Abstract
Purpose
To determine the distribution of central corneal thickness (CCT) and its determinants in an Iranian geriatric population.
Methods
This population-based study was conducted in 2019 in Tehran, the capital of Iran, using stratified multistage random cluster sampling. The study population was all residents ≥60 years of age. First, preliminary optometric and ocular health examinations were performed including the measurement of uncorrected and best-corrected visual acuity, objective and subjective refraction, anterior and posterior segment examination. The study participants then underwent corneal imaging using Pentacam HR.
Results
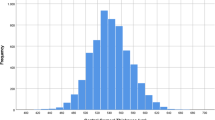
Out of 3791 invitees, 3310 participated in this study (response rate: 87.3%). The mean CCT and apex corneal thicknesses were 528 µ (95% CI: 526–529) and 529 µ (95% CI: 527–530), respectively. The highest and lowest mean corneal thickness was related to the superior (620 µ: 95% CI: 618–622) and the temporal (591 µ: 95% CI: 590–592) paracentral points, respectively. According to the multiple linear regression model, the CCT was significantly inversely related to keratometry readings (K1 and K2) and had a statistically significant direct relationship with intraocular pressure (IOP), corneal eccentricity (ECC), and corneal volume (CV) (all p values <0.05). The CCT was significantly higher in diabetic patients (p = 0.043).
Conclusion
The CCT values in the geriatric Iranian population were lower than the values reported in most previous studies. The CCT is mostly influenced by IOP and corneal parameters (curvature, shape factor, and volume) and is not affected by demographic factors, refractive error, and ocular biometric components.
Similar content being viewed by others
Introduction
Central corneal thickness (CCT) is an important parameter in ocular surgeries including cataract and refractive surgery as well as diagnosis of corneal pathologies especially keratoconus (KCN) [1]. Clinically, changes in CCT can be an indicative of the corneal endothelial cell dysfunction [2]. The CCT is also an influential factor in intraocular pressure measurement (IOP) and serves as a predictor in the progression of ocular hypertension (HTN) to primary open-angle glaucoma [3]. In cataract surgery, CCT measurement is important in some intraocular lens (IOL) power calculation formulas, especially in patients with a history of refractive surgery and also a history of KCN or other corneal abnormalities [4, 5].
The world community is aging [6]. Aging causes changes in the structure of various body tissues, including the eyes [6]. The age-related ocular changes have been studied in many studies. There are conflicting findings in the literature regarding age-related changes in CCT. Several studies have reported a decrease in CCT with age [7,8,9,10]. Some studies reported the stability of CCT from a young age onwards [11]. Due to the importance of CCT, various studies have examined its distribution in different populations. However, most previous studies were non-population-based and had a wide age range. It should be noted that no population-based study has specifically examined the distribution of CCT in the geriatric population. Also, racial differences can affect the distribution of CCT [7]. On the other hand, the related factors of CCT have not been definitively identified. The relationship between CCT and some demographic, ocular factors, and also systemic diseases such as age, sex, refractive errors, ocular biometric components, corneal curvature, diabetes, and hyperglycemia was studied in some previous studies but has been associated with conflicting results [12,13,14,15]. One possible reason for these discrepancies is that each of the previous studies examined a limited number of factors, and the confounding effects of different variables on each other were not controlled by a comprehensive multivariable model. According to the above, the present study aimed to determine the distribution of CCT and its related factors in an Iranian geriatric population.
Methods
Study population and sampling
The present study is part of the Tehran Geriatric Eye Study (TGES); a multi-purpose population-based study that was conducted in Tehran, the capital of Iran in 2019. The target population of TGES was all residents of Tehran above 60 years of age. The sampling was done using the stratified multistage random cluster sampling approach. A total of 165 clusters were randomly selected proportionally to size from 22 strata of Tehran. After identifying each cluster, a sampling team visited the cluster address to perform sampling. After explaining the objectives and steps of the study and ensuring the confidentiality of the information, all individuals over the age of 60 years were invited to participate in the study. This process continued until the sample size in each cluster was completed. When the interviewers went to the doors in each cluster, if the household was not at home, they returned at another time (preferably in the evening). The participants were taken to the examination site on a predetermined day free of charge. Once the study participants presented to the study site, first the signed consent was received, and then the complete demographic, socio-economic, and anthropometric information, as well as the history of previous eye examinations, ocular and systemic diseases, previous ocular surgery, and ocular trauma were collected by the trained interviewer. In the next stage, all participants underwent complete ocular examinations.
