Abstract
Purpose
To report the disease pattern, progression and imaging characteristics in eyes with bilateral central serous chorioretinopathy (CSCR).
Methods
This was a retrospective case review of bilateral CSCR patients with active disease in at least one eye. Multimodal imaging including fundus photography, fundus autofluorescence, optical coherence tomography (OCT), fluorescein and indocyanine angiography (FA/ICGA) was done at baseline and follow-up visits. Disease classification was done using recently described classification criteria. The degree of asymmetry in the disease distribution pattern at baseline and disease progression during follow-up visit with a minimum duration of 12 months was studied.
Results
Among 103 CSCR patients, 36 patients (34.95%) with mean age of 53.6 ± 10.5 years had bilateral CSCR at baseline. Five patients (13.9%) had asymmetrical disease i.e. simple in one eye and complex in fellow eye. The remaining 31 patients had symmetric disease (simple, 2; complex 29). Mean duration of follow up was 17.58 ± 13.84 months. There was no significant difference between both eye parameters at last follow up (best corrected visual acuity, BCVA; central macular thickness, CMT; and subfoveal choroidal thickness, SFCT) (all p > 0.05). At last follow up, 22 eyes (2 simple and 20 complex) remained active whereas none of the eyes converted from simple to complex CSCR.
Conclusion
Bilateral disease was more commonly seen with complex CSCR in contrast to simple CSCR. Moreover, disease distribution in complex CSCR had symmetric pattern if bilateral disease was present. None of the simple CSCR eyes converted to complex type.
Similar content being viewed by others
Background
Central serous chorioretinopathy (CSCR) is a commonly encountered chorioretinal disease in clinical practice [1]. It usually manifests as unilateral neurosensory detachment with or without pigment epithelial detachment without any intraocular inflammation. However, CSCR can also present with a variety of manifestations with bilateral presentation such as chronic or atypical CSCR and masquerade as other chorioretinal disorders [2,3,4].
The recent knowledge in CSCR especially related to choroidal vascular hyperpermeability in uninvolved fellow eyes suggest bilateral, diffuse vascular changes [1]. Previous reports support this observation with 27–40% of CSCR cases showing bilateral involvement either concurrently or during subsequent follow up [1, 5, 6]. Quantifiable parameters such as choroidal thickness and vascularity index are also increased in fellow eyes of CSCR [7, 8]. Moreover, terminologies like pachychoroid morphology and pachychoroid pigment epitheliopathy (PPE) described for eyes with unilateral CSCR eyes with retinal pigment epithelium (RPE) changes in fellow eye also are suggestive of subclinical disease [7,8,9,10].
Common chorioretinal disorders such as age-related macular degeneration (AMD), and diabetic macular oedema (DMO) also show bilateral involvement with almost similar progression in both eyes over long term [11,12,13,14]. Whether the course of illness in CSCR follows asymmetrical pattern in both eyes or each eye behaves independent of fellow eye has not been elucidated in detail. Our group has recently proposed a new classification of CSCR incorporating terminologies such as simple and complex CSCR [15]. Moreover, other variables useful in predicting the visual and anatomical outcomes such as persistent disease, macular neovascularization (MNV), outer retinal changes, foveal involvement or presence of any atypical features i.e. bullous exudative fluid have been included in this classification [15]. We evaluated eyes with bilateral disease using this classification including both major disease subtypes (simple and complex) and report the presentation and disease outcomes in eyes with bilateral, active CSCR at 1 year follow up [15].
Methods
This was a multicentric, retrospective case review and included patients with bilateral CSCR with disease activity in at least one eye during the study period from January 2019 to December 2020. Active CSCR was defined as presence of subretinal fluid (SRF), and/or leakage on fluorescein angiography. Only eyes with good quality multimodal imaging were included for analysis. Institutional review board approval was obtained at each centre. This work was conducted in accordance with the tenets described in declaration of Helsinki. A written, informed consent was obtained from each patient.
