic, and humanistic benefits of testosterone therapy: a narrative review. Clin Outcomes Res. 2021;13:31–8. https://doi.org/10.2147/CEOR.S285434
Majzoub A, Shoskes DA. A case series of the safety and efficacy of testosterone replacement therapy in renal failure and kidney transplant patients. Transl Androl Urol. 2016;5:814–8. https://doi.org/10.21037/tau.2016.07.09
Yang Q, Abudou M, Xie XS, Wu T. Androgens for the anaemia of chronic kidney disease in adults. Cochrane Database Syst Rev. 2014;2014:CD006881. https://doi.org/10.1002/14651858.CD006881.pub2
Jørgensen HS, Winther S, Bøttcher M, Hauge E-M, Rejnmark L, Svensson M, et al. Bioavailable testosterone is positively associated with bone mineral density in male kidney transplantation candidates. Kidney Int Rep. 2018;3:661–70. https://doi.org/10.1016/j.ekir.2018.01.009
Yeo JK, Koo HS, Yu J, Park MG. Effects of testosterone treatment on quality of life in patients with chronic kidney disease. Am J Mens Health. 2020;14:1557988320917258. https://doi.org/10.1177/1557988320917258
Grossmann M, Hoermann R, Ng Tang Fui M, Zajac JD, Ierino FL, Roberts MA. Sex steroids levels in chronic kidney disease and kidney transplant recipients: associations with disease severity and prediction of mortality. Clin Endocrinol. 2015;82:767–75. https://doi.org/10.1111/cen.12656
Barroso LVS, Miranda EP, Cruz NI, Medeiros MaS, Araújo ACO, Mota Filho FHA, et al. Analysis of sexual function in kidney transplanted men. Transplant Proc. 2008;40:3489–91. https://doi.org/10.1016/j.transproceed.2008.07.141
Al Khallaf HH. Analysis of sexual functions in male nondiabetic hemodialysis patients and renal transplant recipients. Transplant Int. 2010;23:176–81. https://doi.org/10.1111/j.1432-2277.2009.00972.x
Shamsa A, Motavalli SM, Aghdam B. Erectile function in end-stage renal disease before and after renal transplantation. Transplant Proc. 2005;37:3087–9. https://doi.org/10.1016/j.transproceed.2005.08.067
Rahman IA, Rasyid N, Birowo P, Atmoko W. Effects of renal transplantation on erectile dysfunction: a systematic review and meta-analysis. Int J Impot Res. 2022;34:456–66. https://doi.org/10.1038/s41443-021-00419-6
Pyrgidis N, Mykoniatis I, Sokolakis I, Minopoulou I, Nigdelis MP, Sountoulides P, et al. Renal transplantation improves erectile function in patients with end-stage renal disease: a systematic review and meta-analysis. J Urol. 2021;205:1009–17. https://doi.org/10.1097/JU.0000000000001577
Thirumavalavan N, Scovell JM, Lo E, Gondokusomo J, Khera M, Pastuszak AW, et al. Is treatment of hypogonadism safe for men after a solid organ transplant? Results from a retrospective controlled cohort study. Int J Impot Res. 2022;34:50–4. https://doi.org/10.1038/s41443-020-00361-z
Stauffer ME, Fan T. Prevalence of anemia in chronic kidney disease in the United States. PLoS ONE. 2014;9:e84943. https://doi.org/10.1371/journal.pone.0084943
Foley RN, Parfrey PS, Harnett JD, Kent GM, Murray DC, Barre PE. The impact of anemia on cardiomyopathy, morbidity, and mortality in end-stage renal disease. Am J Kidney Dis. 1996;28:53–61. https://doi.org/10.1016/s0272-6386(96)90130-4
Akaishi M, Hiroe M, Hada Y, Suzuki M, Tsubakihara Y, Akizawa T. et al. Effect of anemia correction on left ventricular hypertrophy in patients with modestly high hemoglobin level and chronic kidney disease. J Cardiol. 2013;62:249–56. https://doi.org/10.1016/j.jjcc.2013.04.008
Vecchio M, Navaneethan SD, Johnson DW, Lucisano G, Graziano G, Saglimbene V, et al. Interventions for treating sexual dysfunction in patients with chronic kidney disease. Cochrane Database Syst Rev: CD007747. 2010. https://doi.org/10.1002/14651858.CD007747.pub2
Author information
Authors and Affiliations
Contributions
Research conception and design: ND. Drafting of the manuscript: all authors. Critical revision of the manuscript: all authors. Supervision: RR Approval of the final manuscript: all authors.
Corresponding author
Ethics declarations
Competing interests
Dr. Nannan Thirumavalavan is the recipient of the 2021 Young Clinicians Grant from the Sexual Medicine Society of North America. We otherwise declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deebel, N.A., Matthew, A.N., Loloi, J. et al. Testosterone deficiency in men with end stage renal disease and kidney transplantation: a narrative review. Int J Impot Res (2024). https://doi.org/10.1038/s41443-024-00890-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41443-024-00890-x