Examinations
The ocular examinations included preliminary optometric as well as ocular health examinations. The uncorrected distance visual acuity was measured by a LED visual acuity chart (Smart LC 13, Medizs Inc., Korea) at 6 meters (m). The objective refraction was performed using an auto-refractometer (ARK-510A, Nidek Co. LTD, Aichi, Japan). The best distance optical correction was determined using subjective refraction, and the best-corrected distance visual acuity was recorded. In the next step, complete anterior and posterior segment examination was performed using slit-lamp biomicroscopy (Slit-lamp B900, Haag-Streit AG, Bern, Switzerland), direct, and indirect ophthalmoscopy by an ophthalmologist.
In the next step, the study participants underwent corneal imaging using Pentacam HR (Oculus, Optikgeräte GmbH, Wetzlar, Germany) under dim lighting conditions by an experienced operator. The Pentacam is a high-resolution computerized topography system that is based on the Scheimflug principle. Using a blue light-emitting diode with a wavelength of 475 nm as the light source, corneal images are captured by a 1.45-megapixel camera that records up to 138,000 data points within 2 s. The instrument provides a three-dimensional image of the anterior segment as well as various data about the elevation, curvature, thickness, and shape factor of the cornea. Images were obtained using the instrument’s automatic mode. Only measurements were considered which had an “ok” quality statement. To minimize the potential effect of the tear film on the corneal imaging, patients were instructed to blink completely once before imaging. All imaging was done between 10 a.m. and 4 p.m. to account for the potential effects of diurnal variation. Data on pachymetric readings, keratometric readings (K1 and K2), anterior corneal eccentricity (ECC), total corneal volume (CV) in a diameter of 10 mm, anterior chamber depth (ACD), and white to white (WTW) distance were extracted and recorded from Pentacam’s maps. The axial length (AL) measurements were performed using IOL Master 500 (Carl Zeiss Meditec, Jena, Germany). The average of 3 AL measurements was considered as the final AL. The exclusion criteria were any corneal pathology, any anterior segment disease affecting the cornea, a history of corneal surgery, a history of ocular trauma, use of contact lenses at the time of the study, and use of drugs know to affect CCT including anti-glaucomatous medications.
Definitions
The CCT was defined based on the thickness at the pupil center. Also, the corneal thickness at the apex and four paracentral points (superior, inferior, temporal, and nasal on the 3 mm central ring) were extracted. Refractive errors were defined based on the spherical equivalent (SE) of manifest refraction. Myopia and hyperopia were defined as SE ≤−0.50 diopters and >+0.50 D, respectively. Diabetes mellitus (DM) was defined based on a previous diagnosis or hemoglobinA1c level of 6.5% or greater. HTN was defined based on a self-report or a systolic pressure ≥135 mm Hg or a diastolic pressure ≥85 mm Hg.
Statistical analysis
The mean values of corneal thickness with a 95% confidence interval (CI) were reported. The cluster sampling and design effect were considered in calculating the CIs. Simple and multiple linear regression models were used to investigate the relationship between CCT and the study variables. The multiple regression model was run using backward-stepwise method and only statistically significant variables were maintained in the final model. A p value <0.05 was considered statistically significant.
Results
Out of 3791 invitees, 3310 participated in this study (response rate: 87.3%). After applying the exclusion criteria and removing outlier data (values outside of 3 standard deviations from the mean), the final analysis was done on 5110 eyes of 2692 individuals. Of these, 1632 (60.6%) were female and the mean age of the participants was 67.9 ± 6.2 years (range: 60–97 years). The mean (95% CI) corneal thickness at the center, apex, as well as paracentral superior, inferior, temporal, and nasal points are shown in Table 1 according to the age, sex, DM, HTN, and refractive errors groups. The mean CCT and apex corneal thicknesses were 528 µ (95% CI: 526–529) and 529 µ (95% CI: 527–530), respectively. Table 2 shows 25, 75, and 95%, percentiles of corneal thickness in the center, apex, and at different paracentral points. The highest and lowest mean corneal thickness was related to the superior and the temporal paracentral points, respectively. The repeated measures analysis of variance showed a statistically significant difference between these four paracentral points (p < 0.001). Due to the high correlation of mean CCT and apex corneal thickness (r = 0.996), the analytical analysis was done only for CCT.