CSCR was classified according to (i) the traditional classification as acute i.e. SRF ≤ 3 months or chronic and (ii) our recently proposed multimodal imaging based classification as simple or complex CSCR along with subcategorization (persistence, outer retinal atrophy (ORA), foveal involvement and MNV) [15]. In brief, simple CSCR was defined as area of RPE changes ≤2-disc area whereas >2-disc area or multifocal CSCR was classified as complex. The area of involvement was defined based on the multimodal imaging predominantly fundus autofluorescence. Furthermore, both subtypes were subdivided in primary (first known episode), recurrent (active disease with history or signs of previous episodes) and resolved (no disease activity with absence of SRF). Persistent disease included eyes with ≥6 months of SRF. Outer retinal atrophy (ORA) [thinning of outer nuclear layer (ONL) or ellipsoid zone (EZ) and external limiting membrane (ELM) disruption], MNV and involvement of fovea (with SRF/PED/ORA) were also included in the description. This was defined based on rarefaction or loss of ONL/EZ/ELM within central 1000 µm of line scan passing through fovea. Atypical variants such as bullous exudative retinal detachment and RPE tear was also accomodated [15]. Two masked observers classified CSCR based on new CSCR classification. The degree of concordance was later calculated using the kappa coefficient.
Relevant ocular and systemic history (including treatment history) was obtained. Patient with history of focal laser photocoagulation, photodynamic therapy (PDT), anti-vascular endothelial growth factors (VEGF) within 3 months from baseline visit were excluded from the study. Patients with PPE with no obvious CSCR changes were also excluded. Moreover, patients with another ocular comorbidity i.e. diabetic retinopathy, glaucoma, uveitis, vitreoretinal surgeries or any media opacities precluding fundus view were also not considered for the study.
Patients were followed up at 3, 6 and 12 months or as per the treating physician. The minimum duration of follow-up visits was 12 months. Best corrected visual acuity (BCVA) was assessed at both baseline and follow-up visits using Snellen chart. BCVA was later converted to logarithm of minimum angle of resolution (logMAR) for further analysis.
Multimodal imaging including fundus photography, autofluorescence, optical coherence tomography (OCT), fluorescein angiography and indocyanine angiography was performed at baseline and follow-up visits as per the discretion of the treating physician. OCT measurements included central macular thickness (CMT) and subfoveal choroidal thickness (SFCT). CMT was defined as distance between internal limiting membrane and RPE within central 1 mm of macula. SFCT was defined as the linear distance of outer border of RPE and choroidoscleral interface at the subfoveal level. The degree of symmetry based on the subtype of disease at baseline and final visit was studied. Changes in BCVA, CMT, SFCT were compared in right and left eye separately through last visit.
Statistical analysis
Statistical analysis was done using Statistical Package for the Social Sciences (SPSS, version 23, IBM, Armonk, NY, USA). Data were presented as mean ± standard deviation (SD). Normality of the data was determined using Shapiro–Wilk test. The linear mixed effects model with random intercept was used to compare the parameters including change in BCVA, CMT and SFCT at baseline and follow-up visits (3, 6 and 12 months and last follow up) in the entire cohort as well as compare right eye with left eye. Random intercept was used to adjust for inter-eye correlation and was same for both eyes of same patient. P value ≤ 0.05 was considered statistically significant.
Results
A total of 103 CSCR patients were initially evaluated among which 36 patients (34.95%) met the inclusion criteria were included in the final analysis. Mean age of study patients was 53.6 ± 10.5 years. Mean duration of disease was 39 ± 50 months. Thirty-eight eyes among these 72 eyes (52.8%) were treatment naïve and remaining eyes (47.2%) received either monotherapy or a combination of focal laser, PDT or anti vascular endothelial growth factors (anti-VEGF) agents at least 3 months prior to the inclusion (Table 1).
At the baseline, a total of 19 eyes (26.4%) (10 right and 9 left) had presence of acute CSCR. Similarly, 53 eyes (73.6%) (26 right and 27 left) had chronic CSCR. On the other hand, 9 eyes were classified as simple CSCR (6, right and 3, left) whereas remaining 63 eyes were designated as complex CSCR (30, right and 33, left) as shown in Table 1. Two patients (5.5%) had simple CSCR in both eyes at baseline whereas 29 patients (80.5%) showed complex CSCR bilaterally at baseline. Remaining 5 (13.9%) patients had asymmetrical disease i.e. simple, and complex disease in either eye. Mean duration of follow up of the entire cohort was 17.58 ± 13.84 months. On evaluation of the interobserver agreement between the two masked observers who classified the disease as per new CSCR classification, a high concordance rate was obtained (kappa = 0.91).
