Abstract
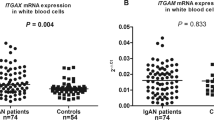
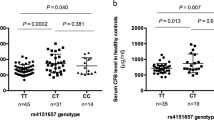
IgA Nephropathy (IgAN) is one of the most common causes of chronic kidney damage worldwide. Identifying new genetic factors associated with IgAN risk is of invaluable importance. To explore the association between polymorphisms of IL-23R and IL-17A and the susceptibility of IgAN, 164 IgAN patients and 192 healthy controls were genotyped for five SNPs in a Chinese Han population. A comparative analysis between genotype distributions, clinical indexes and pathological grades in the IgAN patients was also performed. The GG genotype and a G allele of rs7517847 were associated with a decreased IgAN risk (OR: 0.545; 95% CI: 0.299–0.993; p = 0.046; OR: 0.730; 95% CI: 0.541–0.984; p = 0.039) compared to the TT genotype and T allele respectively. Furthermore, the AA genotype of rs2275913 appeared to reduce the IgAN risk (OR: 0.405; 95% CI: 0.209–0.786; p = 0.007) compared to the GG genotype. Consistently, individuals harboring an AA genotype had a lower IgAN risk (OR: 0.380; 95% CI: 0.211–0.686; p = 0.001) under the recessive model. Our study demonstrated for the first time the significant associations of rs7517847 in IL-23R and rs2275913 in IL-17A with the risk of IgAN in Chinese Han.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Wyatt RJ, Julian BA. IgA nephropathy. N Engl J Med. 2013;368:2402–14.
Suzuki H, Fan R, Zhang Z, Brown R, Hall S, Julian BA, et al. Aberrantly glycosylated IgA1 in IgA nephropathy patients is recognized by IgG antibodies with restricted heterogeneity. J Clin Invest. 2009;119:1668–77.
Reid S, Cawthon PM, Craig JC, Samuels JA, Molony DA, Strippoli GF. Non-immunosuppressive treatment for IgA nephropathy. Cochrane Database Syst Rev. 2011;3:03962.
Geddes CC, Rauta V, Gronhagen-Riska C, Bartosik LP, Jardine AG, Ibels LS, et al. A tricontinental view of IgA nephropathy. Nephrol Dial Transpl. 2003;18:1541–8.
Koyama A, Igarashi M, Kobayashi M. Natural history and risk factors for immunoglobulin A nephropathy in Japan. Research Group on Progressive Renal Diseases. Am J Kidney Dis. 1997;29:526–32.
Gharavi AG, Kiryluk K, Choi M, Li Y, Hou P, Xie J, et al. Genome-wide association study identifies susceptibility loci for IgA nephropathy. Nat Genet. 2011;43:321–7.
Yu XQ, Li M, Zhang H, Low HQ, Wei X, Wang JQ, et al. A genome-wide association study in Han Chinese identifies multiple susceptibility loci for IgA nephropathy. Nat Genet. 2011;44:178–82.
Sallusto F, Baggiolini M. Chemokines and leukocyte traffic. Nat Immunol. 2008;9:949–52.
Cua DJ, Tato CM. Innate IL-17-producing cells: the sentinels of the immune system. Nat Rev Immunol. 2010;10:479–89.
Van Kooten C, Boonstra JG, Paape ME, Fossiez F, Banchereau J, Lebecque S, et al. Interleukin-17 activates human renal epithelial cells in vitro and is expressed during renal allograft rejection. J Am Soc Nephrol. 1998;9:1526–34.
Paust HJ, Turner JE, Steinmetz OM, Peters A, Heymann F, Holscher C, et al. The IL-23/Th17 axis contributes to renal injury in experimental glomerulonephritis. J Am Soc Nephrol. 2009;20:969–79.
Iyoda M, Shibata T, Kawaguchi M, Hizawa N, Yamaoka T, Kokubu F, et al. IL-17A and IL-17F stimulate chemokines via MAPK pathways (ERK1/2 and p38 but not JNK) in mouse cultured mesangial cells: synergy with TNF-alpha and IL-1beta. Am J Physiol Ren Physiol. 2010;298:F779–87.
Kitching AR, Holdsworth SR. The emergence of TH17 cells as effectors of renal injury. J Am Soc Nephrol. 2011;22:235–8.
Gan PY, Steinmetz OM, Tan DS, O’Sullivan KM, Ooi JD, Iwakura Y, et al. Th17 cells promote autoimmune anti-myeloperoxidase glomerulonephritis. J Am Soc Nephrol. 2010;21:925–31.
Lin JR, Wen J, Zhang H, Wang L, Gou FF, Yang M, et al. Interleukin-17 promotes the production of underglycosylated IgA1 in DAKIKI cells. Ren Fail. 2018;40:60–7.