Table 3 shows the results of the simple linear regression to explore the relationship between CCT and any of the study variables. As seen in Table 3, there was no statistically significant difference in mean CCT between males and females (p = 0.567). Considering the age group 60–64 years as a reference, except for the age group 75–79 years (p = 0.255), other groups showed statistically significant lower CCT values, and the largest difference was related to the age group ≥80 years. The simple linear regression model also showed that individuals with HTN had a significantly lower mean CCT (p = 0.018) and the mean CCT was significantly higher in diabetic patients (p = 0.004). Myopes also had lower mean CCT compared to emmetropes (p = 0.005). Evaluation of the relationship between biometric and other ocular indices with CCT in the simple regression model showed that IOP, ECC, and CV, had a statistically significant direct relationship with CCT (all p values < 0.001). On the other hand, the WTW distance, ACD, K1 and K2 were significantly inversely related to CCT (all p values < 0.05).
The association between CCT and all studied variables was evaluated in a backward-stepwise linear multiple regression model. The results of the final adjusted model are shown in Table 3. As seen in Table 3, the CCT was significantly inversely related to keratometry readings (K1 and K2) and had a statistically significant direct association with IOP, ECC, and CV (all p values < 0.001). The CCT was also higher in diabetic patients (p = 0.043). Other variables including age, HTN, myopia, ACD, and WTW distance that had a statistically significant relationship with CCT in the univariate analysis did not maintain their significant association with CCT in the multiple regression model and removed from the final adjusted model (all p values > 0.05).
Discussion
The present study is the first population-based study to examine the distribution of CCT and its determinants specifically in the geriatric population over 60 years of age. According to the results, the mean CCT was 528 µ. Table 4 shows a list of previous studies in this field. This list includes studies that targeted older adults. As shown in Table 4, the mean CCT has been reported in a range of 511–555 µ in different studies [2, 16,17,18,19,20,21,22,23,24,25,26,27]. A comparison of the findings shows that the mean CCT in the present study was lower than in most previous studies. Various factors may influence these differences, including differences in age distribution, measurement tools, and also racial factors. As seen in Table 4, the mean age of the participants in the present study was higher compared to previous studies. Most previous studies used Pentacam and ultrasound pachymetry (US) to measure CCT. According to the literature, despite the high correlation of CCT measurements between Pentacam and the US, these measurements are not interchangeable and Pentacam tends to overestimate CCT compared to the US [28].
In the present study, the relationship between CCT and different demographic and ocular factors was also investigated. Accordingly, no statistically significant difference was found in the mean CCT between males and females. There are conflicting results regarding the association between sex and CCT in the literature. Some studies in line with the present study did not show a significant relationship between sex and CCT [18, 29, 30]. Some other studies have reported sex-related differences in CCT [2, 31, 32]. Some studies reported higher mean CCT in women, whereas others found higher CCT values in men. The cause of these sex-related differences in CCT has been linked to hormonal issues [33]. The lack of relationship between sex and CCT in the present study may be due to the effective control of confounders and also a decrease in hormonal levels considering the age distribution of study participants.
The results of the present study indicated no significant relationship between age and CCT. Although this relationship was statistically significant in the univariate analysis, it did not reach a significant level in the multiple regression model. This means that the significant relationship between age and CCT in univariate analysis is due to the effect of confounding variables, which was controlled in the multiple model. The relationship between age and CCT is also associated with conflicting results in the literature. Some studies in line with the present study did not show a significant relationship between age and CCT [34,35,36]. On the other hand, some studies reported a decrease in CCT with age, so that an age-related decrease in CCT at a rate of 3 to 7 µ per decade has been reported in some racial groups [23, 37,38,39]. This finding has been attributed to a decrease in keratocyte density and possible destruction of collagen fibers [22]. We believe that different factors may affect the relationship between CCT and age, the degree of control of which has led to inconsistencies between different studies. Changes in CCT may also follow different patterns over time, and the extent of these changes may be more pronounced in some stages. Given the age range of the participants in the present study, it is likely that age-related changes in CCT are not noticeable after 60 years of age. Also, controlling the confounding effect of different variables may be another contributing factor. It seems that long-term longitudinal studies are needed to definitively discover the relationship between age and CCT.