Overall, BCVA in the entire cohort improved from 0.45 ± 0.39 at baseline to 0.34 ± 0.36 logMAR at last follow up (p = 0.48). CMT improved from 367.8 ± 204.3 to 269.1 ± 125.1 µm (p = 0.01) whereas SFCT improved from 392.7 ± 142.9 to 324.0 ± 107.6 µm (p = 0.02) at last follow up visit (Table 2). Analysis of right eye (change in BCVA, p = 0.23; CMT, p = 0.12 and SFCT, p < 0.01) and left eye (change in BCVA, p = 0.36; CMT, p = 0.002 and SFCT, p = 0.01) was also done as shown in Table 2. There was no statistically significant difference between the parameters of two eyes through the last follow up (change in BCVA, p = 0.35; CMT, p = 0.96; SFCT, p = 0.44).
Among the 36 patients, 31 (86.1%) had active CSCR in one eye whereas remaining 5 patients (13.9%) had bilateral active CSCR at baseline. Out of 9 simple CSCR eyes at baseline (2 primary, 4 recurrent and 3 resolved), 7 eyes showed resolution (simple, resolved), and 2 eyes remained simple, recurrent CSCR (Table 3). A total of 63 eyes showed complex CSCR at baseline (13 primary, 22 recurrent and 28 resolved). At the last follow-up visit, 20 eyes remained recurrent (11 right, 9 left) and remaining 43 showed resolution of disease (19 right, 24 left). Twenty-two eyes remained active at last follow up (2 simple and 20 complex). None of the eyes with simple CSCR turned into complex over the follow up (Table 3).
At the last follow up, 18 eyes had persistent disease (SRF lasting ≥ 6 months). Three patients had bilateral persistent disease, whereas 12 eyes had asymmetrical i.e. resolved in one and persistent disease in fellow eye. A total of 21 eyes of 15 patients were treated with one session of PDT (12 right, 9 left) following which all except 6 eyes showed non-resolution. Nineteen eyes of 16 patients were treated with focal laser or micropulse laser photocoagulation. (11 right and 8 left eye). Nine patients received tablet eplerenone for a period of 3–4 months. Rest of the patients also received a combination of treatment including anti VEGF injections (48 injections). Representative cases are shown as Figs. 1 and 2.
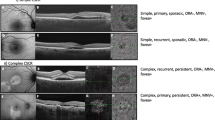
Fundus autofluorescence (FAF) of right (A) and left eye (D) shows multiple hyperautofluorescent areas with few interspersed hypoautofluorescent areas (left > right; more than 2DD, multifocal in both eyes) at the posterior pole. Optical coherence tomography (OCT) of right eye superior to fovea showed pocket of subretinal and intraretinal fluid (SRF/ IRF) (B) whereas foveal scan showed rarefaction of ellipsoid zone and no SRF (not shown). At 3 years follow up, post focal laser photocoagulation and one session of photodynamic therapy (PDT), there was persistent, shallow SRF superior to fovea (C). Subfoveal area showed no SRF at last follow up (not shown). OCT of left eye showed pockets of SRF subfoveally and nasal to fovea with shallow pigment epithelial detachment (PED) (E). Post focal laser and PDT, there was resolution of SRF with loss of outer retinal layers at 3 years follow up (F). Baseline diagnosis was recurrent, complex CSCR in both eyes whereas the final diagnosis was recurrent, complex (persistent) CSCR and resolved, complex CSCR in right and left eye, respectively.
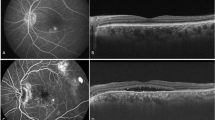
Fundus autofluorescence (FAF) of right (A) and left eye (C) shows hyper-autofluorescence inferior to fovea and disc (more than 2DD, multifocal) in right eye and perifoveal hyperfluorescence expending temporal to fovea (more than 2DD) corresponding to subretinal fluid (SRF) pocket in left eye. OCT of right eye (B) showed focal areas of outer retinal layers loss suggestive of previous episodes of CSCR. Fovea showed normal inner and outer retinal layers. OCT of left eye (D) showed large pocket of SRF involving fovea with serous pigment epithelial detachment. A diagnosis of resolved, complex CSCR and primary, complex CSCR was made.
Discussion
We analysed eyes with bilateral CSCR and reclassified them using our recent classification as simple and complex CSCR. A total of 36 patients (34.95%) among 103 patients had bilateral disease suggestive of the fact that ~1/3rd of all CSCR is bilateral. Among these 36 patients, 5 (13.9%) showed active disease bilaterally at baseline. Majority of the eyes (87.5%) were classified as complex CSCR disease while only 12.5% eye were classified as simple CSCR. This suggest that complex disease is usually bilateral whereas simple disease shows a unilateral predisposition.