Wong CK, Ho CY, Li EK, Lam CW. Elevation of proinflammatory cytokine (IL-18, IL-17, IL-12) and Th2 cytokine (IL-4) concentrations in patients with systemic lupus erythematosus. Lupus. 2000;9:589–93.
Doreau A, Belot A, Bastid J, Riche B, Trescol-Biemont MC, Ranchin B, et al. Interleukin 17 acts in synergy with B cell-activating factor to influence B cell biology and the pathophysiology of systemic lupus erythematosus. Nat Immunol. 2009;10:778–85.
Watorek E, Klinger M. IL-17A as a potential biomarker of IgA nephropathy. Pol Arch Med Wewn. 2015;125:204–6.
Ruszkowski J, Lisowska KA, Pindel M, Heleniak Z, Debska-Slizien A, Witkowski JM. T cells in IgA nephropathy: role in pathogenesis, clinical significance and potential therapeutic target. Clin Exp Nephrol. 2019;23:291–303.
Kuka M, Baronio R, Valentini S, Monaci E, Muzzi A, Aprea S, et al. Src kinases are required for a balanced production of IL-12/IL-23 in human dendritic cells activated by Toll-like receptor agonists. PLoS ONE. 2010;5:e11491.
Miller JM, Bidula SM, Jensen TM, Reiss CS. Vesicular stomatitis virus modified with single chain IL-23 exhibits oncolytic activity against tumor cells in vitro and in vivo. Int J Interferon Cytokine Mediat Res. 2010;2010:63–72.
Peral de Castro C, Jones SA, Ni Cheallaigh C, Hearnden CA, Williams L, Winter J, et al. Autophagy regulates IL-23 secretion and innate T cell responses through effects on IL-1 secretion. J Immunol. 2012;189:4144–53.
McGeachy MJ, Chen Y, Tato CM, Laurence A, Joyce-Shaikh B, Blumenschein WM, et al. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol. 2009;10:314–24.
Parham C, Chirica M, Timans J, Vaisberg E, Travis M, Cheung J, et al. A receptor for the heterodimeric cytokine IL-23 is composed of IL-12Rbeta1 and a novel cytokine receptor subunit, IL-23R. J Immunol. 2002;168:5699–708.
Gaffen SL, Jain R, Garg AV, Cua DJ. The IL-23-IL-17 immune axis: from mechanisms to therapeutic testing. Nat Rev Immunol. 2014;14:585–600.
Duerr RH, Taylor KD, Brant SR, Rioux JD, Silverberg MS, Daly MJ, et al. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science. 2006;314:1461–3.
Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, et al. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol. 2005;6:1123–32.
Langrish CL, Chen Y, Blumenschein WM, Mattson J, Basham B, Sedgwick JD, et al. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J Exp Med. 2005;201:233–40.
McInnes IB, Mease PJ, Ritchlin CT, Rahman P, Gottlieb AB, Kirkham B, et al. Secukinumab sustains improvement in signs and symptoms of psoriatic arthritis: 2 year results from the phase 3 FUTURE 2 study. Rheumatology. 2017;56:1993–2003.
Abdulahad WH, Stegeman CA, Limburg PC, Kallenberg CG. Skewed distribution of Th17 lymphocytes in patients with Wegener’s granulomatosis in remission. Arthritis Rheum. 2008;58:2196–205.
Nogueira E, Hamour S, Sawant D, Henderson S, Mansfield N, Chavele KM, et al. Serum IL-17 and IL-23 levels and autoantigen-specific Th17 cells are elevated in patients with ANCA-associated vasculitis. Nephrol Dial Transpl. 2010;25:2209–17.
Zhang Z, Kyttaris VC, Tsokos GC. The role of IL-23/IL-17 axis in lupus nephritis. J Immunol. 2009;183:3160–9.
Kyttaris VC, Zhang Z, Kuchroo VK, Oukka M, Tsokos GC. Cutting edge: IL-23 receptor deficiency prevents the development of lupus nephritis in C57BL/6-lpr/lpr mice. J Immunol. 2010;184:4605–9.
Ma YC, Zuo L, Chen JH, Luo Q, Yu XQ, Li Y, et al. Modified glomerular filtration rate estimating equation for Chinese patients with chronic kidney disease. J Am Soc Nephrol. 2006;17:2937–44.
Shi YY, He L. SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci. Cell Res. 2005;15:97–8.
Lee HS, Lee MS, Lee SM, Lee SY, Lee ES, Lee EY, et al. Histological grading of IgA nephropathy predicting renal outcome: revisiting H. S. Lee’s glomerular grading system. Nephrol Dial Transpl. 2005;20:342–8.
Working Group of the International Ig ANN, the Renal Pathology Society, Cattran DC, Coppo R, Cook HT, Feehally J, et al. The Oxford classification of IgA nephropathy: rationale, clinicopathological correlations, and classification. Kidney Int. 2009;76:534–45.