The association between DM and CCT was significant in the present study. Various studies have examined this relationship and reported variable results. Some studies, such as those of Wiemer et al. [40], Keoleian et al. [41], Inoue et al. [42], and Canan et al. [43], showed no significant association between DM and CCT. Other studies, such as the studies by Lee et al. [44], Su et al. [45], Roszkowska et al. [46], and Ozdamar et al. [47], reported an increased CCT in diabetic patients. Studies have also shown that the CCT varies at different stages of retinopathy, and increased CCT has also been reported as a risk factor for the progression of diabetic retinopathy [48]. Changes in CCT in diabetic patients appear to be mainly due to the effects of chronic metabolic stress on the corneal epithelium and endothelium [45, 49]. In diabetic patients, corneal endothelial dysfunction and consequent increased corneal hydration may increase CCT [50]. Altered epithelial barrier function can also increase CCT by causing some degree of corneal edema [51]. In our opinion, the differences between the studies in this field are due to the differences in the duration of DM and the level of DM control (blood glucose levels). However, this finding is clinically important and should be considered in measuring the CCT and related clinical settings (especially IOP measurement). Regarding the relationship between HTN and CCT in the present study, although univariate analysis showed a statistically significant inverse relationship, this relationship was not observed in the multiple regression model in the presence of other variables. Therefore, the results of the present study indicated no significant effect of HTN on CCT. Most previous studies in line with the present study did not found a significant relationship between HTN and CCT [27, 31].
Another finding of this study was the significant relationship between CCT and IOP. Based on the results, CCT showed an average increase of 0.64 per mm Hg of increase in IOP. The relationship between CCT and IOP is a relatively well-known finding and several studies have consistently pointed to a significant direct relationship between these two parameters [52,53,54]. In healthy corneas, CCT is related to the corneal rigidity, and this affects the amount of force required to flatten the measurement area in the applanation tonometry [55]. An interesting feature of the present study is that this relationship was investigated in a comprehensive multivariable model and by controlling the effects of different demographic and ocular variables.
The results of the present study showed a significant inverse relationship between CCT and keratometric readings; increased corneal curvature (steeper values) was associated with a decrease in CCT. According to the results, one diopter increase of K1 and K2 was associated with a 1.86 and 3.47 µ decrease in CCT, respectively. The relationship between CCT and corneal curvature was considered in some studies. However, most previous studies used univariate analysis for this purpose or only mentioned the correlation between these two parameters. Accordingly, most previous studies, including those of Ucakhan et al. [56], Torres et al. [57], Cho et al. [35], Iyamu et al. [23], Chen et al. [20], Eysteinsson et al. [58], Casson et al. [59], and Doughty et al. [60], showed no significant association between CCT and corneal curvature. We found three studies that similarly found a significant negative relationship between CCT and corneal curvature. In the study by Almahmoud et al., a significant inverse relationship was observed between the mean keratometry and CCT in the multiple model; age, sex, and SE were covariates (coefficient = −2.08) [14]. A similar finding was observed in the study by Shimmyo et al. based on simple linear regression [61]. Krishnan et al. also showed a statistically significant correlation between the corneal radius of curvature (in mm) and CCT (r = 0.269) [62].
In the present study, an increase in CV was significantly associated with an increase in CCT. We found only one study in this regard in the literature. In this study conducted by Hashemi et al., a similarly significant direct relationship was found between CCT and CV, however the regression coefficient was significantly lower compared to the present study (0.41 vs. 7.66) [2]. CV affects the biomechanical properties of the cornea including corneal hysteresis and corneal resistant factor, so that higher values of these two indices have been reported in corneas with higher CV [63]. On the other hand, these biomechanical characteristics are also directly related to CCT [64]. Therefore, examining the relationship between CCT and CV becomes relevant. Due to the limited information in this field in the literature, further studies are needed to confirm or refute this finding. This relationship should be considered in interpreting studies of factors affecting the corneal biomechanical properties.