Most patients (58.3%) showed asymmetrical disease i.e. active disease in one eye with fellow eye developing disease activity during subsequent visits. However, there was no significant difference in quantitative parameters i.e. BCVA, CMT, SFCT suggestive of the fact that course of illness in both eyes may not be different to a large extent. Although there were limited number of simple CSCR cases, none of them showed conversion to complex CSCR over a mean follow up of 17.58 ± 13.84 months.
There are multiple chorioretinal disorders showing bilateral distribution pattern such as AMD, DMO [11,12,13,14]. On similar lines, complex CSCR in one eye may have a bearing on the fellow eye leading to development of complex CSCR. It is currently unknown whether simple and complex are part of a disease continuum (i.e. simple CSCR can progress to complex CSCR) or separate diseases. Though no genetic transmission pattern has been conclusively proven, prior research has provided some evidence of genetic association such as complement factor H and a possible role of non-genetic factors in progression towards either acute or chronic CSCR [1, 16].
This study has few limitations especially related to the smaller sample size in acute/simple CSCR cases. Patients were not treatment naïve which may have played a role in asymmetricity and natural history of the disease. Moreover, mean duration of disease was 39 months which is relatively shorter. We did not evaluate other risk factors and their relationship with simple or complex disease. Multiple treatment modalities were used based on the discretion of treating physician which could have added an inherent bias. Treatment modalities may have effect on the natural course of disease. The efficacy of eplerenone in CSCR is not universally accepted and has been refuted in recent publications [17]. We have used our novel classification system. The basis of this classification was rectifying the discrepancies of the previous classification and adding multiple descriptors to add to provide adequate information about disease course and its prognosis.
In conclusion, complex CSCR have a higher preponderance of bilateral distribution whereas simple CSCR tends to be unilaterally distributed. Moreover, majority of complex CSCR show higher degree of symmetricity if bilateral disease is present. This information adds to further understand of their pathogenesis. A longer follow up will be useful in understanding the long-term changes. Moreover, further advanced imaging will help to understand subtle changes in fellow eyes of CSCR.
Summary
What was known before
-
Central serous chorioretinopathy (CSCR) is known to occur bilaterally in a third of patients.
-
Our recent classification defined simple (RPE changes ≤ 2-disc area) and complex CSCR (RPE changes > 2-disc area or multifocal) based on the area of extent of RPE involvement.
What this study adds
-
Bilateral disease is more commonly seen with complex CSCR in contrast to simple CSCR. If present, bilateral disease is symmetrical (with respect to disease subtypes) in complex CSCR cases.
-
The conversion rate of simple to complex CSCR over minimum of 12 months (mean duration 39 months) is low.
Data availability
Available upon request.
Code availability
Available upon request.
References
Daruich A, Matet A, Dirani A, Bousquet E, Zhao M, Farman N, et al. Central serous chorioretinopathy: recent findings and new physiopathology hypothesis. Prog Retin Eye Res. 2015;48:82–118. https://doi.org/10.1016/j.preteyeres.2015.05.003
Sahoo NK, Singh SR, Rajendran A, Shukla D, Chhablani J. Masqueraders of central serous chorioretinopathy. Surv Ophthalmol. 2019;64:30–44. https://doi.org/10.1016/j.survophthal.2018.09.001
Balaratnasingam C, Freund KB, Tan AM, Mrejen S, Hunyor AP, Keegan DJ, et al. Bullous Variant of Central Serous Chorioretinopathy: Expansion of Phenotypic Features Using Multimethod Imaging. Ophthalmology. 2016;123:1541–52. https://doi.org/10.1016/j.ophtha.2016.03.017
Hanumunthadu D, Tan ACS, Singh SR, Sahu NK, Chhablani J. Management of chronic central serous chorioretinopathy. Indian J Ophthalmol. 2018;66:1704–14. https://doi.org/10.4103/ijo.IJO_1077_18
