Abstract
Objective
The aim of this work is to evaluate the extent to which the eye’s curvature deformation, due to changes in the intraocular pressure (IOP), can be directly tracked by an overlying contact lens.
Method
In this experimental study, using 12 cadaveric eyes, the IOP was increased from 10 to 36 mmHg, while video imaging was used to capture the three experimental variations. The deformation of the bare eye was used as a control, while the deformation of an overlying silicone grided contact lens and an overlying microfluidic IOP-sensing contact lens were examined and compared.
Results
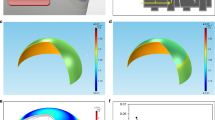
The relation between the slope of the radius of corneal curvature versus the IOP for both the bare eye and the marker contact lens yielded a linear relationship with a R2 value of 0.83. The microfluidic contact lens resulted in an average performance of 0.40 mm indicator movement/mmHg (SD 0.006). Comparing the slope of the marker contact lens deformation, to the performance of the microfluidic contact lens resulted in a R2 value of 0.78. The strain map of the overlaying grided contact lens showed most deformation occurring along the outer edge of the lens with increased deformation as increase IOP occurs; as well as with some negative, compressive movement near the central points.
Conclusion
The deformation from the curvature of the eye is significant enough from 10 to 36 mmHg that a silicone contact lens can capture and mimic those changes. The results show promise for optimization in contact lens-based IOP monitoring.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Kerrigan–Baumrind LA, Quigley HA, Pease ME, Kerrigan DF, Mitchell RS. Number of ganglion cells in glaucoma eyes compared with threshold visual field tests in the same persons. Investig Ophthalmol Vis Sci. 2000;41:741–8.
World Glaucoma Association. Glaucoma informtation. World Glaucoma Association; 2021. https://www.glaucomapatients.org/basic/statistics/.
Kapetanakis VV, Chan MPY, Foster PJ, Cook DG, Owen CG, Rudnicka AR. Global variations and time trends in the prevalence of primary open angle glaucoma (POAG): a systematic review and meta-analysis. Br J Ophthalmol. 2016;100:86–93.
King A, Azuara-Blanco A, Tuulonen A. Glaucoma. Br Med J. 2013;346:f3518. https://doi.org/10.1136/bmj.f3518.
Boyd K. What is glaucoma? American Academy of Ophthalmology; 2019. https://www.aao.org/eye-health/diseases/what-is-glaucoma.
Nayak BK, Maskati QB, Parikh R. The unique problem of glaucoma: under-diagnosis and over-treatment. Indian J Ophthalmol. 2011;559:S1–S2.
Arora T, Jindal Bali S, Arora V, Wadhwani M, Panda A, Dada T. Diurnal versus office-hour intraocular pressure fluctuation in primary adult onset glaucoma. J Optom. 2014;8:239–43.
Arend N, Hirneiss C, Kernt M. Differences in the measurement results of Goldmann applanation tonometry with and without fluorescein. Der Ophthalmol. 2014;111:241–6.
Jasien JV, Zohner YE, Asif SK, Rhodes LA, Samuels BC, Girkin CA, et al. Comparison of extraocular and intraocular pressure transducers for measurement of transient intraocular pressure fluctuations using continuous wireless telemetry. Sci Rep. 2020;10. https://doi.org/10.1038/s41598-020-77880-8.
Wilensky JT, Gieser DK, Mori MT, Langenberg PW, Zeimer RC. Self-tonometry to manage patients with glaucoma and apparently controlled intraocular pressure. JAMA Ophthamol. 1987;105:1072–5.
Pasquale LR, Kang JH. Lifestyle, nutrition and glaucoma. J Glaucoma. 2009;18:423–8.
Brandt JD. The myth of clinical precision. Am Acad Ophthalmol. 2009;116:1–2.
Dunbar GE, Shen BY, Aref AA. The Sensimed Triggerfish contact lens sensor: efficacy, safety, and patient perspectives. Clin Ophthalmol. 2017;11:875–82. https://doi.org/10.2147/OPTH.S109708.
Faschinger C, Mossböck G. 24-hour continuous monitoring of ocular hypertension using Triggerfish™ wireless contact lens sensor. Ophthalmologist. 2010;107:918–22.
Pierscionek BK, Asejczyk‐Widlicka M, Scha RA. The effect of changing intraocular pressure on the corneal and scleral curvatures in the fresh porcine eye. Br J Ophthalmol. 2007;91:801–3.
Douthwaite WA, Lam AKC. The effect of an artificially elevated intraocular pressure on the central corneal curvature. Ophthalmic Physiol Opt. 1997;17:18–24.
Hjortdal JO, Jensen PK. In vitro measurement of corneal strain, thickness, and curvature using digital image processing. Acta Ophthalmol. 1995;73:5–11.
Campigotto A, Ralhan A, Helgason R, Campbell RJ, Lai Y. Effect of variations of corneal physiology on novel non-invasive intraocular pressure monitoring soft contact lens. Biomed Microdevices. 2021;23. https://doi.org/10.1007/s10544-021-00555-2.
Shiku H, Saito T, Wu C-C, Yasukawa T, Yokoo M, Abe H, et al. Oxygen permeability of surface-modified poly(dimethylsiloxane) characterized by scanning electrochemical microscopy. Chem Lett. 2006;35:234–5.
Tran N-P-D, Yang M-C. The ophthalmic performance of hydrogel contact lenses loaded with silicone nanoparticles. Polymers. 2020;12:1128.
Lin C-H, Yeh Y-H, Lin W-C, Yang M-C. Novel silicone hydrogel based on PDMS and PEGMA for contact lens application. Colloids Surf B Biointerfaces. 2014;123:986–94. https://doi.org/10.1016/j.colsurfb.2014.10.053.
Childs A, Li H, Lewittes DM, Dong B, Liu W, Shu X, et al. Fabricating customized hydrogel contact lens. Sci Rep. 2016;6:1–9.
Campigotto A, Leahy S, Zhao G, Campbell JR, Lai Y. Non-invasive intraocular pressure monitoring with contact lens. Br J Ophthalmol. 2019;0:1–15. https://doi.org/10.1136/bjophthalmol-2018-313714.
Rufer F, Schroder A, Erb C. White-to-white corneal diameter: normal values in healthy Iranian population obtained with the Orbscan II. Int J Ophthalmol. 2014;7:309–12.
Jonuscheit S, Doughty MJ, Martin R, Rio-Cristobal A. Relationship between corneal thickness and radius to body height. Optom Vis Sci. 2017;94:380–6.
Brown N. The change in lens curvature with age. Exp Eye Res. 1974;19:175–83.
Lowe RF, Clark BAJ. Radius of curvature of the anterior lens surface. Br J Ophthalmol. 1973;57:471–4.
Acknowledgements
This research was funded by the National Science and Engineering Research Council of Canada and Queen’s University. The authors would like to thank Robert Ewart for his help with setting up the experimentation and data collection.
Author information
Authors and Affiliations
Contributions
Design of study completed by AC, YL, and RJC. Data collection, analysis, and writing completed by AC. Manuscript review completed by AC and YL.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Campigotto, A., Campbell, R.J. & Lai, Y. Correlation between corneal and contact lens deformation with changes in intraocular pressure for wearable monitoring systems. Eye 37, 2055–2060 (2023). https://doi.org/10.1038/s41433-022-02285-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-02285-y