An increase in ECC value (more prolate cornea) was significantly associated with an increase in CCT. It was noteworthy that the highest regression coefficient in the final model belonged to ECC so that the CCT increased 23.74 microns on average per unit of ECC increase. To our knowledge, this is the first time that the effect of corneal shape factor on the CCT has been studied. This association is difficult to justify and may have its origins in the geometric structure of the cornea. It is recommended that this relationship be further considered in future studies.
The results of the present study did not show a significant relationship between CCT and refractive errors. Although there was a statistically significant inverse relationship between myopia and CCT in univariate analysis, the results of multiple regression showed no significant relationship. The association between CCT and refractive errors has also been associated with conflicting results in the literature. Most previous studies failed to demonstrate a significant relationship between CCT and refractive errors [20, 29, 30, 35, 65]. Other studies reported significantly thinner corneas in myopic patients with longer ALs [66, 67]. An important reason for the discrepancy among the studies could be the difference in the degree of control over the effect of confounding variables. Also, in the present study, it is expected that part of myopia has a lenticular origin considering the age distribution of study participants.
In conclusion, the results of the present study showed that the CCT values in the geriatric Iranian population were lower than the values reported in most previous studies. The CCT was significantly directly related to IOP, ECC, and CV and had an indirect association with keratometry readings. Diabetic patients had significantly lower mean CCT.
Summary
What was known before
-
Some studies reported the stability of CCT from a young age onwards.
-
Due to the importance of CCT, various studies have examined its distribution in different populations.
-
However, most previous studies were non-population-based and had a wide age range.
What this study adds
-
The CCT values in the geriatric Iranian population were lower than the values reported in most previous studies.
-
The CCT is mostly influenced by IOP and corneal parameters (curvature, shape factor, and volume) and is not affected by demographic factors, refractive error, and ocular biometric components.
References
Chaudhry IA. Measurement of central corneal thickness in health and disease. Saudi J Ophthalmol. 2009;23:179–80.
Hashemi H, Yekta A, Saatchi M, Moravej R, Aghamirsalim M, Khabazkhoob M. The distribution of corneal thickness in rural population. Acta Med Iran. 2018;56:320–8.
Jonas JB, Stroux A, Oberacher-Velten IM, Kitnarong N, Juenemann A. Central corneal thickness and development of glaucomatous optic disk hemorrhages. Am J Ophthalmol. 2005;140:1139–41.
Seitz B, Langenbucher A. Intraocular lens calculations status after corneal refractive surgery. Curr Opin Ophthalmol. 2000;11:35–46.
Camps VJ, Piñero DP, Caravaca E, De Fez D. Preliminary validation of an optimized algorithm for intraocular lens power calculation in keratoconus. Indian J Ophthalmol. 2017;65:690–9.
Chader GJ, Taylor A. Preface: the aging eye: normal changes, age-related diseases, and sight-saving approaches. Invest Ophthalmol Vis Sci. 2013;54:ORSF1–4.
Aghaian E, Choe JE, Lin S, Stamper RL. Central corneal thickness of Caucasians, Chinese, Hispanics, Filipinos, African Americans, and Japanese in a glaucoma clinic. Ophthalmology. 2004;111:2211–9.
Weizer JS, Stinnett SS, Herndon LW. Longitudinal changes in central corneal thickness and their relation to glaucoma status: an 8 year follow up study. Br J Ophthalmol. 2006;90:732–6.
Rieth S, Engel F, Bühner E, Uhlmann S, Wiedemann P, Foja C. Comparison of data from the rostock cornea module of the heidelberg retina tomograph, the oculus pentacam, and the endothelial cell microscope. Cornea. 2010;29:314–20.
Hashemi H, Asgari S, Emamian MH, Mehravaran S, Fotouhi A. Five year changes in central and peripheral corneal thickness: the Shahroud eye cohort study. Cont Lens Anterior Eye. 2016;39:331–5.
Costantini E, Touzeau O, Gaujoux T, Basli E, Kopito R, Borderie M, et al. Age-related changes in central and peripheral corneal thickness. Inveset Ophthalmol Vis Sci. 2009;50:5107.
Su D, Wong TY, Foster P, Tay WT, Saw S-M, Aung T. Central corneal thickness and its associations with ocular and systemic factors: the Singapore Malay eye study. Am J Ophthalmol. 2009;147:709–16.