McGeachy MJ, Cua DJ, Gaffen SL. The IL-17 family of cytokines in health and disease. Immunity. 2019;50:892–906.
Peng Z, Tian J, Cui X, Xian W, Sun H, Li E, et al. Increased number of Th22 cells and correlation with Th17 cells in peripheral blood of patients with IgA nephropathy. Hum Immunol. 2013;74:1586–91.
Lin FJ, Jiang GR, Shan JP, Zhu C, Zou J, Wu XR. Imbalance of regulatory T cells to Th17 cells in IgA nephropathy. Scand J Clin Lab Invest. 2012;72:221–9.
Cargill M, Schrodi SJ, Chang M, Garcia VE, Brandon R, Callis KP, et al. A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am J Hum Genet. 2007;80:273–90.
Nunez C, Dema B, Cenit MC, Polanco I, Maluenda C, Arroyo R, et al. IL23R: a susceptibility locus for celiac disease and multiple sclerosis? Genes Immun. 2008;9:289–93.
Yang B, Xu Y, Liu X, Huang Z, Wang L. IL-23R and IL-17A polymorphisms correlate with susceptibility of ankylosing spondylitis in a Southwest Chinese population. Oncotarget. 2017;8:70310–6.
Li Y, Mao Q, Shen L, Tian Y, Yu C, Zhu WM, et al. Interleukin-23 receptor genetic polymorphisms and Crohn’s disease susceptibility: a meta-analysis. Inflamm Res. 2010;59:607–14.
Dong H, Li Q, Zhang Y, Tan W, Jiang Z. IL23R gene confers susceptibility to ankylosing spondylitis concomitant with uveitis in a Han Chinese population. PLoS ONE. 2013;8:e67505.
Liu S, He H, Yu R, Han L, Wang C, Cui Y, et al. The rs7517847 polymorphism in the IL-23R gene is associated with gout in a Chinese Han male population. Mod Rheumatol. 2015;25:449–52.
Fischer S, Kovesdi E, Magyari L, Csongei V, Hadzsiev K, Melegh B, et al. IL23R single nucleotide polymorphisms could be either beneficial or harmful in ulcerative colitis. World J Gastroenterol. 2017;23:447–54.
Xu WD, Xie QB, Zhao Y, Liu Y. Association of Interleukin-23 receptor gene polymorphisms with susceptibility to Crohn’s disease: a meta-analysis. Sci Rep. 2015;5:18584.
Zhong L, Wang W, Song H. Complex role of IL-23R polymorphisms on ankylosing spondylitis: a meta-analysis. Expert Rev Clin Immunol. 2018;14:635–43.
Zhai C, Li S, Feng W, Shi W, Wang J, Wang Q, et al. Association of interleukin-17a rs2275913 gene polymorphism and asthma risk: a meta-analysis. Arch Med Sci. 2018;14:1204–11.
Gao JF, Zhang H, Lv J, Wang L, Fan YY. Associations of the IL-17A rs2275913 and IL-17F rs763780 polymorphisms with the risk of digestive system neoplasms: a meta-analysis. Int Immunopharmacol. 2019;67:248–59.
Tabatabaei-Panah PS, Moravvej H, Delpasand S, Jafari M, Sepehri S, Abgoon R, et al. IL12B and IL23R polymorphisms are associated with alopecia areata. Genes Immun. 2020;21:203–10.
Haouami Y, Dhaouadi T, Sfar I, Bacha M, Gargah T, Bardi R, et al. The role of IL-23/IL-17 axis in human kidney allograft rejection. J Leukoc Biol. 2018;104:1229–39.
Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 2007;447:661–78.
Boyd JK, Barratt J. Inherited IgA glycosylation pattern in IgA nephropathy and HSP nephritis: where do we go next? Kidney Int. 2011;80:8–10.
Acknowledgements
We thank Ruyu Ren and Mengyuan Lyu for assistance in statistical analysis.
Funding
This work was sponsored by the National Natural Science Foundation of China [81902129] and Science and Technology Agency of Sichuan Province [2019YFS0313 and 2019YFS0321].
Author information
Authors and Affiliations
Contributions
HL, ZH, JZ, and BY designed the study; ZH and JZ were responsible for recruitment of subjects; HL, ZH, and JZ performed experiments and conducted data management; HL and BY performed statistical analyses and interpreted results; HL wrote the manuscript. All authors revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Liao, H., Huang, Z., Zhang, J. et al. Association of genetic polymorphisms in IL-23R and IL-17A with the susceptibility to IgA nephropathy in a Chinese Han population. Genes Immun 23, 33–41 (2022). https://doi.org/10.1038/s41435-021-00160-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41435-021-00160-6