Gäckle HC, Lang GE, Freissler KA, Lang GK. [Central serous chorioretinopathy. Clinical, fluorescein angiography and demographic aspects]. Ophthalmologe. 1998;95:529–33. https://doi.org/10.1007/s003470050310. Chorioretinopathia centralis serosa. Klinische, fluoreszeinangiographische und demographische Aspekte
Sahoo NK, Singh SR, Kammari P, Jonnadula GB, Das AV, Chhablani J. Prevalence and Profile of Central Serous Chorioretinopathy in an Indian Cohort. Nepal J Ophthalmol 2019;11:5–10. https://doi.org/10.3126/nepjoph.v11i1.25410
Agrawal R, Chhablani J, Tan KA, Shah S, Sarvaiya C, Banker A. Choroidal vascularity index in central serous chorioretinopathy. Retin (Phila, Pa). 2016;36:1646–51. https://doi.org/10.1097/iae.0000000000001040
Kim YT, Kang SW, Bai KH. Choroidal thickness in both eyes of patients with unilaterally active central serous chorioretinopathy. Eye 2011;25:1635–40. https://doi.org/10.1038/eye.2011.258. 2011/12/01
Warrow DJ, Hoang QV, Freund KB. Pachychoroid pigment epitheliopathy. Retina. 2013;33:1659–72. https://doi.org/10.1097/IAE.0b013e3182953df4
Singh SR, Matet A, van Dijk EHC, Daruich A, Fauser S, Yzer S, et al. Discrepancy in current central serous chorioretinopathy classification. Br J Ophthalmol. 2019;103:737–42. https://doi.org/10.1136/bjophthalmol-2018-312435
Gudnadottir GS, Magnusson KP, Stefansson E, Jonasson F, Helgadottir G, Sigurdsson H. The time pattern of bilateral exudative age-related macular degeneration. Acta Ophthalmol Scand. 2005;83:333–6. https://doi.org/10.1111/j.1600-0420.2005.00451.x
Joachim N, Colijn JM, Kifley A, Lee KE, Buitendijk GHS, Klein BEK, et al. Five-year progression of unilateral age-related macular degeneration to bilateral involvement: the Three Continent AMD Consortium report. Br J Ophthalmol. 2017;101:1185–92. https://doi.org/10.1136/bjophthalmol-2016-309729
Gonder JR, Walker VM, Barbeau M, Zaour N, Zachau BH, Hartje JR, et al. Costs and Quality of Life in Diabetic Macular Edema: Canadian Burden of Diabetic Macular Edema Observational Study (C-REALITY). J Ophthalmol. 2014;2014:939315 https://doi.org/10.1155/2014/939315
Mitchell P, Annemans L, Gallagher M, Hasan R, Thomas S, Gairy K, et al. Cost-effectiveness of ranibizumab in treatment of diabetic macular oedema (DME) causing visual impairment: evidence from the RESTORE trial. Br J Ophthalmol. 2012;96:688–93. https://doi.org/10.1136/bjophthalmol-2011-300726
Chhablani J, Cohen FB. Multimodal Imaging-Based Central Serous Chorioretinopathy Classification. Ophthalmol Retin. 2020;4:1043–6. https://doi.org/10.1016/j.oret.2020.07.026
Miki A, Kondo N, Yanagisawa S, Bessho H, Honda S, Negi A. Common variants in the complement factor H gene confer genetic susceptibility to central serous chorioretinopathy. Ophthalmology. 2014;121:1067–72. https://doi.org/10.1016/j.ophtha.2013.11.020
Lotery A, Sivaprasad S, O’Connell A, Harris RA, Culliford L, Ellis L, et al. Eplerenone for chronic central serous chorioretinopathy in patients with active, previously untreated disease for more than 4 months (VICI): a randomised, double-blind, placebo-controlled trial. Lancet 2020;395:294–303. https://doi.org/10.1016/s0140-6736(19)32981-2
Author information
Authors and Affiliations
Contributions
SRS, DCP, SA, JC were involved in the designing of the study. DCP, SA, DSM, NKS, ANK, CI, FT, NGR, RSP, were involved in the collection of data. SRS, SA, NKS, RV, EP, JC were responsible for interpretation and analysis of the data. SRS, DCP, DSM, NKS, FT, EP, JC were involved in paper writing. SRS, DCP, SA, DSM, NKS, ANK, CI, FT, RV, NGR, RSP, EP and JC reviewed the paper. All the authors conducted the study and equally contributed to the preparation, review and approval of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singh, S.R., Parameswarappa, D.C., Arora, S. et al. Imaging characteristics of bilateral CSCR cases:12 months follow up. Eye 37, 97–102 (2023). https://doi.org/10.1038/s41433-021-01885-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01885-4
This article is cited by
-
Biometric Risk Factors for Central Serous Chorioretinopathy
Ophthalmology and Therapy (2023)
-
Ten-year follow-up and sequential evaluation of multifocal retinal pigment epithelium abnormalities in central serous chorioretinopathy
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)