Hashmani N, Hashmani S, Hanfi AN, Ayub M, Saad CM, Rajani H, et al. Effect of age, sex, and refractive errors on central corneal thickness measured by Oculus Pentacam®. Clin Ophthalmol. 2017;11:1233–8.
AlMahmoud T, Priest D, Munger R, Jackson WB. Correlation between refractive error, corneal power, and thickness in a large population with a wide range of ametropia. Invest Ophthalmol Vis Sci. 2011;52:1235–42.
Wang L, Liu S, Wang W, He M, Mo Z, Gong X, et al. Association between ocular biometrical parameters and diabetic retinopathy in Chinese adults with type 2 diabetes mellitus. bioRxiv. 2020. https://doi.org/10.1101/2020.02.06.937144.
Hashemi H, Asgari S, Mehravaran S, Emamian MH, Shariati M, Fotouhi A. The distribution of corneal thickness in a 40-to 64-year-old population of Shahroud, Iran. Cornea. 2011;30:1409–13.
Rashid RF, Farhood QK. Measurement of central corneal thickness by ultrasonic pachymeter and oculus pentacam in patients with well-controlled glaucoma: hospital-based comparative study. Clin Ophthalmol. 2016;10:359–64.
Rüfer F, Schröder A, Bader C, Erb C. Age-related changes in central and peripheral corneal thickness: determination of normal values with the orbscan II topography system. Cornea. 2007;26:1–5.
Landers JA, Billing KJ, Mills RA, Henderson TR, Craig JE. Central corneal thickness of indigenous Australians within Central Australia. Am J Ophthalmol. 2007;143:360–2.
Chen MJ, Liu YT, Tsai CC, Chen YC, Chou CK, Lee SM. Relationship between central corneal thickness, refractive error, corneal curvature, anterior chamber depth and axial length. J Chin Med Assoc. 2009;72:133–7.
Hahn S, Azen S, Ying-Lai M, Varma R. Central corneal thickness in Latinos. Invest Ophthalmol Vis Sci. 2003;44:1508–12.
Galgauskas S, Juodkaite G, Tutkuvienė J. Age-related changes in central corneal thickness in normal eyes among the adult Lithuanian population. Clin Inter Aging. 2014;9:1145–51.
Iyamu E, Osuobeni E. Age, gender, corneal diameter, corneal curvature and central corneal thickness in Nigerians with normal intra ocular pressure. J Optom. 2012;5:87–97.
Gao X, Gauderman WJ, Liu Y, Marjoram P, Torres M, Haritunians T, et al. A genome-wide association study of central corneal thickness in Latinos. Invest Ophthalmol Vis Sci. 2013;54:2435–43.
Tomidokoro A, Araie M, Iwase A, Group TS. Corneal thickness and relating factors in a population-based study in Japan: the Tajimi study. Am J Ophthalmol. 2007;144:152–4.
Nemesure B, Wu SY, Hennis A, Leske MC. Corneal thickness and intraocular pressure in the Barbados eye studies. Arch Ophthalmol. 2003;121:240–4.
Sng C, Barton K, Kim H, Yuan S, Budenz DL. Central corneal thickness and its associations with ocular and systemic factors in an urban West African population. Am J Ophthalmol. 2016;169:268–75.
Al-Ageel S, Al-Muammar AM. Comparison of central corneal thickness measurements by Pentacam, noncontact specular microscope, and ultrasound pachymetry in normal and post-LASIK eyes. Saudi J Ophthalmol. 2009;23:181–7.
Zhang H, Xu L, Chen C, Jonas JB. Central corneal thickness in adult Chinese. Association with ocular and general parameters: the Beijing Eye Study. Graefe’s Arch Clin Exp Ophthalmol. 2008;246:587–92.
Hashemi H, Yazdani K, Mehravaran S, Khabazkhoob M, Mohammad K, Parsafar H, et al. Corneal thickness in a population-based, cross-sectional study: the Tehran Eye Study. Cornea. 2009;28:395–400.
Brandt JD, Beiser JA, Kass MA, Gordon MO, OHTS Group. Central corneal thickness in the ocular hypertension treatment study (OHTS). Ophthalmology. 2001;108:1779–88.
Suzuki S, Suzuki Y, Iwase A, Araie M. Corneal thickness in an ophthalmologically normal Japanese population. Ophthalmology. 2005;112:1327–36.
Giuffre G, Di Rosa L, Fiorino F, Bubella DM, Lodato G. Variations in central corneal thickness during the menstrual cycle in women. Cornea. 2007;26:144–6.
Prasad A, Fry K, Hersh PS. Relationship of age and refraction to central corneal thickness. Cornea. 2011;30:553–5.
Cho P, Lam C. Factors affecting the central corneal thickness of Hong Kong-Chinese. Curr Eye Res. 1999;18:368–74.
Nomura H, Ando F, Niino N, Shimokata H, Miyake Y. The relationship between age and intraocular pressure in a Japanese population: the influence of central corneal thickness. Curr Eye Res. 2002;24:81–5.
Lin N. Central corneal thickness (CCT) decreases with age. Invest Ophthalmol Vis Sci. 2007;48:4332.
Kadhim YJ, Farhood QK. Central corneal thickness of Iraqi population in relation to age, gender, refractive errors, and corneal curvature: a hospital-based cross-sectional study. Clin Ophthalmol. 2016;10:2369–76.
Hashemian H, Lin S, Moghimi S, Torabi H, Amini H. Central corneal thickness in primary angle closure and open angle glaucoma. J Ophthalmic Vis Res. 2014;9:439–43.
Wiemer NG, Dubbelman M, Kostense PJ, Ringens PJ, Polak BC. The influence of chronic diabetes mellitus on the thickness and the shape of the anterior and posterior surface of the cornea. Cornea. 2007;26:1165–70.
Keoleian GM, Pach JM, Hodge DO, Trocme SD, Bourne WM. Structural and functional studies of the corneal endothelium in diabetes mellitus. Am J Ophthalmol. 1992;113:64–70.
Inoue K, Kato S, Inoue Y, Amano S, Oshika T. The corneal endothelium and thickness in type II diabetes mellitus. Japanese. J Ophthalmol. 2002;46:65–9.
Canan H, Sahinoglu-Keskek N, Altan-Yaycioglu R. The relationship of central corneal thickness with the status of diabetic retinopathy. BMC Ophthalmol. 2020;20:1–7.
Lee J, Oum B, Choi H, Lee J, Cho B. Differences in corneal thickness and corneal endothelium related to duration in diabetes. Eye. 2006;20:315–8.
Su D, Wong TY, Wong W-L, et al. Diabetes, hyperglycemia, and central corneal thickness: the Singapore Malay Eye Study. Ophthalmology. 2008;115:964–8.
Roszkowska AM, Tringali CG, Colosi P, Squeri CA, Ferreri G. Corneal endothelium evaluation in type I and type II diabetes mellitus. Ophthalmologica. 1999;213:258–61.
Ozdamar Y, Cankaya B, Ozalp S, Acaroglu G, Karakaya J, Özkan SS. Is there a correlation between diabetes mellitus and central corneal thickness? J Glaucoma. 2010;19:613–6.
Zengin MÖ, Özbek Z, Arıkan G, Durak İ, Saatci A. Does central corneal thickness correlate with haemoglobin A1c level and disease severity in diabetes type II? Turk J Med Sci. 2010;40:675–80.
Calvo-Maroto AM, Perez-Cambrodí RJ, Albarán-Diego C, Pons A, Cerviño A. Optical quality of the diabetic eye: a review. Eye. 2014;28:1271–80.
Briggs S, Osuagwu UL, AlHarthi EM. Manifestations of type 2 diabetes in corneal endothelial cell density, corneal thickness and intraocular pressure. J Biomed Res. 2016;30:46–51.
Gekka M, Miyata K, Nagai Y, Nemoto S, Sameshima T, Tanabe T, et al. Corneal epithelial barrier function in diabetic patients. Cornea. 2004;23:35–7.
Foster PJ, Baasanhu J, Alsbirk PH, Munkhbayar D, Uranchimeg D, Johnson GJ. Central corneal thickness and intraocular pressure in a Mongolian population. Ophthalmology. 1998;105:969–73.
Wolfs RC, Klaver CC, Vingerling JR, Grobbee DE, Hofman A, Jong PT. Distribution of central corneal thickness and its association with intraocular pressure: the Rotterdam study. Am J Opthalmol. 1997;123:767–72.
Vijaya L, George R, Arvind H, Ve Ramesh S, Baskaran M, Raju P, et al. Central corneal thickness in adult south Indians: the Chennai glaucoma study. Ophthalmology. 2010;117:700–4.
Kotecha A, White ET, Shewry JM, Garway-Heath DF. The relative effects of corneal thickness and age on Goldmann applanation tonometry and dynamic contour tonometry. Br J Ophthalmol. 2005;89:1572–75.
Uçakhan OO, Gesoğlu P, Ozkan M, Kanpolat A. Corneal elevation and thickness in relation to the refractive status measured with the Pentacam Scheimpflug system. J Cataract Refract Surg. 2008;34:1900–5.
Torres RJ, Jones E, Edmunds B, Becker T, Cioffi GA, Mansberger SL. Central corneal thickness in Northwestern American Indians/Alaskan Natives and comparison with White and African-American persons. Am J Ophthalmol. 2008;146:747–51.
Eysteinsson T, Jonasson F, Sasaki H, Arnarsson A, Sverrisson T, Sasaki K, et al. Central corneal thickness, radius of the corneal curvature and intraocular pressure in normal subjects using non-contact techniques: Reykjavik Eye study. Acta Ophthalmol Scand. 2002;80:11–15.
Casson RJ, Abraham LM, Newland HS, Muecke J, Sullivan T, Selva D, et al. Corneal thickness and intraocular pressure in a nonglaucomatous burmese population: the Meiktila eye study. Arch Ophthalmol. 2008;126:981–5.
Doughty MJ, Laiquzzaman M, Müller A, Oblak E, Button NF. Central corneal thickness in European (white) individuals, especially children and the elderly, and assessment of its possible importance in clinical measures of intra-ocular pressure. Ophthalmic Physiol Opt. 2002;22:491–504.
Shimmyo M, Ross AJ, Moy A, Mostafavi R. Intraocular pressure, Goldmann applanation tension, corneal thickness, and corneal curvature in Caucasians, Asians, Hispanics, and African Americans. Am J Ophthalmol. 2003;136:603–13.
Krishnan V, Jayalatha K, Vijayakumar C. Correlation of central corneal thickness and keratometry with refraction and axial length: a prospective analytic study. Cureus. 2019;11:e3917.
Doostdar A, Nabovati P, Soori H, Rafati S, Naghdi T, Khabazkhoob M. Corneal biomechanical characteristics and their correlation in an Iranian adult myopic population. Func Diasabil J. 2018;1:9–18.
Mangouritsas G, Morphis G, Mourtzoukos S, Feretis E. Association between corneal hysteresis and central corneal thickness in glaucomatous and non‐glaucomatous eyes. Acta Ophthalmol. 2009;87:901–5.
Tong L, Saw SM, Siak JK, Gazzard G, Tan D. Corneal thickness determination and correlates in Singaporean schoolchildren. Invest Ophthalmol Vis Sci. 2004;45:4004–9.
Chang SW, Tsai IL, Hu FR, Lin LL-K, Shih YF. The cornea in young myopic adults. Br J Ophthalmol. 2001;85:916–20.
Suzuki S, Suzuki Y, Iwase A, Araie M. Corneal thickness in an ophthalmologically normal Japanese population. Ophthalmology. 2005;112:1327–36.
Funding
This project was supported by National Institute for Medical Research Development (NIMAD) affiliated with the Iranian Ministry of Health and Medical Education (grant code: 963660).
Author information
Authors and Affiliations
Contributions
Design and conduct of the study: HH and MK; collection, management of the data: MK, AY, and PN; analysis, and interpretation of the data: MK, PN, HH, and MA; and preparation, review, and approval of the manuscript: HH, MK, AY, PN, FR, and MA.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The tenets of the Declaration of Helsinki were considered in all stages of the study. The protocol of the study was approved by the Ethics Committee of National Institute for Medical Research Development (NIMAD) affiliated with the Iranian Ministry of Health. Informed consent was obtained from all participants (grant code: 963660).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hashemi, H., Nabovati, P., Aghamirsalim, M. et al. Central corneal thickness and its determinants in a geriatric population: a population-based study. Eye 37, 427–433 (2023). https://doi.org/10.1038/s41433-022-01946-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-01946-2
This article is cited by
-
Associations between corneal curvature and other anterior segment biometrics in young myopic adults
Scientific Reports (2024)